Revolutionizing Polysubstituted Pyrrole Manufacturing with Base-Catalyzed Green Chemistry
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex heterocyclic scaffolds, particularly nitrogen-containing five-membered rings like pyrroles which are ubiquitous in bioactive natural products and drug candidates. A significant breakthrough in this domain is documented in Chinese Patent CN110683979B, which discloses a novel method for the synthesis of polysubstituted pyrrole derivatives catalyzed by sodium hydroxide. This technology represents a paradigm shift from traditional transition-metal-catalyzed processes to a greener, base-mediated approach that utilizes readily available aurone derivatives and isocyanoacetates. By leveraging a 1,3-dipolar cycloaddition followed by a ring-opening isomerization, this methodology achieves high atom economy and exceptional yields under remarkably mild conditions. For R&D directors and procurement specialists alike, this patent offers a compelling solution to the longstanding challenges of metal contamination and expensive catalyst loading, positioning it as a critical asset for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted pyrroles via isocyanide chemistry has relied heavily on the use of stoichiometric strong bases or expensive transition metal catalysts, presenting significant hurdles for industrial application. Early methodologies, such as those reported by Sasaki, utilized DBU but suffered from limited substrate scope and modest yields around 10%, restricting their utility. Subsequent advancements by groups like de Meijere, Bi, Lei, and Tiwari introduced copper, silver, and rhodium catalysts to improve efficiency; however, these approaches inherently introduce heavy metals into the reaction system. For pharmaceutical manufacturing, the presence of residual metals like silver, copper, or rhodium in the final active pharmaceutical ingredient (API) is a critical quality attribute that requires stringent control, often necessitating additional purification steps such as metal scavenging or recrystallization. Furthermore, many of these metal-catalyzed protocols demand rigorous anhydrous and anaerobic conditions, increasing operational complexity and energy consumption. The reliance on precious metals also exposes the supply chain to price volatility and availability risks, making cost reduction in API manufacturing difficult to achieve consistently.
The Novel Approach
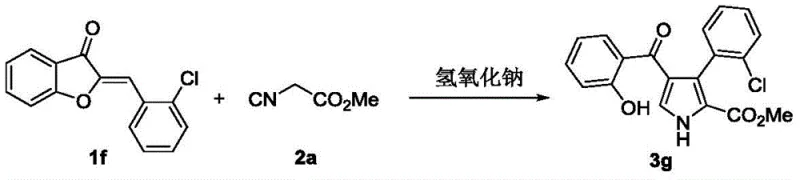
In stark contrast to the metal-dependent legacy methods, the invention detailed in CN110683979B utilizes sodium hydroxide, a ubiquitous and inexpensive inorganic base, to drive the cyclization of aurones and isocyanoacetates. This novel approach completely bypasses the need for transition metals, thereby eliminating the risk of heavy metal contamination at the source. The reaction operates efficiently at temperatures ranging from 0°C to 40°C, with a preference for ambient conditions (25°C), and tolerates common solvents like methanol without the need for specialized drying. As illustrated in the general reaction scheme below, the process converts aurone derivatives and isocyanoacetates directly into 2,3,4-polysubstituted pyrroles through a streamlined cascade.

The operational simplicity is further enhanced by the fact that the reaction does not require inert atmosphere protection, allowing for open-vessel processing which drastically simplifies reactor requirements. Experimental data within the patent demonstrates that while other catalysts like silver oxide or copper acetate yield poor results (often below 25%), the optimized sodium hydroxide system consistently delivers yields exceeding 90% across a wide array of substrates. This robustness makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a direct path to reducing lead time for high-purity pyrrole derivatives.
Mechanistic Insights into NaOH-Catalyzed 1,3-Dipolar Cycloaddition
The mechanistic pathway of this transformation is a sophisticated yet elegant sequence initiated by the deprotonation of the isocyanoacetate alpha-carbon by the hydroxide ion. This deprotonation generates a reactive 1,3-dipole species, which subsequently undergoes a nucleophilic attack on the electron-deficient double bond of the aurone derivative. This initial addition forms a spirocyclic pyrroline intermediate, a key structural motif that dictates the regioselectivity of the final product. Unlike metal-catalyzed pathways that might involve complex coordination spheres or oxidative addition steps, this base-mediated mechanism relies purely on electronic activation and steric factors, ensuring high fidelity in product formation. The spiro-intermediate then undergoes a spontaneous ring-opening isomerization, driven by the restoration of aromaticity in the pyrrole ring and the stabilization of the phenolic hydroxyl group on the benzoyl moiety. This cascade effectively constructs the highly substituted pyrrole core in a single pot.
From an impurity control perspective, the absence of metal catalysts significantly simplifies the impurity profile. In metal-catalyzed reactions, side products often arise from homocoupling of the isocyanide or incomplete conversion due to catalyst deactivation. In this NaOH-catalyzed system, the primary byproducts are minimal, and the reaction proceeds to near-completion as evidenced by the high isolated yields reported in the examples. For instance, the synthesis of compound 3g using an ortho-chloro substituted aurone achieved a remarkable 98% yield, demonstrating the method's tolerance to halogen substituents which are often sensitive in cross-coupling chemistries. The specificity of the base catalysis ensures that functional groups such as esters, ethers, and even sensitive heterocycles like furan and thiophene remain intact, providing a clean reaction profile that facilitates downstream purification.
How to Synthesize Polysubstituted Pyrroles Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves dissolving the aurone substrate and the sodium hydroxide catalyst in methanol, followed by the controlled addition of the isocyanoacetate. Reaction progress is easily monitored via thin-layer chromatography (TLC), and upon completion, the workup involves simple concentration and silica gel column chromatography. This straightforward procedure minimizes the need for specialized equipment or hazardous reagents. For detailed standardized synthesis steps and specific molar ratios optimized for different substrates, please refer to the guide below.
- Dissolve the aurone derivative substrate and sodium hydroxide catalyst (20 mol%) in methanol solvent within a reaction vessel equipped with stirring.
- Add methyl isocyanoacetate dropwise to the mixture at 25°C and stir until TLC monitoring confirms complete consumption of the starting material.
- Concentrate the reaction mixture and purify the crude product via silica gel column chromatography to isolate the high-purity polysubstituted pyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sodium hydroxide-catalyzed methodology offers transformative economic and logistical benefits. The most immediate impact is seen in raw material costs; replacing expensive silver or copper salts with commodity-grade sodium hydroxide results in substantial cost savings per kilogram of product. Additionally, the elimination of metal scavengers and the associated filtration steps reduces the consumption of auxiliary materials and shortens the overall batch cycle time. The robustness of the reaction conditions means that production is less susceptible to delays caused by stringent environmental controls, enhancing supply chain reliability. Furthermore, the high atom economy and yield minimize waste generation, aligning with increasingly strict environmental regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with sodium hydroxide removes a significant cost driver from the bill of materials. Transition metals like silver and rhodium are subject to market fluctuations and can constitute a large portion of the variable cost in fine chemical synthesis. By utilizing a base metal-free approach, manufacturers can achieve a drastically simplified cost structure. Moreover, the high yields (often >90%) reduce the amount of starting material required per unit of output, further driving down the cost of goods sold (COGS). The simplified workup also reduces labor and utility costs associated with extended purification processes.
- Enhanced Supply Chain Reliability: Reliance on specialized transition metal catalysts can introduce supply chain vulnerabilities, especially when specific ligands or high-purity metal salts are required. Sodium hydroxide is a globally available commodity chemical with a stable supply chain, ensuring continuity of production. The ability to run the reaction without inert gas protection (nitrogen or argon) reduces dependency on industrial gases and allows for more flexible scheduling in multipurpose facilities. This operational flexibility translates to shorter lead times and the ability to respond more rapidly to market demand spikes for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling up metal-catalyzed reactions often presents challenges related to heat transfer and metal removal at large volumes. This base-catalyzed process is inherently safer and easier to scale, as it avoids exothermic risks associated with some metal activations and eliminates the need for complex metal recovery systems. From an environmental standpoint, the absence of heavy metals in the waste stream simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This compliance advantage is crucial for maintaining operating licenses and meeting the sustainability goals of modern pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the advantages of using NaOH over transition metal catalysts for pyrrole synthesis?
A: Using sodium hydroxide eliminates the risk of toxic heavy metal residues (like Ag, Cu, Rh) in the final API, simplifies purification by removing metal scavenging steps, and significantly reduces raw material costs compared to precious metal catalysts.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under ambient conditions without the need for rigorous drying of solvents or inert gas protection, facilitating easier scale-up.
Q: What is the substrate scope for this pyrrole synthesis method?
A: The method demonstrates broad compatibility with various aurone derivatives containing electron-donating or withdrawing groups, heterocycles like thiophene and furan, and even bulky groups like ferrocene and naphthyl, yielding products up to 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrroles Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the NaOH-catalyzed pyrrole synthesis in driving the next generation of pharmaceutical developments. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of polysubstituted pyrroles meets the highest international standards for API intermediates. We are committed to leveraging such green chemistry advancements to deliver superior value to our global clientele.
We invite potential partners to engage with our technical procurement team to discuss how this specific technology can be tailored to your project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free route for your specific target molecules. We encourage you to contact us today to obtain specific COA data for our library of pyrrole derivatives and to receive comprehensive route feasibility assessments that will accelerate your drug development timelines.
