Optimizing Carbapenem Intermediate Production via Advanced Metal-Catalyzed Azetidinone Synthesis
Optimizing Carbapenem Intermediate Production via Advanced Metal-Catalyzed Azetidinone Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates, particularly those required for the production of 1-beta-methyl carbapenem derivatives. Patent CN1610663A discloses a groundbreaking process for selectively obtaining azetidinone compounds having a 1'-beta configuration, which serve as pivotal precursors in the synthesis of potent carbapenem-type antibacterial agents. This technology addresses long-standing challenges in the field by enabling the production of these high-value intermediates in a shorter period and at higher yields under mild reaction conditions. By overcoming the limitations of existing methods, this process offers a strategic advantage for manufacturers aiming to secure a stable supply of high-purity pharmaceutical intermediates. The innovation lies in the specific combination of a metal compound and a base to facilitate the reaction between a specialized azetidinone derivative and a diazo compound, ensuring exceptional stereocontrol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-beta-methyl azetidinone compounds has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Traditional routes, such as those depicted in Reaction Scheme 1, often rely on expensive starting materials that drive up the overall cost of goods sold for the final antibiotic product. 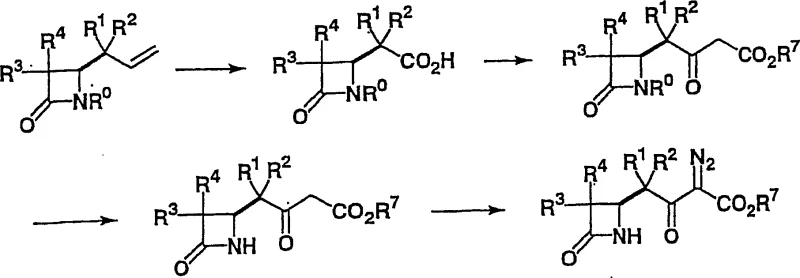 Furthermore, these legacy methods typically involve a large number of synthetic steps, each introducing potential points of failure, yield loss, and impurity generation. The operational complexity associated with multi-step sequences increases the burden on quality control and extends the total production lead time. In addition to cost and complexity, some conventional approaches require the use of unstable intermediates, such as silyl enol ethers, which are difficult to handle safely on an industrial scale. The instability of these reagents necessitates stringent storage conditions and immediate usage, creating logistical bottlenecks that compromise supply chain continuity and increase the risk of batch failures.
Furthermore, these legacy methods typically involve a large number of synthetic steps, each introducing potential points of failure, yield loss, and impurity generation. The operational complexity associated with multi-step sequences increases the burden on quality control and extends the total production lead time. In addition to cost and complexity, some conventional approaches require the use of unstable intermediates, such as silyl enol ethers, which are difficult to handle safely on an industrial scale. The instability of these reagents necessitates stringent storage conditions and immediate usage, creating logistical bottlenecks that compromise supply chain continuity and increase the risk of batch failures.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by introducing a direct and highly selective catalytic method that bypasses the need for unstable intermediates. Instead of relying on fragile silyl enol ethers, this process utilizes a stable diazo compound reacting directly with an azetidinone precursor in the presence of a specific metal compound and a base. This fundamental shift in synthetic strategy drastically simplifies the reaction sequence, reducing the number of unit operations required to reach the target molecule. The ability to conduct the reaction under mild conditions, often at temperatures ranging from -70 to 0 degrees Celsius, minimizes energy consumption and reduces the thermal stress on sensitive functional groups. Consequently, this method not only improves the overall yield but also enhances the safety profile of the manufacturing process, making it an ideal candidate for commercial scale-up in regulated pharmaceutical environments.
Mechanistic Insights into Metal-Catalyzed Diazocarbonylation
The core of this technological advancement lies in the precise interaction between the diazo compound, represented by General Formula [3], and the metal catalyst system. 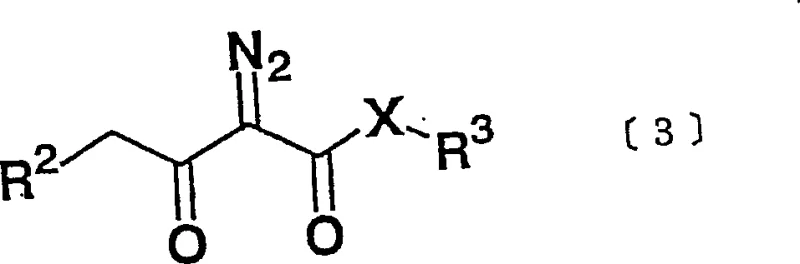 The metal compound, which can be selected from a wide range of options including titanium tetrachloride, zinc chloride, or aluminum chloride, acts as a Lewis acid to activate the diazo species. This activation is critical for generating the reactive carbenoid or metal-carbene intermediate that subsequently attacks the azetidinone ring system. The presence of a base, such as triethylamine or tributylamine, is equally crucial as it neutralizes acidic byproducts and helps maintain the catalytic cycle's efficiency. The synergy between the metal center and the base creates a highly controlled reaction environment that favors the formation of the desired 1'-beta stereoisomer over the alpha counterpart. This mechanistic precision is what allows the process to achieve beta-to-alpha ratios often exceeding 95:5, a level of stereocontrol that is difficult to replicate with non-catalytic methods.
The metal compound, which can be selected from a wide range of options including titanium tetrachloride, zinc chloride, or aluminum chloride, acts as a Lewis acid to activate the diazo species. This activation is critical for generating the reactive carbenoid or metal-carbene intermediate that subsequently attacks the azetidinone ring system. The presence of a base, such as triethylamine or tributylamine, is equally crucial as it neutralizes acidic byproducts and helps maintain the catalytic cycle's efficiency. The synergy between the metal center and the base creates a highly controlled reaction environment that favors the formation of the desired 1'-beta stereoisomer over the alpha counterpart. This mechanistic precision is what allows the process to achieve beta-to-alpha ratios often exceeding 95:5, a level of stereocontrol that is difficult to replicate with non-catalytic methods.
Impurity control is another critical aspect where this mechanism offers substantial benefits to R&D teams focused on product quality. By avoiding the use of unstable silyl enol ethers, the process eliminates a major source of decomposition products and side reactions that typically contaminate the crude reaction mixture. The mild reaction conditions further suppress the formation of thermal degradation byproducts, resulting in a cleaner reaction profile that simplifies downstream purification. The use of standard organic solvents like dichloromethane or toluene ensures that the reaction mixture remains homogeneous and manageable, facilitating efficient work-up procedures such as solvent extraction and crystallization. This high level of chemical fidelity means that the final azetidinone intermediate meets stringent purity specifications with minimal additional processing, directly impacting the cost and speed of the overall manufacturing campaign.
How to Synthesize 1-Beta-Methyl Azetidinone Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the metal-catalyzed system. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent moisture or oxygen from deactivating the sensitive metal catalyst. The detailed standardized synthesis steps provided in the guide below outline the precise addition order and temperature controls necessary to achieve optimal results. Adhering to these protocols ensures that the reactive diazo species is generated in situ and consumed immediately, minimizing safety risks associated with handling high-energy compounds. This structured approach allows manufacturing teams to replicate the high yields and selectivity reported in the patent data consistently across different batch sizes.
- Prepare the reaction system under an inert atmosphere using a suitable organic solvent such as dichloromethane or toluene.
- React the specific diazo compound with a selected metal compound and base at controlled low temperatures to form the active catalytic species.
- Introduce the azetidinone precursor to the mixture, maintaining strict temperature control to ensure high beta-selectivity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible improvements in cost structure and supply reliability. The elimination of expensive and unstable starting materials directly reduces the raw material costs associated with producing carbapenem intermediates. By simplifying the synthetic sequence, the process reduces the consumption of solvents and reagents per kilogram of product, leading to substantial cost savings in waste disposal and material procurement. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring a more consistent supply of the intermediate. This reliability is crucial for maintaining continuous production schedules for downstream antibiotic manufacturing, preventing costly delays caused by batch rejections or supply shortages.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the operational expenditure required for intermediate production. By removing the need for specialized handling of unstable silyl enol ethers, the facility can reduce costs associated with specialized storage and safety protocols. The higher selectivity of the reaction minimizes the loss of valuable starting materials to unwanted isomers, effectively increasing the overall material efficiency of the plant. Additionally, the use of commercially available metal catalysts and common organic solvents ensures that procurement teams can source materials from multiple suppliers, fostering competitive pricing and reducing dependency on single-source vendors.
- Enhanced Supply Chain Reliability: The stability of the reagents used in this process greatly enhances the resilience of the supply chain. Unlike methods relying on freshly prepared unstable intermediates, this route allows for the stocking of key reagents, enabling faster response times to fluctuations in market demand. The mild reaction conditions also reduce the risk of equipment corrosion or failure, leading to higher asset utilization rates and fewer unplanned maintenance shutdowns. This operational stability ensures that delivery commitments to downstream pharmaceutical customers can be met consistently, strengthening long-term partnerships and contractual agreements.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a cleaner and more sustainable manufacturing profile. The reduction in synthetic steps inherently lowers the volume of chemical waste generated, simplifying compliance with increasingly stringent environmental regulations. The ability to use standard solvents facilitates efficient solvent recovery and recycling programs, further reducing the environmental footprint of the production facility. The process is designed to be scalable from laboratory to commercial production without significant re-engineering, allowing for rapid capacity expansion to meet growing global demand for carbapenem antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azetidinone synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They serve to clarify the operational benefits and technical feasibility of adopting this method for large-scale pharmaceutical intermediate production.
Q: What are the primary advantages of this metal-catalyzed method over conventional silyl enol ether routes?
A: This method eliminates the need for unstable silyl enol ether intermediates, which are difficult to handle industrially. It operates under milder conditions with fewer synthetic steps, significantly improving operational safety and process reliability.
Q: How does this process impact the stereochemical purity of the final carbapenem intermediate?
A: The use of specific metal compounds and bases allows for high stereoselectivity, typically achieving a beta-to-alpha isomer ratio exceeding 85:15, often reaching 95:5, which simplifies downstream purification.
Q: Is this synthesis route scalable for commercial manufacturing of antibiotic intermediates?
A: Yes, the process utilizes commercially available metal catalysts and standard organic solvents, avoiding exotic or hazardous reagents, which facilitates straightforward scale-up from laboratory to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azetidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the advanced metal-catalyzed processes described in patent CN1610663A can be successfully transferred to industrial manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of azetidinone intermediate meets the exacting standards required by top-tier pharmaceutical companies. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to optimize your production costs and secure a reliable source of carbapenem intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your long-term strategic goals. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to innovation, quality, and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
