Scalable Synthesis of Cytotoxic Chiral Chroman Compounds via Organocatalysis for Oncology Applications
Introduction to Advanced Chiral Chroman Synthesis
The landscape of oncology drug discovery is constantly evolving, driven by the need for novel scaffolds that exhibit potent cytotoxicity against resistant adenocarcinoma cell lines. A significant breakthrough in this domain is documented in Chinese Patent CN112920173A, which discloses a robust and highly stereoselective method for synthesizing chiral chroman compounds. These heterocyclic structures are of paramount interest in medicinal chemistry due to their privileged status in biological systems, often serving as core motifs in antioxidants and anticancer agents. The patent specifically highlights a synthetic route that leverages asymmetric organocatalysis to construct complex chroman frameworks with exceptional control over stereochemistry. This technological advancement addresses critical pain points in traditional synthesis, such as poor enantiocontrol and harsh reaction conditions, thereby opening new avenues for the development of next-generation therapeutic intermediates. By utilizing a chiral phosphoric acid catalyst, the process achieves high levels of molecular precision, which is a non-negotiable requirement for modern active pharmaceutical ingredient (API) manufacturing.
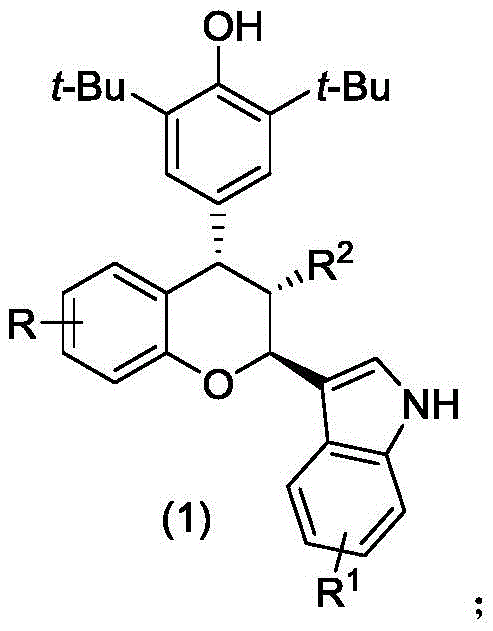
Furthermore, the biological evaluation data presented in the patent underscores the commercial viability of these compounds, demonstrating strong inhibitory effects on HepG2, MDA-MB-231, and SGC-7901 cell lines. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a dual opportunity: access to a high-value pharmacophore and a manufacturing process that aligns with green chemistry principles. The ability to generate structural diversity through simple substrate modification while maintaining high yields makes this technology a strategic asset for companies looking to expand their oncology pipeline. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies is essential for ensuring supply chain continuity and regulatory compliance in the production of complex small molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral chroman skeletons has been fraught with synthetic challenges that hinder efficient commercial production. Traditional approaches often rely on transition metal catalysis, which introduces significant risks regarding heavy metal contamination—a critical quality attribute that requires extensive and costly purification steps to mitigate. Moreover, many classical methods suffer from limited substrate scope, failing to accommodate diverse functional groups without compromising yield or stereoselectivity. The reaction conditions in older protocols can be excessively vigorous, involving high temperatures or strong acids that pose safety hazards and increase the likelihood of side reactions, leading to complex impurity profiles that are difficult to separate. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating bottlenecks for supply chain managers who need to deliver high-purity materials consistently. Additionally, the lack of robust enantioselective control in conventional routes often necessitates resolution steps, which inherently cap the maximum theoretical yield at 50%, representing a massive inefficiency in atom economy and resource utilization.
The Novel Approach
In stark contrast, the methodology described in CN112920173A offers a paradigm shift by employing a chiral phosphoric acid organocatalyst to drive the reaction between p-methylene benzoquinone and 3-indolene derivatives. This metal-free approach eliminates the risk of heavy metal residues, simplifying the downstream purification process and significantly reducing the environmental footprint of the manufacturing operation. The reaction proceeds under remarkably mild conditions, specifically at a controlled temperature of -30°C in dichloromethane, which minimizes thermal degradation of sensitive functional groups and ensures operator safety. The use of molecular sieves as additives further enhances the reaction efficiency by sequestering water, thereby driving the equilibrium towards product formation and improving overall yields. This novel strategy not only broadens the applicability of the synthesis to a wide range of substituted substrates but also delivers products with superior diastereomeric and enantiomeric ratios. For procurement teams, this translates to a more predictable and stable supply of high-quality intermediates, while R&D teams benefit from a versatile platform capable of generating diverse libraries for structure-activity relationship (SAR) studies.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
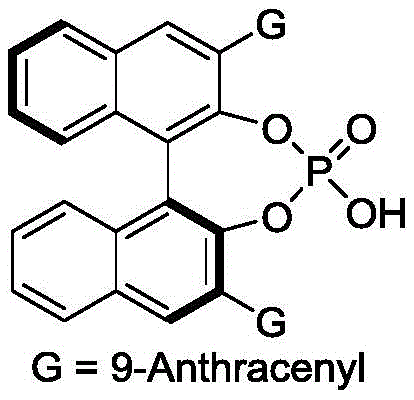
The success of this synthesis hinges on the precise mechanistic action of the chiral phosphoric acid catalyst, which acts as a bifunctional activator to orchestrate the bond-forming events. The catalyst, typically a binaphthyl or spiro-based derivative substituted with bulky groups like 9-anthracenyl, creates a well-defined chiral pocket through hydrogen bonding interactions. In the transition state, the acidic proton of the phosphate activates the electrophilic p-methylene benzoquinone, while the basic phosphoryl oxygen simultaneously coordinates with the nucleophilic indole species. This dual activation lowers the energy barrier for the cyclization step and rigidly locks the substrates into a specific spatial orientation, ensuring that the new stereocenters are formed with high fidelity. The steric bulk of the catalyst's substituents plays a crucial role in shielding one face of the reacting molecules, thereby enforcing the observed enantioselectivity. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as slight modifications to the catalyst structure or solvent environment can have profound effects on the stereochemical outcome.
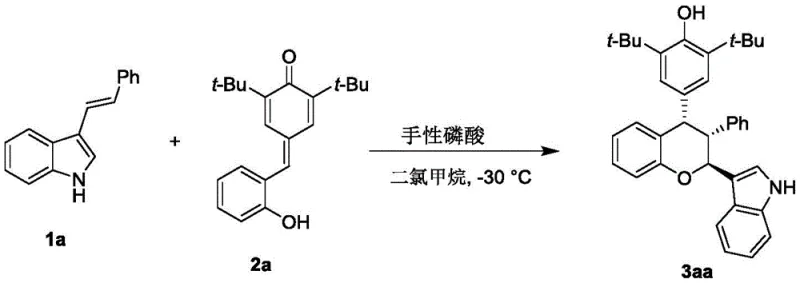
Moreover, the control of impurities is intrinsically linked to the selectivity of this catalytic cycle. By favoring a single enantiomeric pathway, the formation of unwanted diastereomers and racemic byproducts is suppressed, resulting in a cleaner crude reaction mixture. This high level of chemoselectivity reduces the burden on purification units, allowing for simpler workup procedures such as filtration and concentration followed by standard silica gel chromatography. The patent data indicates that yields can reach up to 95% with excellent diastereomeric ratios (dr) of 96:4 in optimized examples, demonstrating the robustness of the catalytic system. For quality assurance professionals, this mechanistic clarity provides confidence in the reproducibility of the process, ensuring that every batch meets stringent purity specifications required for clinical trial materials. The ability to tune the catalyst structure, as shown by the variety of derivatives in the patent, offers a powerful tool for troubleshooting and process intensification during scale-up activities.
How to Synthesize Chiral Chroman Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize the benefits of the organocatalytic system. The process begins with the careful preparation of dry solvents and reagents, as moisture sensitivity can impact the catalyst's performance. The reaction is typically conducted in dichloromethane, a solvent chosen for its ability to dissolve both organic substrates and the catalyst while maintaining stability at low temperatures. Detailed standardized synthetic steps are provided below to guide process development teams in replicating the high yields and selectivity reported in the patent literature. Following these guidelines ensures that the critical parameters, such as catalyst loading and temperature control, are maintained within the optimal window defined by the intellectual property.
- Prepare the reaction mixture by combining p-methylene benzoquinone and 3-indolene derivatives in dichloromethane solvent with molecular sieves as an additive.
- Add the chiral phosphoric acid catalyst (e.g., binaphthyl derivative with 9-anthracenyl group) to the mixture under stirring conditions.
- Maintain the reaction temperature at -30°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organocatalytic route offers substantial strategic advantages for organizations managing the supply of complex pharmaceutical intermediates. The elimination of transition metals from the process flow removes a major cost center associated with metal scavenging resins and rigorous analytical testing for residual metals, directly contributing to cost reduction in API manufacturing. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, enhancing the overall sustainability profile of the production facility. The high atom economy and yield of the reaction mean that less raw material is wasted, optimizing inventory turnover and reducing the environmental impact of chemical waste disposal. These factors combine to create a more resilient supply chain capable of responding quickly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The transition to a metal-free organocatalytic process fundamentally alters the cost structure of producing chiral chromans. By removing the need for expensive palladium or rhodium catalysts and the associated removal technologies, manufacturers can achieve significant savings on raw material costs. Additionally, the simplified purification workflow reduces solvent usage and processing time, further lowering operational expenditures. The high yields reported in the patent minimize the need for recycling unreacted starting materials, ensuring that capital is not tied up in low-efficiency loops. This economic efficiency allows suppliers to offer more competitive pricing models to downstream partners while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain stability. Unlike processes requiring cryogenic temperatures below -70°C or high-pressure hydrogenation, this method operates at a manageable -30°C using standard cooling equipment available in most multipurpose plants. The availability of the chiral phosphoric acid catalysts and simple organic starting materials ensures that sourcing risks are minimized, preventing bottlenecks caused by specialized reagent shortages. This reliability is crucial for long-term project planning, allowing procurement managers to secure multi-year contracts with confidence. The consistency of the process also reduces the risk of batch failures, ensuring a steady flow of materials to support clinical and commercial timelines.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and ton scales is facilitated by the inherent safety and simplicity of the chemistry. The absence of pyrophoric reagents or toxic gases simplifies hazard assessments and regulatory filings, accelerating the path to commercial production. The use of dichloromethane, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols, supporting green chemistry initiatives. The high selectivity of the reaction reduces the generation of hazardous waste streams, aligning with increasingly strict environmental regulations globally. This scalability ensures that the technology can grow with the drug candidate, supporting seamless transitions from preclinical research to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral chroman compounds. The answers are derived directly from the experimental data and technical disclosures found in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. It also clarifies the specific advantages this method holds over legacy synthetic routes.
Q: What represents the primary advantage of this chiral phosphoric acid catalyzed method?
A: The primary advantage is the ability to achieve high enantioselectivity and diastereoselectivity under mild reaction conditions (-30°C) without the need for toxic transition metals, ensuring a cleaner impurity profile suitable for pharmaceutical applications.
Q: Which cancer cell lines show sensitivity to the synthesized chroman compounds?
A: Biological testing indicates strong cytotoxic activity against MDA-MB-231 breast cancer cells, HepG2 liver cancer cells, and SGC-7901 gastric cancer cells, highlighting their potential as oncology leads.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method utilizes conventional solvents like dichloromethane and stable organocatalysts, avoiding hazardous reagents and extreme conditions, which facilitates safer and more cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies disclosed in CN112920173A for the development of novel oncology therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to global supply. We are committed to delivering high-purity chiral chroman intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex organocatalytic reactions allows us to navigate the nuances of stereoselective synthesis, guaranteeing the high enantiomeric excess required for biological efficacy.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your timeline and optimize your budget. Let us be your partner in bringing these promising cytotoxic agents from the laboratory to the patients who need them most.
