Innovative Palladium-Catalyzed Manufacturing Process for High-Purity Quinazolinone Derivatives with Commercial Scale-Up Capability
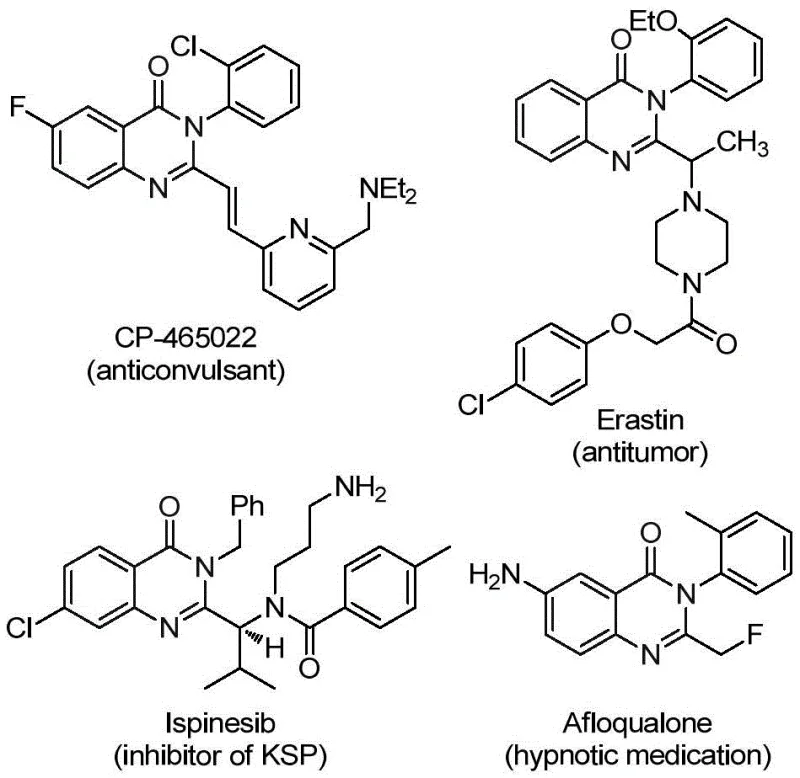
Patent CN112125856A introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone derivatives that serve as critical building blocks in modern pharmaceutical development due to their versatile biological activities including anti-inflammatory, antiviral, antifungal, anticonvulsant, and anticancer properties as evidenced by numerous marketed drugs containing this scaffold. This innovative approach addresses longstanding challenges in the field by utilizing a palladium-catalyzed carbonylation reaction that eliminates the need for hazardous gaseous carbon monoxide through strategic implementation of solid carbon monoxide surrogates like TFBen (1,3,5-tricarboxylate phenol ester), thereby significantly enhancing process safety while maintaining operational simplicity. The process demonstrates exceptional substrate tolerance across diverse functional groups including halogens, alkyl groups, and nitro substituents while maintaining cost-effectiveness through use of readily available starting materials such as o-iodoaniline and trifluoroethylimidoyl chloride precursors. Unlike conventional methods that suffer from narrow substrate scope and demanding reaction conditions requiring expensive pre-activated substrates or unstable reagents like trifluoroacetamide, this patented technique enables synthesis of structurally complex quinazolinone derivatives with unprecedented efficiency and safety profile suitable for commercial manufacturing environments. The significance of this advancement is further underscored by growing market demand for fluorinated compounds in drug discovery pipelines where the trifluoromethyl group enhances key pharmacological properties such as metabolic stability and bioavailability without introducing additional purification challenges.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinazolinone derivatives face significant constraints that hinder their commercial viability including harsh reaction conditions requiring elevated temperatures or pressures that increase operational risks while demanding specialized equipment not commonly available in standard manufacturing facilities. Conventional methods typically employ unstable reagents such as trifluoroacetamide or require expensive pre-functionalized substrates that substantially increase raw material costs while limiting structural diversity through narrow substrate scope that cannot accommodate various functional groups essential for modern drug development programs. These approaches often yield low product quantities due to competing side reactions under aggressive conditions while generating complex impurity profiles that necessitate extensive purification steps adding considerable time and expense to the manufacturing process. Furthermore, methods relying on gaseous carbon monoxide present severe safety hazards requiring specialized containment systems and rigorous monitoring protocols that complicate scale-up efforts and increase regulatory compliance burdens for pharmaceutical manufacturers seeking reliable supply chains. The cumulative effect of these limitations results in higher production costs, reduced process robustness, and limited commercial scalability that cannot meet growing industry demands for high-purity fluorinated intermediates.
The Novel Approach
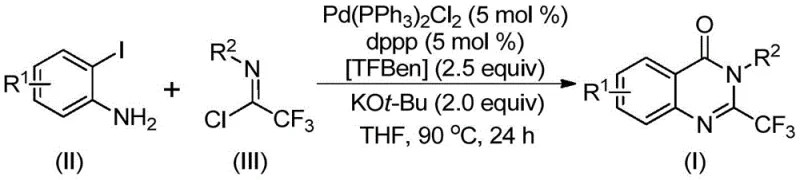
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that utilizes readily available starting materials including o-iodoaniline derivatives and trifluoroethylimidoyl chloride precursors under mild reaction conditions at precisely controlled temperature of 90°C without requiring pressurized systems or hazardous reagents. By implementing TFBen as a solid carbon monoxide surrogate instead of toxic gaseous CO, this approach eliminates significant safety risks while maintaining excellent reaction efficiency across diverse substrate combinations including those with halogen substituents or electron-withdrawing groups that previously caused complications in conventional syntheses. The process demonstrates remarkable functional group tolerance allowing synthesis of various substituted quinazolinones through simple modification of starting materials without requiring additional synthetic steps or specialized handling procedures. This streamlined methodology achieves high yields through optimized catalyst system comprising Pd(PPh₃)₂Cl₂/dppp combination with potassium tert-butoxide base while operating under standard atmospheric pressure conditions compatible with existing manufacturing infrastructure. The elimination of hazardous reagents combined with simplified workup procedures significantly enhances process safety profile while reducing overall production costs through decreased waste generation and minimized purification requirements.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism begins with potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate that establishes the foundational molecular architecture required for subsequent cyclization steps. This key transformation occurs under mild conditions without requiring additional catalysts or harsh reagents while maintaining excellent regioselectivity across diverse substrate combinations. The palladium catalyst then inserts into the carbon-iodine bond of the aryl iodide substrate generating a divalent palladium species that serves as the catalytic center for the carbonylation process through oxidative addition mechanism. Under thermal activation at precisely controlled temperature of 90°C, TFBen decomposes to release carbon monoxide in situ which subsequently inserts into the carbon-palladium bond forming an acyl palladium intermediate critical for carbonyl group incorporation essential to quinazolinone ring formation. The base facilitates deprotonation and nitrogen coordination creating a seven-membered palladacycle intermediate through intramolecular cyclization that directs regioselectivity toward desired product formation while minimizing competing pathways. Finally reductive elimination releases the desired quinazolinone product regenerating the active palladium catalyst for further catalytic cycles while maintaining high atom economy throughout the transformation sequence.
Impurity control is achieved through precise management of reaction parameters combined with inherent selectivity of the catalytic system where TFBen's controlled decomposition kinetics prevent side reactions by providing gradual carbon monoxide release rather than sudden gas introduction that could cause undesired pathways. The well-defined coordination environment created by dppp ligand stabilizes palladium intermediates against β-hydride elimination pathways that typically generate impurities in similar catalytic systems while directing regioselectivity toward desired cyclization products. Strategic substrate design allows placement of substituents that minimize competing reaction channels through electronic effects without requiring additional purification steps beyond standard workup procedures. The post-treatment protocol involving filtration followed by silica gel-assisted column chromatography effectively removes residual catalyst species and unreacted starting materials while preserving product integrity through gentle separation techniques compatible with sensitive fluorinated compounds. This integrated approach ensures consistent production of high-purity quinazolinone derivatives meeting stringent pharmaceutical quality standards across diverse substrate combinations while maintaining excellent yield profiles without introducing additional impurities during purification.

How to Synthesize Quinazolinone Derivatives Efficiently
This innovative synthesis route represents a significant advancement over conventional methodologies by enabling efficient production of structurally diverse quinazolinone derivatives through a streamlined catalytic process that eliminates hazardous reagents while maintaining excellent yield profiles across multiple substrate combinations as demonstrated in patent examples. The methodology leverages readily available starting materials including o-iodoaniline derivatives with various substituents (R¹ = H, F, Cl, Br, CF₃) combined with different aryl groups on trifluoroethylimidoyl chloride (R² = phenyl derivatives) to produce a wide range of target compounds with consistent quality metrics suitable for pharmaceutical applications. Detailed standardized synthesis steps incorporating precise stoichiometric ratios and optimized reaction parameters are provided below to ensure successful implementation in manufacturing environments where reproducibility and quality control are paramount considerations for regulatory compliance.
- Combine palladium catalyst (Pd(PPh₃)₂Cl₂), ligand (dppp), TFBen (1,3,5-tricarboxylate phenol ester), potassium tert-butoxide, trifluoroethylimidoyl chloride, and o-iodoaniline in THF solvent under inert atmosphere with precise stoichiometric ratios.
- Heat the reaction mixture to precisely controlled temperature of 90°C and maintain for optimal duration between 16 to 30 hours to ensure complete conversion while preventing side reactions.
- Perform post-treatment through filtration to remove solids, followed by silica gel mixing and column chromatography purification to isolate high-purity quinazolinone derivatives meeting pharmaceutical standards.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial commercial benefits specifically addressing critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its elimination of hazardous materials handling requirements while maintaining excellent process economics suitable for large-scale production environments where reliability is paramount. The approach directly tackles common supply chain vulnerabilities by utilizing widely available starting materials that can be sourced from multiple global suppliers without requiring specialized handling or storage conditions that typically complicate logistics networks for sensitive chemical intermediates.
- Cost Reduction in Manufacturing: Elimination of toxic carbon monoxide gas handling reduces safety-related operational costs through simplified facility requirements while avoiding expensive containment systems typically needed for pressurized gas operations; substitution with solid TFBen surrogate streamlines regulatory compliance procedures reducing associated administrative burdens; utilization of inexpensive starting materials like o-iodoaniline derivatives combined with efficient catalytic system minimizes raw material expenses without compromising product quality; simplified workup procedure eliminates multiple purification steps required by conventional methods thereby reducing overall production time and associated labor costs.
- Enhanced Supply Chain Reliability: Broad substrate availability ensures consistent access to starting materials from multiple global suppliers reducing single-source dependency risks; elimination of hazardous reagents simplifies transportation logistics by removing special handling requirements; modular nature allows rapid adaptation to different product variants without major process modifications enabling flexible response to changing market demands; compatibility with standard manufacturing equipment facilitates seamless integration into existing production facilities without requiring capital-intensive upgrades.
- Scalability and Environmental Compliance: Process operates under standard atmospheric pressure conditions enabling straightforward scale-up from laboratory to commercial production volumes; elimination of toxic gaseous reagents significantly reduces environmental impact while simplifying waste treatment protocols; simplified purification requirements minimize solvent usage contributing to greener manufacturing practices; robust performance across diverse substrates ensures consistent quality during scale-up without requiring extensive reoptimization efforts typically associated with complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding implementation feasibility and quality assurance aspects specific to this patented manufacturing process based on detailed analysis of patent documentation including experimental procedures and performance metrics reported in supporting examples.
Q: How does this method overcome limitations of conventional quinazolinone synthesis?
A: This patented approach eliminates harsh reaction conditions by replacing toxic gaseous carbon monoxide with solid TFBen surrogate while maintaining excellent substrate tolerance across diverse functional groups. The process avoids expensive pre-activated substrates through direct coupling of readily available o-iodoaniline and trifluoroethylimidoyl chloride precursors under mild conditions.
Q: What are key advantages of using TFBen as carbon monoxide substitute?
A: TFBen provides controlled in situ CO release that prevents hazardous gas handling while ensuring consistent reaction kinetics. This solid surrogate eliminates safety risks associated with pressurized CO systems and simplifies regulatory compliance without compromising yield or purity in pharmaceutical intermediate manufacturing.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The catalytic system's inherent selectivity minimizes side reactions through precise ligand control and optimized reaction parameters. Combined with straightforward post-treatment involving filtration and column chromatography, this method consistently delivers high-purity quinazolinone derivatives meeting stringent pharmaceutical quality requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivative Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-million levels. NINGBO INNO PHARMCHEM's CDMO expertise ensures seamless transition from laboratory-scale development to full commercial manufacturing through comprehensive process validation protocols that guarantee consistent product quality meeting international regulatory standards across all production volumes.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements; our specialists stand ready to collaborate on optimizing this innovative synthesis pathway for your particular application needs while ensuring maximum value delivery throughout your supply chain.
