Advanced Nine-Step Synthesis of Thiazole Derivatives for Commercial Pharmaceutical Applications
Advanced Nine-Step Synthesis of Thiazole Derivatives for Commercial Pharmaceutical Applications
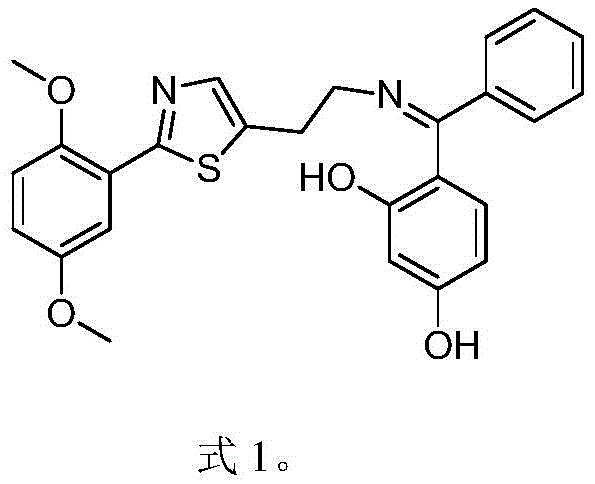
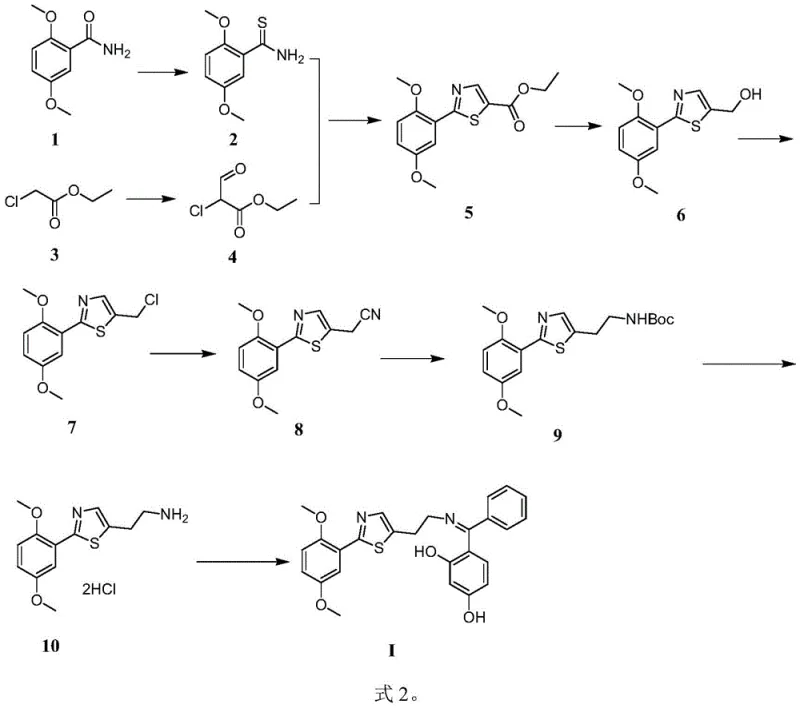
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for complex heterocyclic compounds that serve as critical building blocks for new drug candidates. Patent CN111875557B introduces a significant advancement in this domain by detailing a comprehensive synthesis method for a specific thiazole derivative, chemically identified as (Z)-4-((2-(2-(2-(2,5-dimethoxyphenyl)thiazol-5-yl)ethylimino)(phenyl)methyl)-1,3-benzenediol. This molecule represents a high-value pharmaceutical intermediate with potential applications in the development of novel therapeutic agents targeting various physiological pathways. The disclosed methodology offers a structured nine-step pathway that begins with readily available starting materials such as 2,5-dimethoxybenzamide and ethyl 2-chloroacetate. By establishing a clear and reproducible protocol, this patent addresses the common challenges associated with the synthesis of functionalized thiazole cores, particularly regarding yield consistency and impurity control. The strategic design of this route allows for precise manipulation of the molecular architecture, ensuring that the final product meets the stringent quality requirements necessary for downstream drug development. For R&D teams evaluating new chemical entities, understanding the nuances of this synthesis provides a competitive edge in optimizing lead compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex thiazole derivatives has been plagued by several inherent inefficiencies that hinder commercial viability and research progress. Conventional routes often rely on harsh reaction conditions that can compromise the integrity of sensitive functional groups, leading to the formation of difficult-to-remove impurities and byproducts. Many existing methods suffer from low overall yields due to non-selective reactions or the requirement for multiple protection and deprotection steps that add unnecessary complexity and cost to the process. Furthermore, the use of expensive or hazardous reagents in traditional protocols can pose significant safety risks and environmental burdens, making scale-up problematic for manufacturing facilities. The lack of standardized procedures in older literature often results in poor reproducibility, where slight variations in temperature or reagent quality can drastically alter the outcome. These limitations create bottlenecks in the supply chain for high-purity intermediates, forcing pharmaceutical companies to invest heavily in process optimization before any clinical progress can be made. Consequently, there is a pressing need for improved synthetic strategies that offer better control over reaction parameters and product quality.
The Novel Approach
The methodology outlined in patent CN111875557B presents a novel approach that systematically overcomes the drawbacks associated with conventional thiazole synthesis. This new route utilizes a stepwise construction strategy that builds the molecular complexity gradually, allowing for better control over each transformation. By selecting specific reagents such as phosphorus pentasulfide for thionation and employing controlled temperature gradients ranging from 0°C to 95°C, the process minimizes side reactions and maximizes the conversion of starting materials. The integration of a cyanation step followed by a selective reduction allows for the precise installation of the ethylamine side chain, which is crucial for the biological activity of the final derivative. Moreover, the use of common organic solvents like acetonitrile, ethanol, and dichloromethane ensures that the process remains compatible with standard industrial equipment and safety protocols. The final condensation step with 2-hydroxy-4-methoxybenzophenone is optimized to favor the Z-isomer, enhancing the stereochemical purity of the product. This holistic approach not only improves the chemical efficiency but also aligns with modern green chemistry principles by reducing waste and improving atom economy.
Mechanistic Insights into Thiazole Ring Formation and Functionalization
The core of this synthetic strategy lies in the efficient construction of the thiazole ring, which is achieved through a condensation reaction between a thioamide and an alpha-haloketone derivative. In the third step of the sequence, 2,5-dimethoxybenzothioamide reacts with ethyl 2-chloro-3-oxopropionate in the presence of an organic base such as triethylamine. This reaction proceeds via a nucleophilic attack of the sulfur atom on the carbonyl carbon, followed by cyclization and dehydration to form the stable thiazole heterocycle. The choice of acetonitrile as the solvent and the maintenance of a temperature between 75-95°C are critical for driving this equilibrium towards the desired product while suppressing polymerization or decomposition. Following the ring formation, the ester group is selectively reduced to a primary alcohol using reducing agents like sodium borohydride or lithium aluminum hydride, depending on the specific example embodiment. This reduction step is pivotal as it sets the stage for subsequent functionalization of the side chain without affecting the sensitive methoxy groups on the phenyl ring. The mechanistic pathway ensures that the electronic properties of the thiazole ring are preserved, maintaining its potential for hydrogen bonding and pi-stacking interactions in biological systems.
Impurity control is rigorously managed throughout the synthesis through the use of Thin-Layer Chromatography (TLC) monitoring at every stage. This analytical technique allows chemists to visualize the consumption of starting materials and the appearance of products in real-time, ensuring that reactions are quenched at the optimal point to prevent over-reaction or degradation. For instance, during the chlorination step using thionyl chloride, strict temperature control between 0-35°C prevents the formation of dichloro byproducts which could complicate downstream purification. The cyanation step, which introduces the nitrogen atom required for the final amine functionality, is conducted in a mixed solvent system of DMF and water to enhance the solubility of the inorganic cyanide salt while maintaining reaction homogeneity. The final condensation reaction is driven by the basicity of potassium carbonate, which facilitates the formation of the imine bond while promoting the specific Z-configuration. By understanding these mechanistic details, process chemists can identify critical control points that guarantee the consistency and quality of the final pharmaceutical intermediate.
How to Synthesize (Z)-4-((2-(2-(2-(2,5-dimethoxyphenyl)thiazol-5-yl)ethylimino)(phenyl)methyl)-1,3-benzenediol Efficiently
The synthesis of this complex thiazole derivative requires a disciplined approach to reaction management and purification to ensure high yields and purity. The process begins with the thionation of the benzamide precursor, followed by a series of functional group transformations that build the molecular complexity step-by-step. Each intermediate must be carefully isolated and characterized before proceeding to the next stage to prevent the carryover of impurities that could poison subsequent catalysts or reagents. The patent provides detailed experimental conditions including specific molar ratios, solvent volumes, and reaction times that have been optimized through extensive laboratory testing. Adhering to these parameters is essential for replicating the success of the disclosed examples and achieving the reported yields. For industrial applications, it is recommended to implement in-process controls that monitor key quality attributes such as water content and residual solvent levels. The detailed standardized synthesis steps see the guide below.
- Preparation of 2,5-dimethoxybenzothioamide via thionation of 2,5-dimethoxybenzamide using phosphorus pentasulfide in dioxane at 70-90°C.
- Condensation with ethyl 2-chloro-3-oxopropionate followed by cyclization to form the ethyl thiazole-5-carboxylate core structure.
- Sequential reduction, chlorination, cyanation, and protection steps to functionalize the thiazole side chain before final condensation with benzophenone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in patent CN111875557B offers several distinct advantages that translate into tangible business value. The reliance on commercially available starting materials such as 2,5-dimethoxybenzamide and ethyl 2-chloroacetate ensures a stable and reliable supply chain, reducing the risk of production delays caused by raw material shortages. These commodities are produced by multiple vendors globally, which fosters competitive pricing and allows procurement managers to negotiate favorable terms. The synthetic pathway avoids the use of exotic or highly regulated reagents that often require special handling permits and incur significant compliance costs. Instead, it utilizes standard organic solvents and inorganic salts that are easy to source and dispose of in accordance with environmental regulations. This simplicity in material sourcing significantly reduces the administrative burden on the supply chain team and enhances the overall agility of the manufacturing operation. Furthermore, the robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, ensuring supply continuity even in the face of geopolitical or logistical disruptions.
- Cost Reduction in Manufacturing: The streamlined nature of this nine-step synthesis contributes to substantial cost savings by minimizing the number of unit operations and purification steps required. By achieving high yields in key transformation steps, such as the reduction and chlorination reactions, the process reduces the amount of raw material needed per kilogram of final product. The elimination of expensive transition metal catalysts in favor of organic bases and common reducing agents further lowers the direct material costs. Additionally, the ability to perform reactions at moderate temperatures reduces energy consumption for heating and cooling, leading to lower utility costs over the lifecycle of the product. These cumulative efficiencies result in a more cost-effective manufacturing process that improves the margin profile for the final pharmaceutical intermediate. Procurement teams can leverage these efficiencies to secure better pricing structures for long-term supply agreements.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction conditions enhances the reliability of the supply chain by reducing the likelihood of batch failures. Each step in the synthesis has been optimized to tolerate minor variations in reagent quality, which is crucial when sourcing materials from different suppliers. The modular nature of the synthesis allows for the potential outsourcing of specific intermediate steps, providing flexibility in capacity management. This decentralization of production risk ensures that the supply of the final thiazole derivative remains uninterrupted even if one part of the chain faces temporary constraints. Supply chain heads can benefit from this resilience by maintaining lower safety stock levels while still meeting customer demand. The predictability of the process also facilitates more accurate forecasting and inventory planning, optimizing working capital utilization.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, as evidenced by the use of standard reactor sizes and common workup procedures in the patent examples. The process avoids the generation of hazardous waste streams that would require expensive treatment or disposal, aligning with increasingly strict environmental regulations. The use of aqueous workups and recyclable solvents minimizes the environmental footprint of the manufacturing process. This compliance with green chemistry principles not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturer. Scalability is further supported by the linear progression of the synthesis, which allows for easy expansion from pilot plant to commercial scale without significant re-engineering. This readiness for scale-up ensures that the supply can grow in tandem with the clinical and commercial demand for the drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this thiazole derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for stakeholders evaluating the feasibility of incorporating this intermediate into their development pipelines. The information covers aspects of reaction safety, quality control, and logistical considerations that are vital for decision-making. We encourage technical teams to review these points closely to ensure alignment with their specific project requirements.
Q: What are the critical reaction conditions for the thiazole ring formation?
A: The thiazole ring formation occurs in step 3, requiring a temperature range of 75-95°C in acetonitrile with an organic base such as triethylamine, ensuring complete cyclization over 12 hours.
Q: How is the purity of the final thiazole derivative monitored?
A: The patent specifies the use of Thin-Layer Chromatography (TLC) at each stage to analyze raw material consumption and reaction completion before proceeding to workup and purification.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the examples demonstrate scalability using 2L to 10L reactors with standard reagents and manageable temperature profiles ranging from 0°C to 95°C, indicating industrial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazole Derivative Supplier
NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the nuances of heterocyclic synthesis and can adapt the patented route to meet your specific purity and throughput requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of thiazole derivative meets the highest industry standards. Our commitment to quality and reliability makes us an ideal partner for pharmaceutical companies seeking a stable source of critical intermediates. By leveraging our infrastructure, you can accelerate your timeline from research to commercialization with confidence.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of partnering with us. We are prepared to provide specific COA data and route feasibility assessments to support your due diligence process. Let us help you secure a reliable supply of high-quality thiazole derivatives for your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
