Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The rapid evolution of medicinal chemistry demands robust, safe, and scalable synthetic routes for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous azide chemistry towards a safer, base-promoted cyclization strategy. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this patent offers a compelling solution for generating high-purity building blocks essential for modern drug discovery programs. The methodology eliminates the need for transition metal catalysts and explosive reagents, thereby streamlining the supply chain and reducing regulatory burdens associated with hazardous material handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation steps. Alternatively, organocatalytic 1,3-dipolar cycloadditions involving azides and trifluoromethyl ketones have been employed. These conventional pathways suffer from critical drawbacks that hinder their utility in large-scale manufacturing. The primary concern is the reliance on organic azides, which are notoriously toxic and possess high explosion risks, necessitating specialized equipment and stringent safety protocols that drive up operational costs. Furthermore, the use of copper catalysts introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), requiring expensive and time-consuming purification steps to meet strict residual metal limits set by regulatory bodies.
The Novel Approach
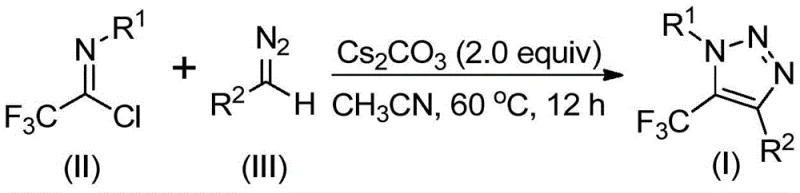
In stark contrast, the technology described in patent CN113121462B utilizes cheap and readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials. This metal-free, azide-free approach fundamentally alters the risk profile of the synthesis. By employing a simple base promotion system, specifically cesium carbonate, the reaction proceeds efficiently without the need for toxic heavy metals or dangerous azide reagents. This not only enhances the safety of the operation but also simplifies the downstream processing, as there is no need for complex metal scavenging procedures. The result is a cleaner reaction profile that facilitates cost reduction in pharmaceutical intermediate manufacturing while maintaining high yields and excellent functional group tolerance.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is both elegant and efficient, proceeding through a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step promotes the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization reaction. This cyclization step is pivotal as it constructs the five-membered triazole ring system characteristic of the target molecule. The presence of the trifluoromethyl group at the 5-position is established directly from the imidoyl chloride precursor, ensuring precise regiocontrol without the need for additional trifluoromethylation reagents which can often be costly or difficult to handle.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of transition metals eliminates a major class of potential impurities related to catalyst ligands or metal residues. Furthermore, the reaction conditions are mild enough to preserve sensitive functional groups on the aromatic rings of the substrates, such as halogens, alkoxy groups, or esters. This high chemoselectivity ensures that the impurity profile of the crude product is relatively simple, primarily consisting of unreacted starting materials or minor hydrolysis byproducts, which are easily removed during standard workup procedures like filtration and silica gel chromatography. This predictability is crucial for R&D teams aiming to establish robust quality control specifications for clinical trial materials.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory scale-up and commercial production. The process involves mixing the key reactants—trifluoroethylimidoyl chloride and the specific diazo compound—with cesium carbonate in an aprotic organic solvent. Acetonitrile is identified as the preferred solvent due to its ability to dissolve the reactants effectively and promote high conversion rates. The reaction is typically heated to a moderate temperature range of 50°C to 70°C, with 60°C being the optimal point for many substrates, and maintained for 8 to 16 hours. Upon completion, the mixture is filtered to remove inorganic salts, and the product is isolated via standard purification techniques. The detailed standardized synthesis steps are outlined in the guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical yield. The elimination of hazardous azides and transition metals translates directly into reduced logistical complexity and lower insurance premiums for storage and transport. Since the starting materials are commercially available and stable, the risk of supply disruption due to the regulation of controlled precursors is significantly minimized. This stability ensures a continuous flow of materials, which is vital for maintaining production schedules in a fast-paced pharmaceutical environment.
- Cost Reduction in Manufacturing: The removal of expensive copper catalysts and the associated purification resins required for metal scavenging leads to substantial cost savings. Additionally, the avoidance of specialized safety infrastructure needed for handling explosive azides reduces capital expenditure and operational overhead. The use of inexpensive bases like cesium carbonate further drives down the raw material costs, making the overall process economically superior to traditional methods.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted aromatic amines and diazo precursors, are widely available from global chemical suppliers. This broad supplier base mitigates the risk of single-source dependency. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch reproducibility which is critical for long-term supply contracts.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be easily extended to the gram level and beyond, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates. The generation of waste is minimized as the byproducts are largely inorganic salts that are easy to dispose of, aligning with green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations regarding heavy metal discharge and hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, significantly reducing safety hazards and handling risks in the laboratory and plant.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds under mild thermal conditions between 50°C and 70°C, often optimized at 60°C, using cesium carbonate as a promoter in acetonitrile solvent for 8 to 16 hours.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent explicitly states that the method is easily scalable from gram-level laboratory synthesis to industrial production, offering high reaction efficiency and simple post-treatment procedures suitable for large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced metal-free technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while optimizing your overall budget.
