Revolutionizing Amlodipine Intermediate Production: A Safer, Scalable Synthetic Route for Global Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe synthetic routes for critical cardiovascular medications, and the production of amlodipine besylate remains a cornerstone of hypertension management globally. Patent CN111303006B introduces a transformative preparation method for a key amlodipine intermediate that addresses long-standing safety and efficiency bottlenecks inherent in legacy manufacturing processes. This innovation shifts the paradigm from hazardous batch operations involving pyrophoric reagents to a streamlined, Lewis acid-catalyzed sequence that prioritizes operator safety and supply chain stability. For R&D directors and procurement leaders, understanding this technological shift is vital, as it offers a pathway to secure high-purity intermediates while mitigating the operational risks associated with traditional sodium hydride (NaH) chemistry. The method leverages a sophisticated Swern oxidation followed by a precise C-H bond insertion, demonstrating how modern organic synthesis can reconcile high yield with industrial safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of the amlodipine key intermediate has been plagued by reliance on sodium hydride (NaH) as a base for alkylation steps, a practice that introduces severe safety liabilities and logistical constraints. As illustrated in the conventional pathway below, the use of NaH requires stringent anhydrous conditions and specialized equipment to prevent spontaneous combustion upon contact with moisture or air, limiting reactor sizes and complicating scale-up efforts.
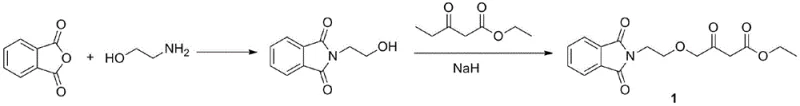
Furthermore, traditional routes often depend on ethyl 4-chloroacetoacetate, a raw material subject to volatile pricing and supply chain disruptions due to regulatory restrictions on its precursors like chlorine and diketene. The combination of hazardous reagents and unstable raw material sourcing creates a fragile supply chain where production halts due to weather conditions or regulatory inspections are a constant threat. Additionally, the multi-step nature of older methodologies, such as those involving Meldrum's acid or tosylate intermediates, accumulates impurities and lowers overall throughput, forcing manufacturers to invest heavily in purification and waste treatment infrastructure that erodes profit margins.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a concise two-step sequence that bypasses these historical pitfalls by employing diglycolamine, an abundant and cost-effective byproduct of morpholine manufacturing, as the starting scaffold. This strategic choice of raw materials not only stabilizes the supply chain against market fluctuations but also eliminates the need for expensive, regulated chlorinated intermediates. The core of this innovation lies in the replacement of dangerous strong bases with a controlled Swern oxidation system, which operates under mild conditions to generate the critical aldehyde functionality with high fidelity.
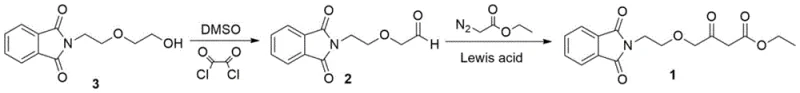
Following the oxidation, the process employs a Lewis acid-catalyzed C-H insertion reaction with ethyl diazoacetate, a transformation that constructs the essential beta-keto ester motif with remarkable atom economy. This methodology avoids the generation of stoichiometric salt waste typical of alkylation reactions, thereby simplifying downstream processing and reducing the environmental footprint. By decoupling the synthesis from hazardous reagents and unstable supply chains, this novel approach offers a resilient manufacturing platform capable of sustaining continuous production schedules even in challenging regulatory environments.
Mechanistic Insights into Swern Oxidation and Lewis Acid Catalyzed C-H Insertion
The first stage of this advanced synthesis involves a Swern oxidation, where dimethyl sulfoxide (DMSO) is activated by oxalyl chloride at cryogenic temperatures to form a reactive chlorosulfonium ion species. This electrophilic species attacks the hydroxyl group of the diglycolamine derivative, facilitating a smooth conversion to the corresponding aldehyde without over-oxidation to the carboxylic acid, a common side reaction in less controlled oxidation protocols. The meticulous control of temperature during the activation and addition phases ensures that the exothermic nature of the reaction is managed effectively, preserving the integrity of the sensitive isoindoline protecting group and preventing decomposition pathways that could compromise purity.
Subsequently, the generated aldehyde undergoes a Lewis acid-catalyzed C-H insertion reaction with ethyl diazoacetate, a step that is mechanistically distinct from traditional nucleophilic substitutions. In this transformation, the Lewis acid catalyst, such as tin(II) chloride or zinc chloride, coordinates with the diazo compound to generate a metal-carbenoid species that selectively inserts into the alpha-C-H bond of the aldehyde. This mechanism bypasses the need for pre-functionalized leaving groups and avoids the formation of regioisomers that often plague conventional alkylation strategies. The result is a highly selective formation of the beta-keto ester linkage, which is crucial for the subsequent Hantzsch condensation in the final amlodipine assembly, ensuring that the impurity profile remains within strict pharmacopeial limits.
How to Synthesize Amlodipine Key Intermediate Efficiently
Implementing this synthesis requires precise adherence to the optimized molar ratios and temperature profiles defined in the patent to maximize yield and safety. The process begins with the activation of DMSO followed by the controlled addition of the substrate, ensuring complete conversion before proceeding to the insertion step. While the laboratory procedure outlines specific solvent choices like dichloromethane, the protocol is robust enough to accommodate alternative aprotic solvents depending on facility capabilities. Detailed standardized operating procedures for scaling this route from pilot to commercial production are outlined in the technical guide below.
- Perform Swern oxidation on 2-(2-(2-hydroxyethoxy)ethyl)isoindoline-1,3-dione using DMSO and oxalyl chloride at low temperature to generate the aldehyde intermediate.
- Conduct a Lewis acid catalyzed C-H bond insertion reaction between the aldehyde intermediate and ethyl diazoacetate to form the final beta-keto ester structure.
- Purify the crude product through standard aqueous workup and solvent evaporation to obtain the high-purity amlodipine key intermediate ready for downstream cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational resilience and significant cost optimization opportunities. By eliminating the dependency on sodium hydride, facilities can remove the costly engineering controls and safety protocols required for pyrophoric materials, leading to substantial reductions in overhead and insurance premiums. Furthermore, the shift towards diglycolamine as a primary feedstock insulates the production schedule from the volatility associated with chlorinated intermediates, ensuring a more predictable and reliable flow of goods to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of high-cost, hazard-associated reagents with commodity chemicals that are widely available in the global market. Eliminating the NaH step removes the need for specialized quenching procedures and reduces the consumption of solvents required for extensive washing to remove inorganic salts. Additionally, the higher overall yield of the two-step sequence compared to multi-step traditional routes means less raw material is wasted per kilogram of final product, driving down the cost of goods sold significantly without compromising quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically improved by sourcing diglycolamine, a stable byproduct of the mature morpholine industry, rather than relying on ethyl 4-chloroacetoacetate which faces periodic supply shortages. The robustness of the Lewis acid catalysts used in the second step also allows for flexibility in sourcing, as several metal salts can achieve similar results, preventing single-supplier bottlenecks. This diversification of raw material inputs ensures that production can continue uninterrupted even if specific chemical markets experience temporary disruptions or logistics delays.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers a cleaner profile that aligns with increasingly stringent global regulations on hazardous waste. The absence of heavy metal catalysts in the final product simplifies purification, reducing the load on wastewater treatment plants and minimizing the generation of hazardous solid waste. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without the need for exotic materials of construction, facilitating rapid scale-up from pilot batches to multi-ton commercial production with minimal capital expenditure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational advantages and technical feasibility for potential partners evaluating this technology for their supply chains.
Q: Why is the new synthetic route for amlodipine intermediates considered safer than conventional methods?
A: Conventional methods rely heavily on sodium hydride (NaH), a pyrophoric reagent that poses significant fire and explosion risks, especially in humid conditions or large-scale reactors. The patented route eliminates NaH entirely, replacing it with a controlled Swern oxidation and Lewis acid catalysis, drastically reducing workplace hazards and insurance liabilities.
Q: How does this process impact the cost structure of amlodipine manufacturing?
A: By utilizing diglycolamine, a readily available byproduct of morpholine production, instead of expensive and supply-constrained ethyl 4-chloroacetoacetate, the raw material costs are significantly lowered. Additionally, the simplified two-step sequence reduces energy consumption and waste treatment expenses associated with multi-step traditional routes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. It avoids the reactor size limitations imposed by NaH safety concerns, allowing for the use of standard 1000L+ reactors without special atmospheric controls. The high yields and robust catalyst systems ensure consistent quality suitable for commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amlodipine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is essential for maintaining competitiveness in the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN111303006B process are translated into reliable supply realities. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of amlodipine intermediate meets the exacting standards required by top-tier regulatory agencies worldwide.
We invite forward-thinking procurement leaders to collaborate with us to leverage this advanced technology for their upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective supply of high-purity pharmaceutical intermediates that drives value for your organization.
