Revolutionizing Triazole Chemistry: Commercial-Scale Production of High-Purity 5-Trifluoromethyl Compounds Without Metal Catalysts
Chinese patent CN113121462B discloses a groundbreaking metal-free synthesis methodology for 5-trifluoromethyl-substituted 1,2,3-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative approach eliminates the need for hazardous azide compounds and transition metal catalysts that have traditionally plagued triazole synthesis, thereby addressing critical safety and purity concerns in API intermediate manufacturing. The patented process utilizes readily available starting materials—trifluoroethyl imidoyl chloride and diazo compounds—under mild reaction conditions with cesium carbonate as a base promoter in acetonitrile solvent at 60°C. This methodology not only enhances operational safety but also demonstrates exceptional scalability from laboratory to industrial production scales, making it particularly valuable for global pharmaceutical manufacturers seeking reliable and high-purity triazole intermediates. The strategic elimination of toxic reagents and metal catalysts directly translates to reduced purification complexity and improved environmental compliance, positioning this technology as a superior alternative to conventional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
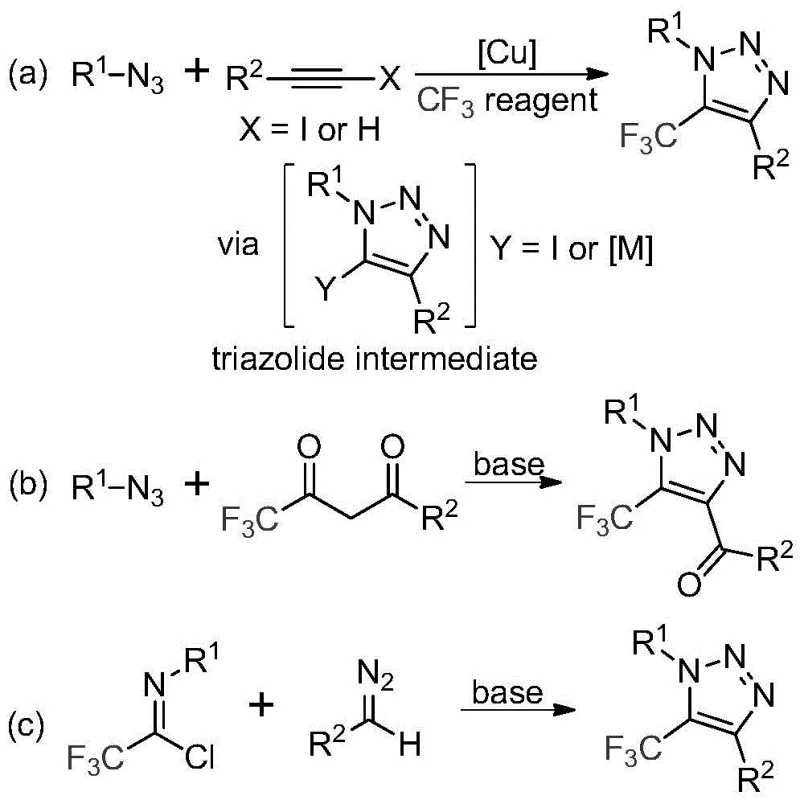
Traditional synthetic approaches for trifluoromethyl-substituted triazoles predominantly rely on two methodologies that present significant operational challenges for pharmaceutical manufacturers. The first method employs copper-catalyzed [3+2] cycloaddition between alkynes and organic azides to form triazole metal complexes, followed by reaction with various trifluoromethyl reagents; this process necessitates handling highly toxic and potentially explosive azide compounds that pose serious safety risks in manufacturing environments. The second conventional approach utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly requires hazardous azide precursors and often suffers from inconsistent yields and difficult purification processes due to metal contamination. Both established methods generate complex mixtures requiring extensive chromatographic separation, resulting in higher production costs, longer lead times, and compromised batch-to-batch consistency that are unacceptable in regulated pharmaceutical manufacturing. Furthermore, the presence of transition metals in final products necessitates additional purification steps to meet stringent regulatory requirements for API intermediates, significantly increasing the cost of goods and complicating scale-up procedures for commercial production.
The Novel Approach
The patented methodology presented in CN113121462B introduces a fundamentally different synthetic pathway that completely eliminates the need for both azide compounds and transition metal catalysts while maintaining excellent reaction efficiency and product purity. This innovative process utilizes trifluoroethyl imidoyl chloride and diazo compounds as starting materials, with cesium carbonate serving as an effective base promoter in acetonitrile solvent at moderate temperatures of 60°C for 12 hours. The reaction mechanism proceeds through a base-promoted intermolecular nucleophilic addition and elimination process followed by intramolecular 5-endo-dig cyclization, avoiding the formation of hazardous intermediates altogether. This approach delivers multiple advantages including simplified reaction workup (requiring only filtration and column chromatography), exceptional functional group tolerance across diverse substrates, and the ability to produce high-purity triazole compounds without metal contamination concerns. The elimination of hazardous reagents not only enhances workplace safety but also significantly reduces regulatory compliance burdens and associated costs for pharmaceutical manufacturers seeking to produce these valuable heterocyclic intermediates at commercial scale.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism described in patent CN113121462B represents a significant departure from conventional triazole synthesis methodologies through its elegant base-promoted pathway that avoids hazardous intermediates. The process begins with the nucleophilic attack of the diazo compound on the electrophilic carbon of trifluoroethyl imidoyl chloride, facilitated by cesium carbonate as a mild base that promotes deprotonation without generating unstable intermediates. This initial step forms a key intermediate that undergoes spontaneous intramolecular cyclization through a 5-endo-dig process, where the nitrogen atom attacks the electron-deficient carbon center to form the characteristic triazole ring structure with perfect regioselectivity at the 5-position for the trifluoromethyl group. The mild reaction conditions (60°C in acetonitrile) prevent decomposition of sensitive functional groups while ensuring complete conversion of starting materials, as evidenced by the consistent yields across diverse substrate combinations documented in the patent examples. This mechanism operates without transition metals or explosive azide compounds, eliminating potential sources of metal contamination that would otherwise require extensive purification to meet pharmaceutical quality standards.
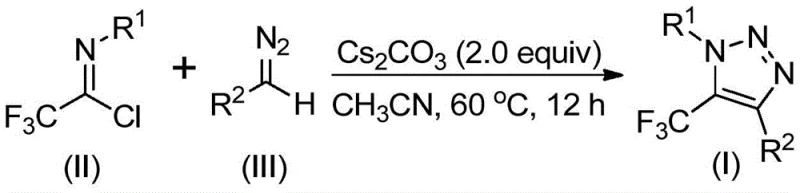
Regarding impurity control, the patented methodology demonstrates exceptional selectivity that minimizes unwanted byproduct formation through several inherent design features. The precise stoichiometric ratio (trifluoroethyl imidoyl chloride:diazo compound:cesium carbonate = 1:1.5:2) ensures complete consumption of starting materials while preventing side reactions from excess reagents. The use of molecular sieves (4Å) effectively controls moisture that could otherwise lead to hydrolysis byproducts, while the choice of acetonitrile as solvent provides optimal polarity for intermediate stabilization without promoting decomposition pathways. The moderate reaction temperature (60°C) avoids thermal degradation that could generate impurities common in higher-temperature processes, and the well-defined reaction time (12 hours) prevents over-reaction that might lead to secondary products. Most significantly, the complete absence of transition metals eliminates metal-catalyzed side reactions and removes the need for costly metal removal steps that often introduce additional impurities in conventional triazole syntheses, resulting in products with inherently higher purity profiles suitable for pharmaceutical applications without extensive post-processing.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This patented methodology represents a significant advancement in triazole synthesis that addresses critical challenges faced by pharmaceutical manufacturers in producing high-purity heterocyclic intermediates. The process eliminates hazardous reagents while maintaining excellent yield and selectivity across diverse substrate combinations, making it particularly valuable for commercial-scale production of API intermediates. Detailed standardized synthesis procedures have been developed based on the patent specifications to ensure consistent product quality and operational efficiency; the following step-by-step guide provides essential information for successful implementation in manufacturing environments.
- Combine trifluoroethyl imidoyl chloride (II), diazo compound (III), cesium carbonate (2.0 equiv), and molecular sieves in acetonitrile solvent under nitrogen atmosphere
- Heat the reaction mixture to 60°C with stirring for 12 hours while monitoring reaction progress by analytical methods
- Perform workup by filtration through silica gel followed by column chromatography purification to obtain high-purity triazole product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value to procurement and supply chain decision-makers by addressing multiple pain points inherent in traditional triazole manufacturing processes. The elimination of hazardous materials reduces regulatory compliance burdens while improving operational safety profiles, directly impacting both cost structures and supply chain reliability for global pharmaceutical manufacturers seeking consistent access to high-quality intermediates.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes significant expenses associated with catalyst procurement, recovery systems, and extensive purification processes required to meet regulatory limits for metal residues in pharmaceutical intermediates. By avoiding toxic azide compounds, manufacturers eliminate specialized handling equipment and safety protocols that substantially increase operational costs while reducing production cycle times through simplified workup procedures that require only filtration and standard column chromatography rather than complex multi-step purification sequences.
- Enhanced Supply Chain Reliability: The use of readily available starting materials with established commercial supply chains ensures consistent access to raw materials without dependency on specialized or restricted reagents that often cause supply disruptions. The robust nature of the reaction across diverse substrates provides flexibility to adapt to raw material availability fluctuations while maintaining product quality, and the simplified process design reduces vulnerability to equipment failures or operator errors that commonly cause batch failures in more complex synthetic routes.
- Scalability and Environmental Compliance: The mild reaction conditions (60°C) and straightforward workup procedures enable seamless scale-up from laboratory to commercial production without requiring specialized equipment or infrastructure modifications typically needed for high-pressure or cryogenic processes. The elimination of hazardous reagents significantly reduces environmental impact by minimizing waste streams requiring special treatment and eliminating potential safety incidents that could disrupt production schedules, while the absence of metal catalysts simplifies waste disposal procedures and reduces regulatory reporting requirements associated with heavy metal handling.
Frequently Asked Questions (FAQ)
The following questions address common concerns from technical procurement teams regarding implementation of this patented technology for commercial production of high-purity triazole intermediates. Each response is based on detailed analysis of the patent specifications and experimental data provided in CN113121462B.
Q: How does this method eliminate metal contamination concerns compared to conventional approaches?
A: The patented process completely avoids transition metal catalysts by utilizing a base-promoted mechanism with cesium carbonate instead of copper catalysts used in traditional methods. This eliminates the need for extensive metal removal steps and ensures final products meet strict regulatory requirements for metal residues without additional purification.
Q: What are the key advantages regarding supply chain reliability for this synthesis?
A: The methodology uses readily available starting materials with established commercial supply chains while eliminating hazardous azide compounds that often cause regulatory delays and supply disruptions. The robust reaction tolerates variations in raw material quality while maintaining consistent product specifications.
Q: How does this process support scalability from laboratory to commercial production?
A: The mild reaction conditions (60°C), straightforward workup procedures requiring only filtration and standard chromatography, and absence of specialized equipment requirements enable seamless scale-up from gram-scale laboratory reactions to multi-ton commercial production without process revalidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds, with stringent purity specifications consistently achieved through our rigorous QC labs and advanced analytical capabilities. Our specialized expertise in triazole chemistry enables us to deliver high-purity 5-trifluoromethyl-substituted intermediates that meet exacting pharmaceutical standards while optimizing cost structures through efficient process design and scale-up strategies developed from years of CDMO experience across multiple therapeutic areas.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can enhance your supply chain resilience while reducing total cost of ownership; please contact us for specific COA data and route feasibility assessments tailored to your manufacturing requirements.
