Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
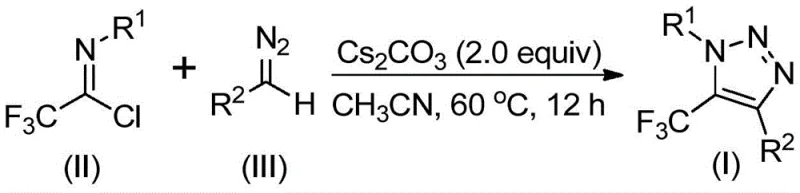
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding safety profiles and synthetic efficiency. A groundbreaking development detailed in Chinese Patent CN113121462B introduces a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that fundamentally shifts the paradigm away from hazardous reagents. This technology leverages a base-promoted cyclization strategy using readily available trifluoroethylimidoyl chloride and diazo compounds, effectively bypassing the need for toxic organic azides or expensive transition metal catalysts. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline the supply chain for critical pharmaceutical intermediates while enhancing operational safety. The method operates under mild thermal conditions, typically around 60°C, utilizing cesium carbonate as a promoter in acetonitrile, ensuring high atom economy and simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,3-triazole scaffold, a privileged structure in medicinal chemistry, has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions. While effective, these traditional pathways suffer from inherent safety liabilities due to the use of organic azides, which are known to be toxic, mutagenic, and potentially explosive upon isolation or heating. Furthermore, alternative organocatalytic approaches involving trifluoromethyl ketones still necessitate the handling of hazardous azide precursors. From a manufacturing perspective, the reliance on copper catalysts introduces another layer of complexity; residual heavy metals must be rigorously removed to meet stringent regulatory limits for active pharmaceutical ingredients (APIs), often requiring additional chelating resins or complex purification protocols that drive up production costs and extend lead times. These factors collectively create a bottleneck for the reliable supply of high-purity triazole intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121462B offers a transformative solution by employing a metal-free, azide-free protocol. By utilizing trifluoroethylimidoyl chloride and diazo compounds as starting materials, the process eliminates the explosion risks associated with azides entirely. The reaction proceeds through a base-promoted mechanism that is both robust and tolerant of various functional groups, allowing for the synthesis of diverse derivatives including esters, ketones, and phosphonates. This approach not only enhances the safety profile of the manufacturing facility but also simplifies the regulatory burden regarding heavy metal residues. The use of cesium carbonate, a mild and commercially abundant base, ensures that the reaction conditions remain gentle, preserving sensitive functional groups that might otherwise degrade under harsher catalytic conditions typical of older methodologies.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is a fascinating example of efficient bond construction without transition metal mediation. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion, generating a reactive intermediate. Subsequently, the system undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is crucial as it closes the five-membered triazole ring, establishing the aromatic heterocyclic core. The presence of the trifluoromethyl group at the 5-position is retained throughout this process, imparting the desired lipophilicity and metabolic stability to the final molecule. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrates.
Furthermore, the choice of cesium carbonate as the promoter plays a pivotal role in the reaction kinetics and impurity profile. Cesium ions, being large and soft, can effectively coordinate with the nitrogen atoms of the diazo and triazole intermediates, stabilizing the transition states and lowering the activation energy for the cyclization step. This specific interaction helps to suppress side reactions such as dimerization of the diazo compound or hydrolysis of the imidoyl chloride, thereby ensuring a cleaner crude reaction mixture. The result is a product with a superior impurity profile, reducing the load on purification columns and increasing the overall yield of the isolated API intermediate. This level of control over the reaction pathway is essential for maintaining batch-to-batch consistency in a commercial setting.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction environment is required. The patent suggests a molar ratio where the diazo compound is used in slight excess relative to the trifluoroethylimidoyl chloride, typically around 1.5 equivalents, to drive the reaction to completion given the reactive nature of the diazo species. Cesium carbonate is employed at approximately 2.0 equivalents to ensure sufficient basicity for the promotion of the cyclization. The reaction is best conducted in anhydrous acetonitrile, which provides optimal solubility for all reagents and supports the ionic mechanism. Following the reaction period, standard workup procedures involving filtration to remove inorganic salts and silica gel chromatography yield the pure product.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in anhydrous acetonitrile under inert atmosphere.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The elimination of explosive azides significantly lowers the insurance premiums and safety infrastructure costs associated with manufacturing facilities, directly impacting the bottom line. Moreover, the removal of transition metal catalysts like copper simplifies the purification train, reducing the consumption of expensive scavenging resins and shortening the overall production cycle time. This streamlined process enhances the reliability of supply, as the risk of batch failures due to metal contamination or azide instability is drastically minimized. Consequently, this method supports a more resilient supply chain capable of meeting the fluctuating demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic advantages of this route are multifaceted, primarily driven by the substitution of expensive and hazardous reagents with commodity chemicals. By avoiding the use of specialized trifluoromethylating reagents and copper catalysts, the raw material costs are significantly lowered. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a greener and more cost-effective manufacturing process. The high atom economy of the reaction ensures that a greater proportion of the starting materials ends up in the final product, further optimizing resource utilization and minimizing waste generation.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as trifluoroethylimidoyl chloride and diazo compounds ensures a consistent supply of feedstock. Unlike azides, which often require on-site generation due to storage instability, these precursors can be sourced from multiple vendors and stored safely for extended periods. This stability allows for better inventory management and reduces the risk of production stoppages caused by reagent degradation or supply shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in temperature or mixing, leading to higher reproducibility across different manufacturing sites.
- Scalability and Environmental Compliance: Scaling this reaction from the laboratory to industrial production is straightforward due to the absence of exothermic hazards associated with azide chemistry. The mild reaction temperature of 60°C is easily maintainable in large-scale reactors without requiring complex cooling systems. Furthermore, the metal-free nature of the synthesis aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge. This compliance reduces the regulatory burden and facilitates faster approval processes for new drug applications, making it an attractive option for companies aiming to bring products to market rapidly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific application needs.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety hazards associated with handling high-energy azide intermediates.
Q: What represents the primary cost driver in this manufacturing process?
A: The process utilizes cesium carbonate as a promoter, which is relatively inexpensive compared to transition metal catalysts like copper or rhodium. Additionally, the absence of heavy metals removes the need for costly purification steps to meet residual metal specifications.
Q: Can this reaction be scaled for industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with mild reaction conditions (60°C) and simple workup procedures involving filtration and column chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global fine chemicals market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the advanced methodology described in CN113121462B, we can offer our partners a superior product with a reduced environmental footprint and enhanced safety profile.
We invite you to collaborate with us to explore how this technology can optimize your specific supply chain requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your goals for cost reduction and supply security in the manufacturing of complex pharmaceutical intermediates.
