Advanced Silver-Catalyzed Synthesis of Polysubstituted Pyrroles for Pharmaceutical Intermediates
Advanced Silver-Catalyzed Synthesis of Polysubstituted Pyrroles for Pharmaceutical Intermediates
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation driven by the demand for greener, more efficient methodologies. Patent CN103145600A introduces a groundbreaking approach to constructing polysubstituted pyrrole scaffolds, which are ubiquitous motifs in bioactive molecules and advanced materials. This technology leverages a silver carbonate (Ag2CO3) catalyzed [3+2] cycloaddition between readily available alkynes and isocyanides. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift away from classical, waste-intensive routes toward a streamlined, atom-economical process. The method operates under remarkably mild conditions, typically utilizing 1,4-dioxane as a solvent at temperatures ranging from 20°C to 100°C, ensuring high purity and simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrole ring has relied heavily on classical condensation reactions such as the Knorr pyrrole synthesis, the Hantzsch pyrrole synthesis, or the Paal-Knorr condensation. While these methods are well-established, they suffer from inherent limitations that hinder modern process chemistry objectives. Traditional routes often necessitate the use of strong acids or bases, leading to harsh reaction environments that are incompatible with sensitive functional groups frequently found in complex drug candidates. Furthermore, these multi-step sequences frequently exhibit poor atom economy, generating substantial quantities of stoichiometric salt waste and requiring rigorous purification steps to remove trace metal impurities or side products. The reliance on specific 1,4-dicarbonyl precursors also limits the structural diversity accessible in a single step, often forcing synthetic chemists into lengthy linear sequences to install diverse substituents at the 2, 3, 4, and 5 positions of the pyrrole ring.
The Novel Approach
In stark contrast, the silver-catalyzed methodology described in the patent offers a convergent strategy that directly assembles the heterocyclic core from simple building blocks. By utilizing terminal or internal alkynes and isocyanides, this [3+2] cyclization allows for the simultaneous introduction of multiple substituents in a single operational step. The use of silver carbonate as a mild Lewis acid catalyst activates the alkyne moiety towards nucleophilic attack by the isocyanide carbon, facilitating ring closure without the need for extreme thermal energy or corrosive reagents. As demonstrated in the specific example of converting phenylacetylene derivatives into ethyl 4-phenyl-1H-pyrrole-3-carboxylates, the reaction proceeds with high efficiency. This approach not only simplifies the synthetic route but also significantly enhances the structural diversity accessible to medicinal chemists, enabling the rapid generation of libraries for SAR studies.
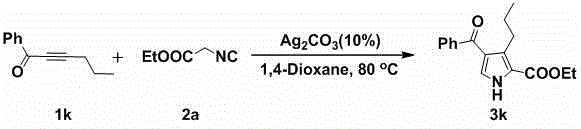
Mechanistic Insights into Ag2CO3-Catalyzed [3+2] Cycloaddition
The mechanistic pathway of this transformation involves the coordination of the silver cation to the pi-system of the alkyne, which increases the electrophilicity of the triple bond. This activation is crucial for the subsequent nucleophilic attack by the divalent carbon of the isocyanide species. Following the initial addition, a series of intramolecular rearrangements and proton transfers occur, ultimately leading to the aromatization of the five-membered ring. The choice of silver carbonate is particularly strategic; it acts as a heterogeneous or semi-heterogeneous catalyst that provides a steady release of active silver species while minimizing the formation of colloidal silver aggregates that could complicate filtration. The reaction environment, typically polar aprotic solvents like 1,4-dioxane or DMF, stabilizes the charged intermediates formed during the cyclization process, ensuring smooth progression to the final polysubstituted pyrrole product.
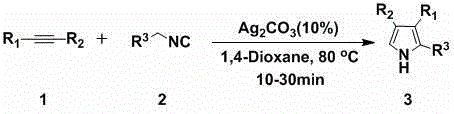
From an impurity control perspective, the mildness of the silver catalytic system is a distinct advantage. High-temperature processes often lead to polymerization of alkynes or decomposition of the sensitive isocyanide functionality, resulting in complex tarry byproducts that are difficult to separate. By maintaining reaction temperatures between 60°C and 80°C for most substrates, this method suppresses these degradation pathways. Furthermore, the specificity of the silver catalyst for the alkyne-isocyanide coupling minimizes homocoupling side reactions of the alkyne (Glaser coupling), which is a common issue in copper-catalyzed systems. This high chemoselectivity translates directly to cleaner crude reaction profiles, reducing the burden on purification teams and improving the overall yield of the desired high-purity pharmaceutical intermediate.
How to Synthesize Polysubstituted Pyrroles Efficiently
The operational simplicity of this silver-catalyzed protocol makes it highly attractive for both laboratory scale-up and industrial manufacturing. The procedure typically involves charging a pressure-resistant vessel with the alkyne substrate, the isocyanide component (often used in a slight excess to drive conversion), and the silver carbonate catalyst in the chosen solvent. The detailed standardized synthesis steps, including precise stoichiometric ratios, agitation speeds, and workup procedures, are outlined in the guide below to ensure reproducibility and safety compliance.
- Charge a pressure-resistant reactor with 1,4-dioxane solvent, the terminal or internal alkyne substrate, and ethyl isocyanoacetate in a molar ratio of approximately 1: 1.5.
- Add 10 mol% of silver carbonate (Ag2CO3) catalyst to the reaction mixture and stir thoroughly to ensure homogeneous suspension before heating.
- Heat the reaction mixture to 80°C in an oil bath until TLC indicates complete consumption of the starting alkyne, then proceed to aqueous workup and silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the accessibility and cost profile of the raw materials. Alkynes and isocyanides are commodity chemicals available from a robust global supply chain, reducing the risk of raw material shortages that often plague specialized reagent-dependent processes. Additionally, the elimination of harsh acidic or basic reagents simplifies the engineering requirements for the reaction vessels, allowing for the use of standard stainless steel reactors without the need for exotic corrosion-resistant linings. This compatibility with standard manufacturing infrastructure significantly lowers the barrier to entry for commercial scale-up.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the [3+2] cycloaddition, where nearly all atoms from the starting materials are incorporated into the final product. By avoiding multi-step protection and deprotection sequences typically required in classical pyrrole synthesis, the overall number of unit operations is drastically reduced. This consolidation of steps leads to substantial savings in labor, energy consumption, and solvent usage. Furthermore, the use of a relatively inexpensive silver salt catalyst, as opposed to precious metals like palladium or rhodium, ensures that the catalyst cost does not become a prohibitive factor in large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain stability. Because the reaction tolerates a wide range of functional groups and proceeds under mild thermal conditions, the risk of batch failure due to thermal runaway or sensitivity to moisture is minimized. This reliability ensures consistent delivery schedules for downstream customers who rely on these intermediates for API synthesis. The ability to source generic solvents like 1,4-dioxane or DMF further decouples the manufacturing process from volatile specialty chemical markets, providing a stable foundation for long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with green chemistry principles. The reduction in waste generation and the avoidance of toxic heavy metals or corrosive acids simplify the wastewater treatment process. The straightforward workup procedure, often involving simple extraction and crystallization, reduces the volume of organic waste solvents requiring disposal. This environmental profile not only lowers compliance costs but also enhances the sustainability credentials of the final product, a factor increasingly weighted by global pharmaceutical buyers in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed pyrrole synthesis technology. These insights are derived directly from the experimental data and scope evaluation presented in the patent literature, providing a realistic overview of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: What are the advantages of using silver carbonate over traditional pyrrole synthesis methods?
A: Unlike traditional Knorr or Paal-Knorr syntheses which often require harsh acidic conditions and generate significant waste, the silver carbonate catalyzed [3+2] cycloaddition operates under mild neutral conditions (20-100°C) with high atom economy and broader functional group tolerance.
Q: Can this method tolerate sensitive functional groups like ketones or esters?
A: Yes, the protocol demonstrates excellent compatibility with various functional groups including esters, ketones, hydroxyls, and heteroaryl rings, allowing for the direct synthesis of complex intermediates without extensive protecting group strategies.
Q: Is the silver catalyst recoverable for large-scale production?
A: While the patent specifies a 10% loading of Ag2CO3, silver salts can often be recovered from the reaction residue through standard filtration and regeneration processes, contributing to long-term cost efficiency in industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrroles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocycle synthesis plays in accelerating drug discovery and development pipelines. Our technical team has extensively evaluated the silver-catalyzed pathway described in CN103145600A and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profiles of these complex pyrrole derivatives to meet the highest pharmacopeial standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecule. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our catalog compounds, and receive comprehensive route feasibility assessments that demonstrate how we can optimize your supply chain for cost and efficiency.
