Revolutionizing Amlodipine Intermediate Production: A Safe and Scalable Synthetic Route
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical cardiovascular medications, and the synthesis of Amlodipine Besylate remains a focal point due to its widespread use in treating hypertension and angina. A pivotal breakthrough in this domain is detailed in patent CN107935912B, which outlines a novel, safe, and highly efficient preparation process for a key Amlodipine intermediate. This technology addresses long-standing safety and efficiency bottlenecks associated with traditional etherification reactions. By shifting away from hazardous reagents toward a more benign chemical architecture, this patent offers a compelling value proposition for manufacturers aiming to optimize their production lines. The core innovation lies in the strategic use of a tosylate-activated intermediate, which fundamentally alters the reaction kinetics and safety profile of the synthesis.
For R&D directors and process chemists, the implications of this patent extend beyond mere regulatory compliance; it represents a tangible opportunity to refine impurity profiles and streamline purification protocols. The transition from direct chloro-substitution to a tosylate-mediated pathway allows for milder reaction conditions while maintaining high conversion rates. This report analyzes the technical merits of this approach, evaluating its potential to serve as a reliable standard for high-purity pharmaceutical intermediates in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
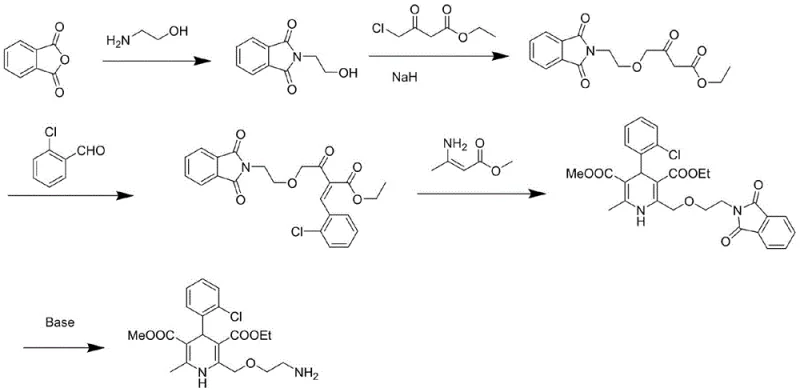
Historically, the industrial synthesis of the critical ether linkage in Amlodipine intermediates has relied heavily on strong, hazardous bases to drive nucleophilic substitution. As illustrated in the prior art, a common method involves the reaction of N-hydroxyethylphthalimide with ethyl 4-chloroacetoacetate using Sodium Hydride (NaH) in tetrahydrofuran (THF). While chemically effective, this approach introduces severe operational risks that complicate commercial scale-up of complex pharmaceutical intermediates. Sodium Hydride is a pyrophoric solid that reacts violently with moisture, releasing flammable hydrogen gas, which necessitates rigorous exclusion of water and specialized explosion-proof equipment.
Furthermore, alternative methods utilizing Sodium tert-butoxide have been explored, yet they present their own set of challenges, including high reagent costs and sensitivity to storage conditions. Literature reports also indicate that attempts using Sodium Ethoxide often result in suboptimal yields or fail to produce the target compound entirely under certain conditions. These limitations create a fragile supply chain where minor deviations in raw material quality or environmental controls can lead to batch failures, increased waste generation, and significant safety liabilities for the manufacturing facility.
The Novel Approach
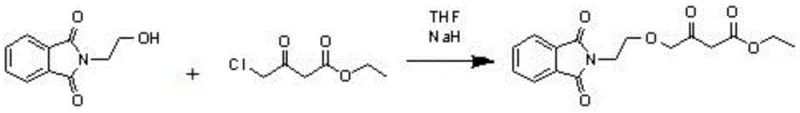
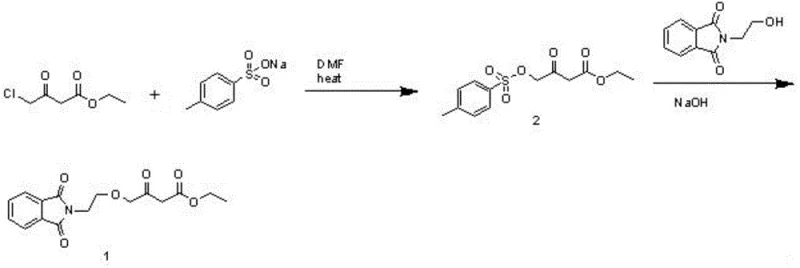
The methodology disclosed in patent CN107935912B circumvents these issues by introducing a two-step activation strategy that replaces the direct use of dangerous hydrides. The process begins by converting ethyl 4-chloroacetoacetate into a tosylate derivative (Compound 2) using sodium p-toluenesulfonate. This activated intermediate possesses a superior leaving group capability compared to the chloride, facilitating the subsequent etherification step under much milder conditions. In the second step, this tosylate intermediate reacts with N-hydroxyethylphthalimide in the presence of a simple base like sodium hydroxide and a phase transfer catalyst.
This strategic modification drastically reduces the energy barrier for the reaction, allowing it to proceed efficiently without the need for pyrophoric reagents. The result is a process that not only mitigates safety risks but also improves the overall economic feasibility of the synthesis. By decoupling the activation step from the coupling step, manufacturers gain greater control over reaction parameters, leading to more consistent product quality and reduced downtime associated with safety incidents or complex quenching procedures required for hydride-based chemistry.
Mechanistic Insights into Tosylate-Mediated Etherification
The core chemical transformation in this novel route is a nucleophilic substitution where the hydroxyl group of N-hydroxyethylphthalimide attacks the activated methylene carbon of the acetoacetate derivative. In the first stage, the displacement of the chloride ion by the p-toluenesulfonate anion generates Compound 2. This step is crucial because the tosylate group is a much better leaving group than chloride, stabilizing the transition state during the subsequent nucleophilic attack. The reaction is typically conducted in polar aprotic solvents like DMF at elevated temperatures (100-105°C), which enhances the solubility of the ionic reagents and accelerates the formation of the activated ester.
In the second stage, the introduction of a phase transfer catalyst (PTC) such as PEG-400 plays a pivotal role in determining the purity of the final product. The PTC facilitates the transport of the hydroxide anion into the organic phase, where it deprotonates the N-hydroxyethylphthalimide to form the reactive alkoxide nucleophile. Comparative data within the patent indicates that reactions performed without a PTC yield products with significantly lower purity (e.g., dropping from over 92% to roughly 59%). This suggests that the PTC not only accelerates the reaction rate but also suppresses side reactions, possibly by ensuring a more homogeneous reaction environment and preventing localized high concentrations of base that could degrade the sensitive beta-keto ester moiety.
How to Synthesize Ethyl 4-(2-phthalimidoethoxy)acetoacetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into two distinct stages: the preparation of the tosylate intermediate and the subsequent coupling reaction. Operators must strictly adhere to the specified molar ratios, particularly maintaining a slight excess of sodium p-toluenesulfonate in the first step to ensure complete conversion of the chloro-precursor. In the second step, the selection of the phase transfer catalyst is critical; PEG-400 has shown superior performance in enhancing purity compared to other options like tetrabutylammonium bromide. Detailed standardized operating procedures for this synthesis are provided below to guide technical teams in implementation.
- React ethyl 4-chloroacetoacetate with sodium p-toluenesulfonate in a solvent like DMF at elevated temperatures to form the activated tosylate intermediate.
- Combine the isolated tosylate intermediate with N-hydroxyethylphthalimide in the presence of a base such as sodium hydroxide.
- Add a phase transfer catalyst like PEG-400 to the reaction mixture and heat to reflux to facilitate the etherification and ensure high product purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits beyond simple chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for Sodium Hydride and Sodium tert-butoxide, facilities can remove high-hazard materials from their inventory, thereby reducing insurance premiums, storage costs, and the regulatory burden associated with handling dangerous goods. This shift directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering the total cost of ownership for the production line.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with commodity chemicals like sodium p-toluenesulfonate and sodium hydroxide creates a more resilient cost structure. Sodium Hydride requires specialized disposal protocols and inert handling systems, which add significant overhead to every batch produced. By switching to a base-mediated tosylation and coupling sequence, the process utilizes widely available, low-cost inputs. Furthermore, the improved purity achieved through phase transfer catalysis reduces the load on downstream purification units, potentially lowering solvent consumption and waste treatment costs associated with removing impurities.
- Enhanced Supply Chain Reliability: Dependence on specialized, high-risk reagents often creates single points of failure in the supply chain. Sodium Hydride, for instance, has a limited shelf life once opened and requires strict moisture control during transport. The new method relies on stable, shelf-stable solids that are easily sourced from multiple global suppliers. This diversification of the supply base ensures reducing lead time for high-purity pharmaceutical intermediates and protects production schedules from disruptions caused by the unavailability of niche reagents. The robustness of the chemistry also means that raw material variability is less likely to cause batch failures.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently greener. Avoiding the generation of hydrogen gas eliminates the risk of explosion and the need for complex venting systems. Additionally, the higher selectivity of the reaction minimizes the formation of by-products, leading to a lower E-factor (mass of waste per mass of product). This aligns with modern sustainability goals and simplifies the permitting process for capacity expansion, making it easier to scale from pilot batches to multi-ton commercial production without triggering major new environmental impact assessments.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is the new process considered safer than traditional Sodium Hydride methods?
A: Traditional methods rely on Sodium Hydride (NaH), which is pyrophoric and releases explosive hydrogen gas upon contact with moisture. The new process utilizes sodium p-toluenesulfonate and sodium hydroxide, eliminating the fire hazards and stringent inert atmosphere requirements associated with NaH.
Q: How does the phase transfer catalyst impact the final product quality?
A: Comparative experiments within the patent data demonstrate that adding a phase transfer catalyst, such as PEG-400, significantly enhances the purity of the final intermediate compared to reactions run without one, likely by improving the interaction between the organic substrate and the inorganic base.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It avoids expensive reagents like sodium tert-butoxide and dangerous reagents like sodium hydride, using cheap and readily available materials like sodium p-toluenesulfonate, which simplifies supply chain logistics and reduces operational risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amlodipine Intermediate Supplier
The technological advancements described in patent CN107935912B highlight the continuous evolution of process chemistry towards safer and more efficient manufacturing. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovations to maintain competitiveness in the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of intermediate delivered meets the highest standards required for API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the performance of this safer, tosylate-mediated synthesis in your own quality systems, ensuring a seamless transition to a more robust and cost-effective supply model.
