Scalable Enantioselective Synthesis of High-Purity Tetrahydroquinoline Cardiovascular Intermediate
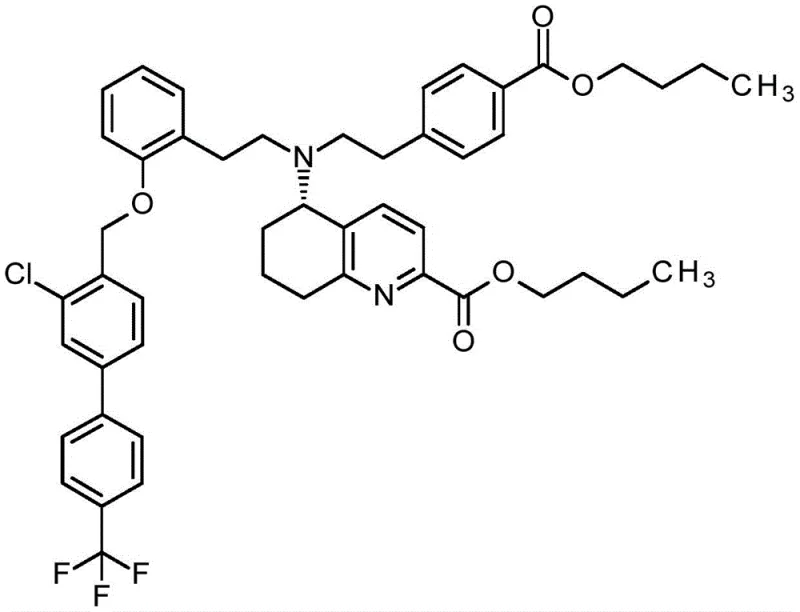
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex active pharmaceutical ingredients, particularly those targeting critical cardiovascular conditions. Patent CN115803318A introduces a groundbreaking methodology for the preparation of (5S)-5-({2-[4-(butoxycarbonyl)phenyl]ethyl}[2-(2-{[3-chloro-4'-(trifluoromethyl)[biphenyl]-4-yl]methoxy}phenyl)ethyl]amino)-5,6,7,8-tetrahydroquinoline-2-carboxylic acid butyl ester, a pivotal intermediate for soluble guanylate cyclase activators. This compound class holds immense therapeutic potential for treating pulmonary arterial hypertension and related cardiopulmonary diseases. The disclosed innovation addresses long-standing manufacturing bottlenecks by implementing an enantioselective synthesis strategy that bypasses the need for inefficient chiral chromatography. By integrating advanced catalytic reduction and optimized purification via salt formation, this technical breakthrough offers a viable route for producing high-purity pharmaceutical intermediates with enhanced reproducibility. For global supply chains, this represents a shift towards more sustainable and economically feasible production of life-saving cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for this class of tetrahydroquinoline derivatives, such as those described in WO 2014/012934, suffer from severe industrial limitations that hinder cost-effective mass production. The prior art methodologies typically require an excessive number of synthetic steps, often exceeding seventeen distinct stages, which inherently compounds yield losses and operational complexity. A critical drawback is the reliance on late-stage chiral chromatography to separate enantiomers from a racemic mixture, a process that is technically demanding, solvent-intensive, and economically prohibitive at scale. Furthermore, conventional methods often involve reaction times extending over several days and utilize high dilution conditions that drastically reduce batch throughput. The accumulation of impurities necessitates up to seven chromatographic purification steps, creating significant waste streams and increasing the environmental footprint. These factors collectively render traditional approaches unsuitable for the rigorous demands of modern commercial pharmaceutical manufacturing.
The Novel Approach
The innovative process detailed in CN115803318A fundamentally reengineers the synthetic pathway to overcome these inefficiencies through early-stage stereocontrol and simplified purification. By employing an enantioselective reduction at the beginning of the synthesis, the method establishes the required chiral center with high fidelity, thereby eliminating the need for downstream chiral resolution. The strategy replaces multiple chromatographic steps with robust crystallization and salt formation techniques, which are far more amenable to large-scale processing. This approach significantly reduces the total number of synthetic stages and minimizes solvent consumption, leading to a drastic simplification of the overall workflow. The use of specific sterically hindered bases and optimized reaction conditions further enhances yield and purity, ensuring that the final product meets stringent regulatory standards without the burden of complex purification. This novel methodology transforms the production landscape for these complex intermediates.
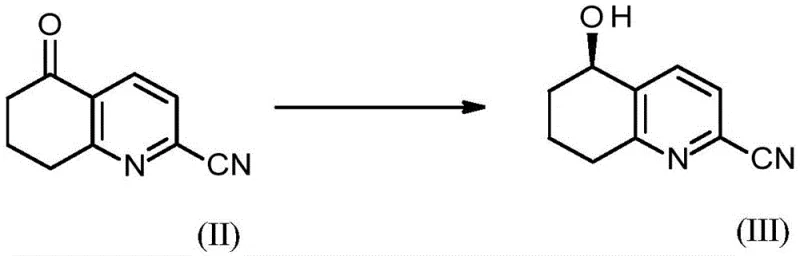
Mechanistic Insights into Enantioselective Reduction and Coupling
The cornerstone of this advanced synthesis lies in the enantioselective reduction of 5-oxo-5,6,7,8-tetrahydroquinoline-2-carbonitrile to form the chiral alcohol intermediate. This transformation utilizes a sophisticated ruthenium catalyst system, specifically (p-cymene)[(R,R)-N-(p-toluenesulfonyl)-1,2-diphenylethylenediamine]ruthenium, in the presence of formic acid and a tertiary amine base. The reaction proceeds under mild thermal conditions, typically between 20°C to 50°C, to achieve high conversion rates while maintaining exceptional enantiomeric excess. This catalytic cycle ensures that the chiral information is introduced early, preventing the formation of unwanted diastereomers in subsequent steps. The mechanism avoids the thermodynamic pitfalls of racemic synthesis, providing a direct route to the desired (5R)-hydroxy configuration which is essential for the biological activity of the final API. This level of stereochemical control is critical for ensuring the safety and efficacy of the resulting cardiovascular therapeutic.
Following the establishment of chirality, the synthesis proceeds through a series of carefully orchestrated coupling and protection steps designed to maximize efficiency. The process involves the alkylation of the chiral intermediate with protected amine fragments using sterically hindered secondary amines like diisopropylamine as bases. This specific choice of base is crucial as it suppresses polyalkylation side reactions and promotes the formation of the desired monoalkylated product with high selectivity. Subsequent steps involve the strategic use of silyl protecting groups to mask reactive hydroxyl functionalities, allowing for orthogonal deprotection later in the sequence. The final assembly includes hydrolysis of nitrile groups to carboxylic acids followed by esterification and a final alkylation with a biphenyl derivative. Each step is optimized to allow for isolation via crystallization or extraction, avoiding the need for chromatography and ensuring that impurities are effectively removed at each stage of the synthesis.
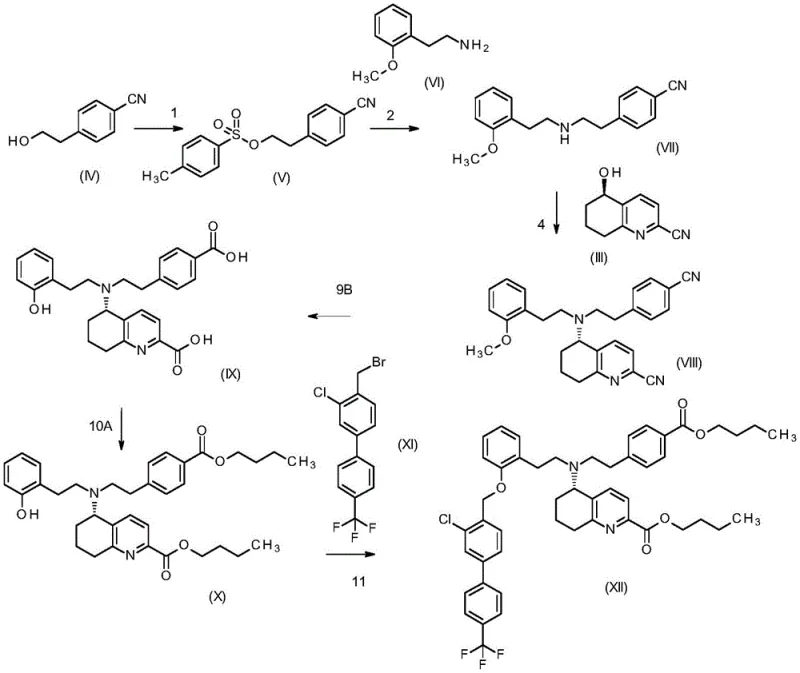
How to Synthesize Tetrahydroquinoline Derivative Efficiently
Implementing this synthesis requires precise control over reaction parameters and a deep understanding of the intermediate stability profiles. The process begins with the catalytic reduction step, followed by coupling reactions that must be conducted under anhydrous conditions to prevent catalyst deactivation. Detailed standardized synthesis steps see the guide below.
- Perform enantioselective reduction of 5-oxo-5,6,7,8-tetrahydroquinoline-2-carbonitrile using a ruthenium catalyst to establish chirality.
- Execute coupling reactions with protected amine intermediates using sterically hindered bases to ensure high yield and purity.
- Complete the synthesis via hydrolysis, esterification, and final alkylation with biphenyl derivatives to form the target butyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic benefits for procurement and supply chain management within the pharmaceutical sector. The elimination of chiral chromatography and the reduction in total synthetic steps directly translate to a significant decrease in manufacturing costs and production lead times. By removing the dependency on expensive chiral stationary phases and vast quantities of chromatographic solvents, the process achieves a leaner operational model that is less susceptible to raw material price volatility. The robustness of the crystallization-based purification steps enhances supply chain reliability, as these unit operations are more easily scaled and controlled compared to complex chromatographic separations. This stability ensures a consistent supply of high-quality intermediates, mitigating the risk of production delays that can impact downstream API manufacturing schedules.
- Cost Reduction in Manufacturing: The streamlined synthesis route drastically lowers the cost of goods sold by removing expensive purification technologies and reducing solvent waste. The avoidance of chiral chromatography eliminates a major cost driver associated with resin replacement and solvent disposal, while the reduced step count minimizes labor and equipment occupancy time. Furthermore, the use of readily available reagents and catalysts in catalytic amounts ensures that material costs remain optimized without compromising on reaction efficiency. This economic efficiency allows for more competitive pricing structures in the global market for cardiovascular drug intermediates.
- Enhanced Supply Chain Reliability: The simplified process flow enhances supply chain resilience by reducing the number of critical control points and potential failure modes. With fewer synthetic steps and more robust purification methods, the risk of batch failure due to purification issues is significantly diminished. The ability to isolate intermediates as stable salts or crystalline solids improves inventory management and allows for flexible production scheduling. This reliability is crucial for maintaining continuous supply to API manufacturers, ensuring that patient demand for essential cardiovascular medications is met without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing unit operations that are standard in industrial chemical manufacturing. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations and corporate sustainability goals. By minimizing the environmental footprint through efficient atom economy and waste reduction, manufacturers can achieve compliance more easily and reduce the costs associated with environmental remediation. This scalability ensures that production can be ramped up to meet market demand without the need for specialized or hard-to-source equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis pathway. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: How does this process improve upon prior art synthesis methods?
A: The new process eliminates the need for seven chromatographic purification steps and a chiral chromatography stage required in previous methods, significantly reducing solvent consumption and technical complexity.
Q: What is the key mechanism for ensuring enantiomeric purity?
A: Enantiomeric purity is achieved early in the synthesis via an enantioselective reduction using a ruthenium-p-cymene-R,R-TsDPEN catalyst, avoiding the need for late-stage resolution of racemates.
Q: Is this synthesis suitable for industrial scale-up?
A: Yes, the process is designed for industrial feasibility by replacing complex chromatographic separations with crystallization and salt formation steps, which are more robust and cost-effective at large scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of enantioselective catalysis and purification are maintained at scale. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetrahydroquinoline derivative meets the highest international standards. Our commitment to quality and technical excellence makes us the ideal partner for pharmaceutical companies seeking to secure their supply chain for critical cardiovascular intermediates.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant efficiencies in your manufacturing operations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your project goals. Let us help you navigate the complexities of fine chemical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
