Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
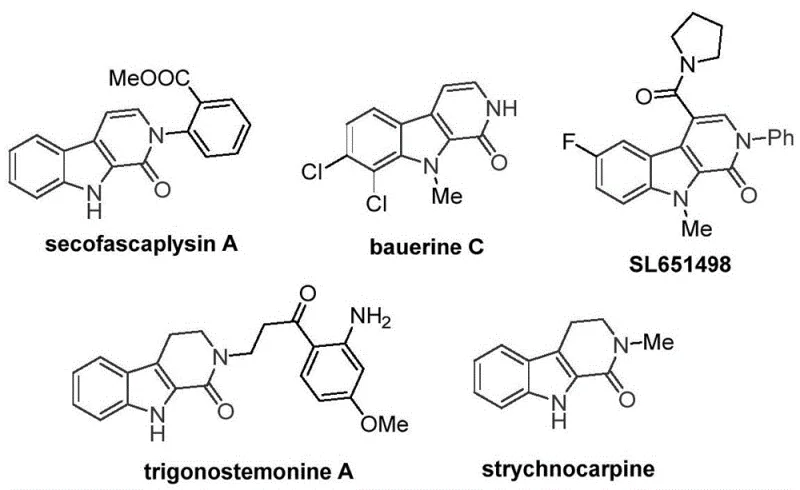
The tetrahydro-beta-carbolinone scaffold represents a privileged structural motif found extensively in bioactive natural products and pharmaceutical candidates, ranging from antiviral agents like bauerine C to anxiolytic drug candidates such as SL651498. Despite its therapeutic significance, the efficient construction of this core heterocycle has historically presented substantial synthetic challenges, often relying on precious metal catalysis or harsh conditions. A groundbreaking development disclosed in Chinese Patent CN115260188A introduces a robust, cobalt-catalyzed C-H activation carbonylation strategy that fundamentally reshapes the manufacturing landscape for these valuable intermediates. This innovation not only streamlines the synthetic route but also aligns perfectly with the industry's shift towards sustainable, base-metal catalysis, offering a reliable pharmaceutical intermediate supplier pathway for complex nitrogen-containing heterocycles.
Traditionally, the assembly of the tetrahydro-beta-carbolinone framework via carbonylation reactions has been dominated by palladium-catalyzed protocols. While effective, these conventional methods suffer from inherent limitations that hinder their widespread adoption in large-scale industrial settings. The primary drawback is the reliance on palladium, a scarce and exorbitantly expensive transition metal, which drastically inflates the raw material costs for cost reduction in API manufacturing. Moreover, palladium residues are strictly regulated in final drug substances due to their toxicity, necessitating complex and costly purification steps such as scavenging or recrystallization to meet stringent ppm limits. Additionally, many traditional carbonylation procedures require the use of gaseous carbon monoxide, a highly toxic and flammable gas that demands specialized high-pressure equipment and rigorous safety protocols, thereby complicating the commercial scale-up of complex pharmaceutical additives.
In stark contrast, the novel approach detailed in the patent leverages an earth-abundant cobalt catalyst system to drive the cyclization efficiently. By utilizing cobalt acetate tetrahydrate as the precatalyst in conjunction with silver carbonate as an oxidant and a phenolic ester as a solid carbon monoxide surrogate, this method circumvents the need for hazardous CO gas and expensive palladium complexes. The reaction operates under relatively mild thermal conditions (120-140°C) in dioxane, demonstrating exceptional functional group tolerance across a wide array of tryptamine derivatives. This shift to base-metal catalysis not only reduces the direct cost of the catalyst by orders of magnitude but also simplifies the downstream processing, as cobalt is generally easier to manage and remove compared to noble metals, presenting a transformative solution for high-purity OLED material and pharmaceutical intermediate production.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
The mechanistic pathway proposed for this transformation involves a sophisticated catalytic cycle initiated by the oxidation of the cobalt(II) species. Initially, the cobalt(II) acetate precursor is oxidized by silver carbonate to generate a reactive cobalt(III) species, which subsequently coordinates with the nitrogen atom of the tryptamine derivative substrate. This coordination is crucial as it directs the metal center to the proximal C-H bond at the 2-position of the indole ring, facilitating a directed C-H activation event that forms a stable five-membered cobaltacycle intermediate. This step is the turnover-limiting feature of the cycle and highlights the efficacy of the cobalt system in activating relatively inert C-H bonds without the need for pre-functionalized halide handles, a common requirement in cross-coupling chemistry.

Following the C-H activation, the key carbonylation step occurs through the insertion of carbon monoxide, which is released in situ from the decomposition of the 1,3,5-tricarboxylic acid phenol ester (TFBen). This insertion generates an acyl-cobalt(III) intermediate, effectively building the carbonyl functionality directly into the growing molecular framework. The cycle concludes with a reductive elimination step that forms the new C-C bond, closing the six-membered lactam ring characteristic of the tetrahydro-beta-carbolinone structure, followed by hydrolysis to release the final product and regenerate the active catalyst species. This elegant mechanism ensures high atom economy and minimizes the formation of side products, as the pyridine directing group installed on the starting material is designed to leave spontaneously during the reaction, further simplifying the impurity profile and enhancing the overall purity of the isolated compound.
How to Synthesize Tetrahydro-beta-carbolinone Efficiently
The experimental protocol outlined in the patent provides a straightforward and reproducible method for accessing these valuable scaffolds. The procedure involves charging a Schlenk tube with the cobalt catalyst, base, additive, oxidant, CO surrogate, and the specific tryptamine derivative in dioxane solvent. The mixture is then heated to the specified temperature range, allowing the cascade of C-H activation and carbonylation to proceed to completion. Detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Charge a reaction vessel with cobalt catalyst (Co(OAc)2·4H2O), base (Et3N), additive (PivOH), oxidant (Ag2CO3), CO surrogate (TFBen), and tryptamine derivative in dioxane.
- Heat the mixture to 120-140°C and stir for 16-24 hours under inert atmosphere to facilitate C-H activation and carbonylation.
- Upon completion, filter the mixture, concentrate, and purify the crude residue via silica gel column chromatography to isolate the pure tetrahydro-beta-carbolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from palladium-based to cobalt-based synthesis offers profound strategic benefits beyond mere chemical curiosity. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials; cobalt salts are commodity chemicals with stable pricing and abundant global availability, unlike palladium which is subject to volatile geopolitical supply shocks. By eliminating the dependence on precious metals, manufacturers can secure long-term supply continuity and hedge against the fluctuating costs associated with noble metal markets, ensuring a more predictable cost structure for long-term projects.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive cobalt acetate results in significant direct material cost savings. Furthermore, the use of a solid CO surrogate eliminates the capital expenditure and maintenance costs associated with high-pressure gas infrastructure and safety monitoring systems required for gaseous CO. The simplified workup procedure, which avoids complex heavy metal scavenging resins, further reduces the consumption of auxiliary materials and lowers the overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against various functional groups means that a single, standardized protocol can be applied to a diverse library of substrates without extensive re-optimization. This flexibility allows for rapid response to changing R&D demands and reduces the lead time for high-purity pharmaceutical intermediates. The use of commercially available and stable reagents ensures that production schedules are not disrupted by the scarcity of specialized ligands or sensitive catalysts often required in fine-tuned palladium cycles.
- Scalability and Environmental Compliance: From an environmental and safety perspective, replacing toxic CO gas with a solid surrogate significantly lowers the risk profile of the manufacturing process, facilitating easier regulatory approval for plant operations. The reaction demonstrates excellent scalability, having been validated on gram scales with high conversion rates, indicating a smooth path to kilogram and ton-scale production. Additionally, the reduced heavy metal load in the waste stream simplifies effluent treatment and aligns with increasingly strict environmental regulations regarding metal discharge in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this synthesis method for industrial partners.
Q: What are the advantages of using cobalt over palladium for this synthesis?
A: Cobalt is significantly more earth-abundant and cost-effective than palladium. Furthermore, using a base metal catalyst eliminates the stringent regulatory requirements and costly processing steps associated with removing toxic heavy metal residues from pharmaceutical intermediates.
Q: What is the role of 1,3,5-tricarboxylic acid phenol ester in this reaction?
A: This compound acts as a solid carbon monoxide (CO) surrogate. It safely releases CO in situ under the reaction conditions, avoiding the need for handling hazardous high-pressure CO gas cylinders, which greatly enhances operational safety and scalability.
Q: Can this method tolerate diverse functional groups on the indole ring?
A: Yes, the patent demonstrates excellent substrate compatibility. Various substituents including methyl, methoxy, chloro, bromo, benzyl, and allyl groups at different positions (R1 and R2) are well-tolerated, yielding products with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the cobalt-catalyzed pathway described in CN115260188A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to translate this laboratory-scale breakthrough into a robust, GMP-compliant manufacturing process, ensuring that our clients receive high-quality intermediates that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite potential partners to engage with our technical sales division to explore how this cost-effective synthesis route can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your project volume. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of efficiency and reliability in the industry.
