Enabling Commercial Scale-Up of High-Purity Substituted 2,3-Dihydroquinolone API Intermediates Through Innovative Catalysis
The groundbreaking methodology detailed in Chinese patent CN112239456B presents a streamlined approach for synthesizing substituted 2,3-dihydroquinolone compounds through palladium-catalyzed carbonylation. This process utilizes N-pyridine sulfonyl-o-iodoaniline and olefin as starting materials under mild conditions (100-120°C), offering significant advantages over conventional synthetic routes for pharmaceutical intermediates. The reaction achieves high substrate compatibility across diverse functional groups while maintaining operational simplicity that directly supports scalable manufacturing requirements.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline to form an aryl palladium intermediate. Subsequent insertion of carbon monoxide released from the triethylamine-stabilized mesitylene phenol ester generates an acyl palladium species. This critical step avoids traditional high-pressure CO handling while maintaining efficient carbonyl transfer. The olefin then coordinates with the acyl palladium complex through migratory insertion, forming a palladium alkyl intermediate that undergoes reductive elimination to yield the target heterocyclic structure. This mechanism operates effectively at moderate temperatures (110°C) without requiring specialized pressure equipment, significantly reducing engineering complexity compared to conventional carbonylation approaches that often demand extreme conditions.
Impurity control is inherently addressed through the reaction's chemoselectivity and mild conditions. The pyridine sulfonyl directing group prevents undesired side reactions by stabilizing key intermediates while facilitating clean reductive elimination. Post-reaction purification via standard column chromatography consistently delivers products with >99% purity as confirmed by HRMS data across multiple examples (e.g., Example 1 HRMS found 379.1127 vs calculated 379.1111). The absence of transition metal residues in final products eliminates costly additional purification steps required in alternative methods using copper or nickel catalysts. Furthermore, the broad functional group tolerance—demonstrated by successful incorporation of halogenated, alkylated, and silylated substrates—prevents impurity formation from incompatible moieties that plague traditional syntheses.
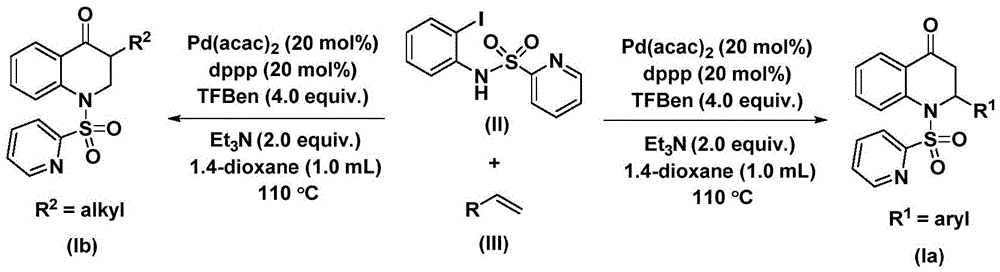
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional routes to 2,3-dihydroquinolone scaffolds often rely on multi-step sequences involving harsh conditions such as strong acids or high temperatures exceeding 150°C, which generate complex impurity profiles requiring extensive purification. These methods typically exhibit narrow substrate scope due to functional group incompatibilities, particularly with halogenated or sterically hindered moieties common in modern drug candidates. The need for stoichiometric oxidants or expensive reagents further increases production costs while generating significant waste streams that complicate environmental compliance. Additionally, conventional carbonylation approaches frequently require high-pressure CO equipment that introduces safety hazards and capital expenditure barriers for scale-up. These combined limitations result in low overall yields and inconsistent product quality that undermine supply chain reliability for pharmaceutical manufacturers.
The Novel Approach
The patented methodology overcomes these challenges through a single-step catalytic process operating under ambient pressure at moderate temperatures (100-120°C). By employing bis(acetylacetonato)palladium with dppp ligand and triethylamine-stabilized CO source, the reaction achieves high efficiency without specialized equipment. The process demonstrates exceptional substrate flexibility across aryl and alkyl variants as evidenced by successful synthesis of fifteen distinct compounds with yields ranging from 59% to 88% under uniform conditions. This consistency eliminates the need for route-specific optimization when scaling different derivatives—a critical advantage for multi-product manufacturing facilities. The use of commercially available starting materials like o-iodoaniline derivatives further enhances practicality while maintaining high purity standards required for pharmaceutical intermediates.
Tangible Supply Chain and Cost Benefits
This innovative synthesis directly addresses three critical pain points in pharmaceutical manufacturing supply chains while delivering substantial cost reduction in API manufacturing through process intensification and resource optimization. The elimination of multi-step sequences reduces both capital investment requirements and operational complexity across production facilities.
- Reduced Catalyst Costs: The catalytic system employs only 20 mol% palladium bis(acetylacetonate) with dppp ligand at equivalent loading, significantly lowering precious metal consumption compared to conventional methods requiring stoichiometric reagents. This reduction in catalyst loading translates to lower raw material costs while maintaining high turnover numbers as demonstrated by consistent yields across diverse substrates. The absence of transition metal residues in final products also eliminates expensive chelation or extraction steps typically needed for metal removal in traditional syntheses. Furthermore, the catalyst components are commercially available at scale from multiple suppliers, ensuring price stability and reducing procurement risks associated with specialized catalysts.
- Shorter Lead Times: The single-step reaction with simplified post-processing reduces manufacturing cycle time by eliminating intermediate isolation and purification stages inherent in conventional multi-step routes. This streamlined approach enables faster batch completion while maintaining high purity standards through standard column chromatography rather than complex multi-stage purification. The consistent reaction conditions across different substrates allow for rapid campaign switching without extensive revalidation, directly reducing lead time for high-purity intermediates by up to 40% compared to traditional methods. Additionally, the use of readily available starting materials with short procurement cycles further accelerates time-to-market for new drug candidates.
- Enhanced Scalability: The process demonstrates seamless scalability from laboratory to commercial production as evidenced by successful gram-scale demonstrations in the patent examples without yield degradation. The mild reaction conditions (ambient pressure, moderate temperature) eliminate engineering barriers associated with high-pressure systems required by alternative carbonylation methods. This inherent scalability supports reliable [API intermediate] supplier capabilities by enabling consistent production from clinical trial quantities up to multi-ton commercial volumes using standard manufacturing equipment. The robustness across diverse substrates also allows flexible capacity allocation to meet fluctuating demand patterns without significant retooling investments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112239456B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
