Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Manufacturing
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Manufacturing
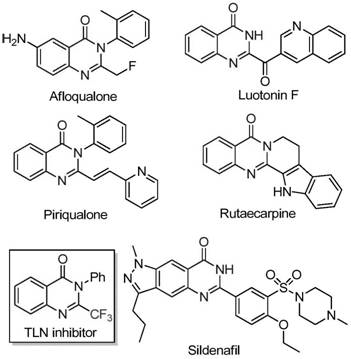
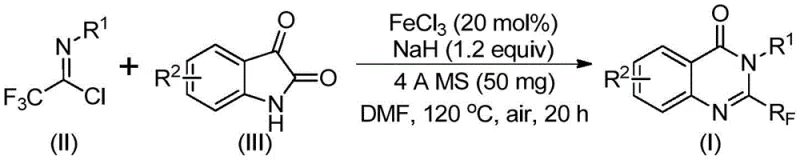
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages an iron-catalyzed cyclization strategy that circumvents the limitations of traditional precious metal catalysis, offering a streamlined pathway for generating complex molecular scaffolds found in numerous bioactive agents. The process utilizes readily available starting materials such as trifluoroethylimidoyl chloride and isatin derivatives, reacting them in the presence of ferric chloride and sodium hydride to achieve high conversion rates. By addressing key pain points in organic synthesis such as catalyst cost and reaction severity, this patent provides a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives containing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons like trifluoroacetic anhydride or ethyl trifluoroacetate with substrates such as anthranilamide or isatoic anhydride. These conventional pathways are frequently plagued by severe reaction conditions that necessitate stringent control over temperature and pressure, often leading to safety concerns in large-scale operations. Furthermore, the substrates required for these traditional methods can be prohibitively expensive or difficult to source in bulk quantities, creating bottlenecks in the procurement process for contract development and manufacturing organizations. The narrow substrate scope associated with these older techniques also limits the structural diversity achievable, restricting the ability of R&D teams to rapidly iterate on lead compounds. Additionally, the reliance on specific activating agents often generates significant chemical waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a highly efficient route utilizing trifluoroethylimidoyl chloride and isatin as the core building blocks, catalyzed by inexpensive ferric chloride. This novel approach dramatically simplifies the synthetic sequence by enabling a direct cyclization that tolerates a wide array of functional groups on both the aromatic amine and the isatin ring systems. The reaction proceeds under relatively mild thermal conditions, initially at 40°C and subsequently heating to 120°C, which reduces energy consumption and equipment stress compared to harsher alternatives. The use of iron as a catalyst not only lowers the raw material costs significantly but also eliminates the need for complex removal of toxic heavy metal residues, a critical factor for regulatory compliance in API production. This method represents a paradigm shift towards sustainable and economically viable manufacturing of complex heterocyclic intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway for this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and transition metal-catalyzed rearrangement. Initially, the sodium hydride acts as a strong base to deprotonate the isatin nitrogen, facilitating a nucleophilic attack on the imidoyl chloride to form a carbon-nitrogen bond. This intermediate trifluoroacetamidine species then undergoes an iron-catalyzed decarbonylation process, which is the rate-determining step that drives the formation of the quinazolinone core. The ferric chloride likely coordinates with the carbonyl oxygen or the nitrogen atoms to lower the activation energy for the elimination of carbon monoxide, thereby promoting the cyclization. Understanding this mechanism is vital for process chemists as it highlights the dual role of the reagents: NaH for activation and FeCl3 for the skeletal rearrangement, ensuring that stoichiometry is carefully managed to prevent side reactions.
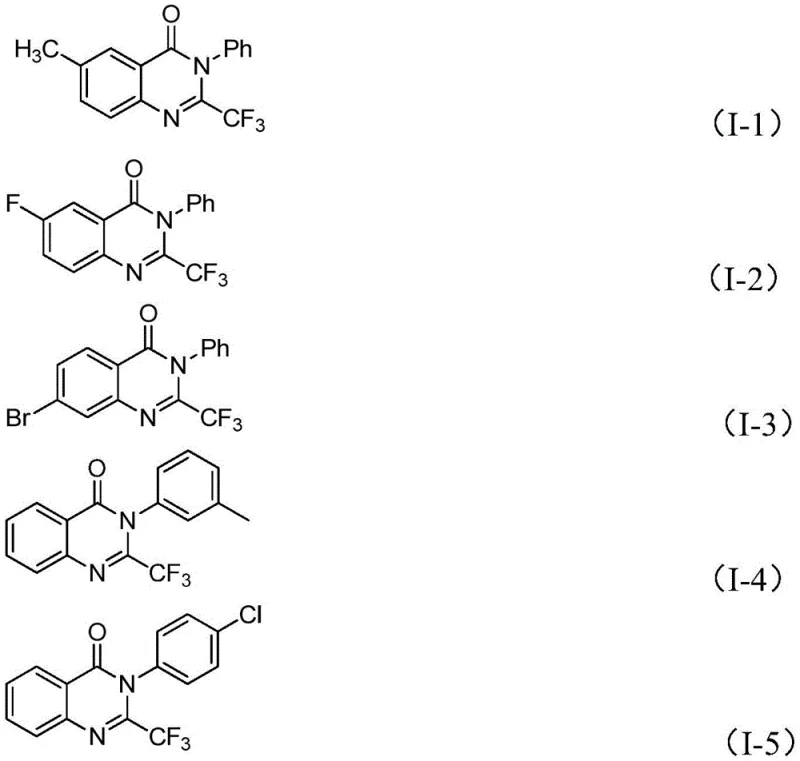
From an impurity control perspective, the specificity of this iron-catalyzed cycle offers distinct advantages over non-catalytic thermal cyclizations. The presence of the metal center directs the reaction trajectory towards the desired quinazolinone product, minimizing the formation of regioisomers or polymeric byproducts that often contaminate crude reaction mixtures. The tolerance for various substituents, such as halogens (fluorine, chlorine, bromine) and alkyl groups on the aromatic rings, suggests that the electronic properties of the substrate do not drastically inhibit the catalytic cycle. This robustness allows for the synthesis of diverse analogues, such as compounds I-1 through I-5, with consistent purity profiles. For quality assurance teams, this implies a more predictable impurity profile, simplifying the validation of analytical methods and ensuring that the final API intermediate meets stringent pharmacopeial standards without extensive recrystallization steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol dictates a two-stage heating profile where the reaction mixture is first maintained at a lower temperature to allow for the initial coupling, followed by a higher temperature phase to drive the cyclization to completion. The use of 4A molecular sieves is critical to sequester moisture, which could otherwise deactivate the sodium hydride or hydrolyze the sensitive imidoyl chloride starting material. Operators must ensure that the ferric chloride is added in the correct molar ratio, typically around 20 mol%, to balance catalytic activity with cost efficiency. Detailed standard operating procedures regarding the addition rates of reagents and the quenching of excess sodium hydride are essential for safe scale-up.
- Charge a reaction vessel with ferric chloride (20 mol%), sodium hydride (1.2 equiv), and 4A molecular sieves in anhydrous DMF solvent under air atmosphere.
- Add trifluoroethylimidoyl chloride and the corresponding isatin derivative to the mixture and stir at 40°C for approximately 10 hours to initiate C-N bond formation.
- Heat the reaction mixture to 120°C and maintain for 20 hours to complete the decarbonylation and cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that extend beyond simple chemistry. The shift from precious metal catalysts to abundant iron salts fundamentally alters the cost structure of the manufacturing process, removing exposure to the volatile pricing of rhodium, palladium, or ruthenium. Furthermore, the simplicity of the workup procedure, which involves basic filtration and standard chromatography, reduces the demand for specialized processing equipment and shortens the overall production cycle time. This efficiency gain allows suppliers to respond more agilely to market demands, reducing lead times for critical intermediates needed in drug development pipelines. The robustness of the reaction also implies higher batch-to-batch consistency, mitigating the risk of supply disruptions caused by failed runs or off-spec material.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with ferric chloride results in a drastic reduction in direct material costs, which is a primary driver for margin improvement in competitive generic markets. Additionally, the use of commodity chemicals like DMF and sodium hydride, which are sourced from established global supply chains, ensures price stability and availability. The elimination of complex ligand systems further simplifies the bill of materials, reducing the administrative burden of sourcing and qualifying multiple specialized reagents. Consequently, the overall cost of goods sold (COGS) for the final quinazolinone intermediate is significantly optimized, providing a competitive edge in tender negotiations.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are commercially available from multiple vendors, preventing single-source dependency risks. The reaction's tolerance to air and its operation in common solvents mean that storage and handling requirements are less stringent than for air-sensitive organometallic reagents. This resilience ensures that production schedules can be maintained even under fluctuating logistical conditions, securing the continuity of supply for downstream API manufacturers. Moreover, the scalability demonstrated in the patent suggests that transitioning from kilogram to ton-scale production can be achieved with minimal process re-engineering.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional methods that might utilize toxic tin or lead reagents, aligning with modern green chemistry principles and regulatory expectations. The simplified purification workflow reduces solvent consumption and waste generation, lowering the costs associated with waste disposal and environmental compliance reporting. As regulatory bodies increasingly scrutinize the environmental footprint of pharmaceutical manufacturing, adopting this cleaner iron-catalyzed route future-proofs the supply chain against tightening emissions and effluent standards. This sustainability aspect is increasingly becoming a key differentiator when selecting long-term partners for API production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the balance between theoretical potential and practical execution observed in the reported examples.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational benefits compared to precious metal catalysts. It is inexpensive, readily available, and demonstrates high tolerance for various functional groups, allowing for a broader substrate scope without the need for rigorous exclusion of air or moisture in some steps.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states that the method is suitable for gram-level expansion and provides possibilities for industrial scale application. The use of common solvents like DMF and simple post-treatment procedures like filtration and column chromatography supports scalability.
Q: What is the typical yield range for this trifluoromethylation cyclization reaction?
A: According to the experimental data in the patent, the reaction yields vary depending on the substituents but generally range from moderate to excellent. For instance, electron-deficient or neutral substrates often achieve yields above 80%, with specific examples reaching up to 93% yield under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest quality standards required for clinical and commercial applications. Our infrastructure is designed to handle complex chemistries safely, leveraging our expertise in iron-catalyzed transformations to deliver cost-effective solutions.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this iron-catalyzed process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your drug development journey.
