Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The strategic incorporation of fluorinated heterocycles into small molecule therapeutics has become a cornerstone of modern medicinal chemistry, driven by the need to enhance metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in the synthesis of this privileged scaffold. The 1,2,4-triazole ring is ubiquitous in high-value pharmaceuticals, serving as a key bioisostere in drugs such as Sitagliptin and various antifungal agents, where the trifluoromethyl group further modulates lipophilicity and binding affinity. 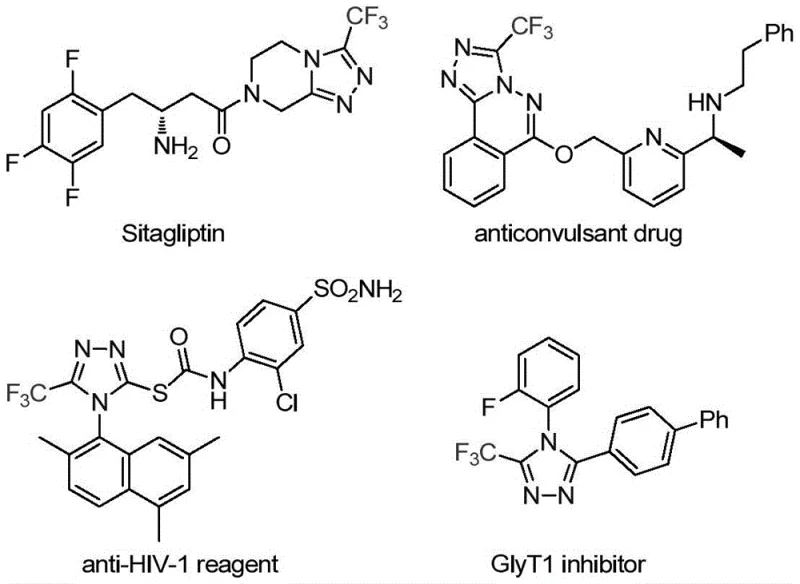 This innovation provides a reliable pharmaceutical intermediate supplier with a robust pathway to access these complex motifs efficiently. By leveraging a dual catalytic system involving molybdenum and copper, the disclosed technology enables the direct construction of the triazole core from readily available precursors, bypassing the multi-step sequences and harsh conditions that have historically plagued this area of organic synthesis.
This innovation provides a reliable pharmaceutical intermediate supplier with a robust pathway to access these complex motifs efficiently. By leveraging a dual catalytic system involving molybdenum and copper, the disclosed technology enables the direct construction of the triazole core from readily available precursors, bypassing the multi-step sequences and harsh conditions that have historically plagued this area of organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied heavily on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of oxazolinone derivatives. These traditional pathways are fraught with significant operational challenges, including the handling of unstable and potentially explosive hydrazine intermediates, which pose severe safety risks in large-scale manufacturing environments. Furthermore, existing copper-catalyzed multi-component reactions often require diazonium salts or trifluorodiazoethane, reagents that are not only hazardous but also demand stringent temperature control and specialized equipment to manage gas evolution. The reliance on such dangerous starting materials inevitably increases the cost of goods sold (COGS) due to enhanced safety protocols, specialized waste disposal requirements, and lower overall throughput caused by cautious reaction pacing.
The Novel Approach
In stark contrast, the methodology described in CN113307778A utilizes a benign and atom-economical strategy centered on the cycloaddition of trifluoroethylimidoyl chloride and functionalized isonitriles. This novel approach operates under remarkably mild thermal conditions, typically between 70°C and 90°C, which drastically reduces energy consumption compared to high-temperature reflux methods. The use of stable solid or liquid reagents eliminates the need for cryogenic conditions or high-pressure reactors, thereby simplifying the engineering controls required for production. Moreover, the reaction demonstrates exceptional functional group tolerance, allowing for the direct synthesis of diverse derivatives without the need for extensive protecting group strategies, which streamlines the synthetic route and accelerates the timeline from benchtop discovery to commercial supply.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The efficacy of this transformation hinges on a sophisticated synergistic catalytic cycle involving molybdenum hexacarbonyl and cuprous acetate. Mechanistically, the molybdenum species acts as a potent activator for the functionalized isonitrile, coordinating to the nitrogen lone pair and increasing the electrophilicity of the isonitrile carbon. Simultaneously, the copper catalyst facilitates the activation of the trifluoroethylimidoyl chloride, promoting the initial nucleophilic attack. 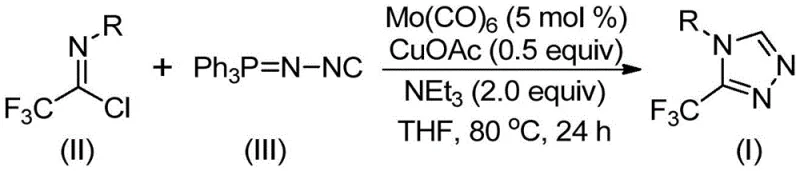 This cooperative interaction lowers the activation energy barrier for the [3+2] cycloaddition step, enabling the formation of the five-membered triazole ring with high regioselectivity. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water or base in the system, ensures the irreversible formation of the aromatic triazole product, driving the equilibrium towards completion and minimizing the accumulation of partially reacted intermediates.
This cooperative interaction lowers the activation energy barrier for the [3+2] cycloaddition step, enabling the formation of the five-membered triazole ring with high regioselectivity. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water or base in the system, ensures the irreversible formation of the aromatic triazole product, driving the equilibrium towards completion and minimizing the accumulation of partially reacted intermediates.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity pharmaceutical intermediates. The specificity of the metal-ligand interactions minimizes side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride, which are common pitfalls in non-catalyzed thermal reactions. The use of triethylamine as a base scavenges the hydrochloric acid byproduct generated during the cyclization, preventing acid-catalyzed decomposition of the sensitive triazole ring. Consequently, the crude reaction profile is significantly cleaner, reducing the load on downstream purification units like chromatography columns or crystallization tanks, which directly translates to higher recovery yields and reduced solvent usage in the final isolation steps.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemistry teams aiming to establish robust manufacturing procedures. The reaction is performed in common aprotic solvents such as tetrahydrofuran (THF), which are easily recovered and recycled, aligning with green chemistry principles. The standardized procedure involves charging the reactor with the catalysts, base, and substrates under an inert atmosphere, followed by heating to the specified temperature range for a defined period.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition centered on cost reduction in API manufacturing and supply chain resilience. The primary drivers of cost efficiency lie in the selection of starting materials; both trifluoroethylimidoyl chlorides and functionalized isonitriles are derived from commodity chemicals that are widely available in the global market, mitigating the risk of supply disruptions associated with exotic or proprietary reagents. Furthermore, the elimination of hazardous hydrazines and diazo compounds removes the need for expensive safety infrastructure and specialized containment systems, leading to substantial capital expenditure (CAPEX) savings for facilities upgrading their production lines.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the high atom economy and the use of inexpensive catalysts like cuprous acetate. Unlike precious metal catalysts such as palladium or rhodium, copper and molybdenum complexes are significantly more affordable and do not require rigorous recovery processes to meet residual metal specifications in the final drug substance. The mild reaction conditions also contribute to lower utility costs, as the process does not demand extreme heating or cooling, allowing for the use of standard jacketed reactors and conventional heating media like steam or hot oil.
- Enhanced Supply Chain Reliability: By relying on a synthetic route that utilizes stable, shelf-stable reagents, manufacturers can maintain leaner inventory levels without compromising production continuity. The robustness of the reaction against moisture and oxygen variations, facilitated by the molecular sieves and inert atmosphere, ensures consistent batch-to-batch quality, reducing the incidence of failed batches that can disrupt delivery schedules. This reliability is crucial for maintaining just-in-time delivery models required by major pharmaceutical clients who demand strict adherence to production timelines.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is streamlined by the homogeneous nature of the reaction mixture and the absence of gas-generating steps that complicate reactor headspace management. The simplified workup procedure, involving filtration and standard chromatography or crystallization, generates less hazardous waste compared to traditional methods that produce heavy metal sludge or toxic hydrazine byproducts. This alignment with environmental regulations reduces the burden on waste treatment facilities and minimizes the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this catalytic system for industrial applications.
Q: What are the primary advantages of this Mo/Cu co-catalytic system over traditional hydrazine-based cyclizations?
A: This novel approach eliminates the need for hazardous hydrazine derivatives and harsh acidic conditions often required in traditional methods. By utilizing stable trifluoroethylimidoyl chlorides and functionalized isonitriles, the process offers superior functional group tolerance and significantly simplified downstream purification, reducing the burden on waste treatment facilities.
Q: Is this synthetic route scalable for industrial production of API intermediates?
A: Yes, the patent data explicitly confirms that the reaction conditions are mild (70-90°C) and the methodology has been successfully demonstrated at the gram level with high efficiency. The use of common solvents like THF and commercially available catalysts suggests a straightforward path to kilogram and metric ton scale-up without requiring specialized high-pressure equipment.
Q: How does the substrate scope impact the versatility of this method for drug discovery?
A: The method exhibits broad substrate tolerance, accommodating various aryl substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like halogens and nitro groups. This flexibility allows medicinal chemists to rapidly generate diverse libraries of 3-trifluoromethyl-1,2,4-triazoles for structure-activity relationship (SAR) studies without optimizing conditions for each new derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs.
