Advanced One-Pot Synthesis of Pyrazolopyrimidine Derivatives for Commercial Scale-Up
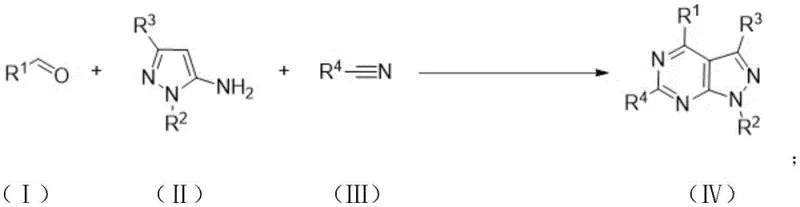
The chemical landscape for nitrogen-containing fused ring compounds is continuously evolving, driven by the demand for more efficient and environmentally benign synthetic routes. Patent CN115304607A introduces a groundbreaking methodology for the preparation of pyrazolopyrimidine derivatives, utilizing an iodine-catalyzed one-pot reaction system. This innovation addresses critical bottlenecks in the synthesis of these valuable scaffolds, which are increasingly recognized for their utility in both pharmaceutical interventions and advanced electronic materials. By leveraging a three-component coupling strategy involving aldehydes, 5-aminopyrazoles, and nitriles, the process achieves high atom economy and operational simplicity. For industry leaders seeking a reliable pyrazolopyrimidine supplier, this technology represents a significant leap forward in process chemistry, offering a robust pathway to access complex molecular architectures that were previously difficult to manufacture cost-effectively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrazolopyrimidine molecular skeleton has been fraught with significant technical challenges that hinder large-scale adoption. Traditional synthetic methodologies often necessitate multi-step sequences, which inherently accumulate inefficiencies and reduce overall throughput in a manufacturing setting. These legacy processes frequently rely on harsh reaction conditions and the use of toxic, hazardous organic reagents that pose substantial safety risks and environmental compliance burdens. Furthermore, conventional routes often suffer from poor substrate compatibility, limiting the diversity of derivatives that can be practically accessed without extensive process re-optimization. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and incapable of meeting the rigorous purity standards required for high-value applications in medicine and optoelectronics.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a streamlined one-pot cyclization strategy that fundamentally reshapes the production economics. By employing an iodine catalyst under oxidative conditions, the reaction efficiently constructs polysubstituted pyrazolopyrimidine derivatives with exceptional selectivity and yield. This method eliminates the need for intermediate isolation steps, thereby drastically reducing solvent consumption and waste generation associated with traditional multi-step syntheses. The use of low-toxicity or non-toxic organic reagents further aligns the process with modern green chemistry principles, ensuring a safer working environment and simplified regulatory approval pathways. This technological shift enables cost reduction in pharmaceutical intermediate manufacturing by simplifying the workflow and enhancing the overall sustainability profile of the production line.
Mechanistic Insights into Iodine-Catalyzed Cyclization
The underlying mechanism of this transformation involves a sophisticated sequence of nucleophilic additions and cyclization events orchestrated by the iodine catalyst. The reaction initiates with the activation of the aldehyde component, facilitating a condensation with the 5-aminopyrazole to form a key intermediate. Subsequently, a Ritter-type amination occurs involving the nitrile species, which is critical for introducing the necessary nitrogen atoms into the forming ring system. The final cyclization step closes the pyrimidine ring, driven by the oxidative conditions provided by oxygen or air, which regenerates the active catalytic species. This mechanistic pathway is highly advantageous because it avoids the use of expensive transition metal catalysts that often leave behind difficult-to-remove residues, thus ensuring high-purity pyrazolopyrimidine derivatives suitable for sensitive electronic applications.
Impurity control is a paramount concern for R&D directors, and this catalytic system demonstrates remarkable specificity in managing side reactions. The high selectivity of the iodine-mediated process minimizes the formation of by-products that typically complicate downstream purification efforts. By operating under mild thermal conditions, typically between 110°C and 140°C, the process prevents the thermal degradation of sensitive functional groups that might be present on the diverse array of substrates. This robustness allows for the commercial scale-up of complex heterocyclic intermediates without compromising on the quality of the final output. The ability to tolerate a wide range of substituents, including halogens and electron-withdrawing groups, ensures that the impurity profile remains consistent and manageable across different batches.
How to Synthesize Pyrazolopyrimidine Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and reproducibility. The process begins with the careful selection of stoichiometric ratios, where the aldehyde and 5-aminopyrazole are typically mixed in a molar ratio ranging from 1:1 to 2:1 to drive the equilibrium forward. The reaction is conducted in a solvent such as acetonitrile, which can also serve as a reactant in certain variations, under an atmosphere of oxygen or air to sustain the catalytic cycle. Heating is maintained for a duration of 10 to 20 hours to ensure complete conversion, after which the mixture is cooled and subjected to standard extraction and purification protocols. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Mix aldehyde compounds, 5-aminopyrazole compounds, nitrile compounds, and an iodine catalyst in a suitable organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 110°C and 140°C under an oxygen or air atmosphere for 10 to 20 hours.
- Cool the reaction to room temperature, perform extraction and washing, then purify the crude product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes the need for costly and time-consuming heavy metal scavenging steps, which directly contributes to substantial cost savings in the overall manufacturing budget. Additionally, the use of readily available starting materials such as common aldehydes and nitriles ensures a stable supply chain that is less susceptible to market volatility compared to specialized reagents. The simplicity of the workup procedure, involving basic extraction and crystallization, reduces the operational burden on production facilities and shortens the turnaround time for batch completion. These factors collectively enhance supply chain reliability and position the manufacturer to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by removing the dependency on precious metal catalysts that are traditionally used in similar cyclization reactions. This substitution not only lowers the direct material costs but also reduces the expenses associated with waste disposal and catalyst recovery systems. Furthermore, the high atom economy of the one-pot reaction ensures that a greater proportion of raw materials are converted into the desired product, minimizing waste and maximizing resource utilization. These efficiencies compound to deliver a more competitive pricing structure for the final chemical intermediates without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the requisite raw materials are commodity chemicals with established global supply networks. Unlike proprietary reagents that may face availability constraints, the aldehydes and aminopyrazoles used in this process are widely produced, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing an additional layer of security against supply chain disruptions. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream clients.
- Scalability and Environmental Compliance: The method is explicitly designed to be scalable from gram-level laboratory experiments to multi-kilogram or ton-scale commercial production without fundamental changes to the chemistry. The use of green solvents and the absence of highly toxic reagents simplify the environmental permitting process and reduce the liability associated with hazardous chemical handling. Waste streams are easier to treat due to the simpler composition of the reaction by-products, aligning with increasingly stringent global environmental regulations. This scalability ensures that the technology can grow with the business, supporting increasing volume requirements while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new product development pipelines. We encourage stakeholders to review these insights to fully appreciate the value proposition offered by this advanced chemical process.
Q: What are the advantages of this iodine-catalyzed method over traditional synthesis?
A: This method offers high atom economy, excellent selectivity, and operates under mild conditions using low-toxicity reagents, overcoming the multi-step and hazardous nature of conventional routes.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is scalable to gram-level or larger production, utilizing common organic solvents and simple post-reaction processing suitable for manufacturing.
Q: What are the primary applications of these derivatives?
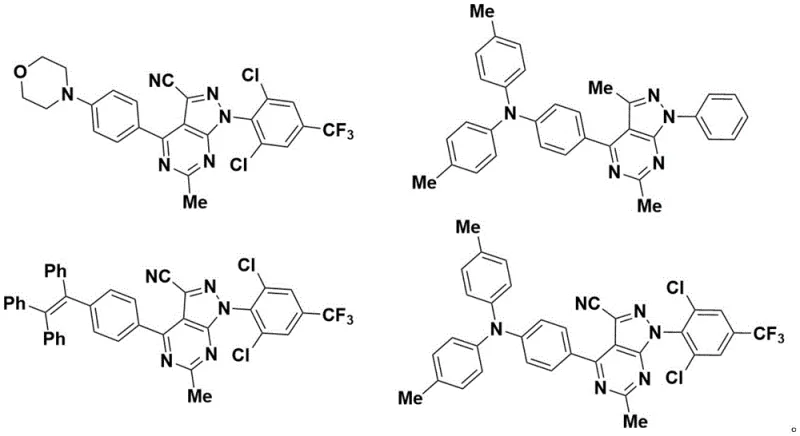
A: These derivatives exhibit significant photoluminescence properties, making them suitable for organic light-emitting devices, fluorescent probes, and potentially as bioactive pharmaceutical agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to translate this innovative patent data into commercial reality for our global clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of pyrazolopyrimidine derivatives meets the highest industry standards for both pharmaceutical and electronic applications. Our commitment to quality and process excellence makes us the ideal partner for companies looking to secure a stable source of high-performance chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume and purity needs. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Contact us today to explore how we can collaborate to bring these advanced materials to your market efficiently and reliably.
