Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Quinazolinone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Quinazolinone Production
The pharmaceutical landscape is continuously evolving, driven by the demand for heterocyclic scaffolds that offer superior metabolic stability and bioavailability. Among these, quinazolinone derivatives stand out as privileged structures found in numerous bioactive molecules, ranging from anticonvulsants like CP-465022 to antitumor agents such as Erastin. 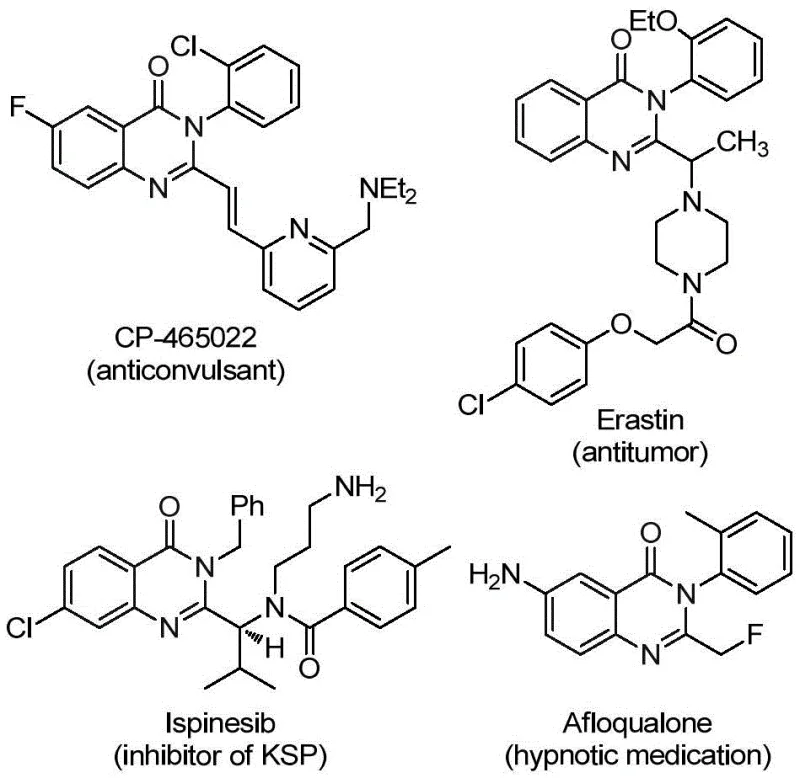 The introduction of a trifluoromethyl group at the 2-position of the quinazolinone ring significantly enhances these pharmacological properties by modulating electronegativity and lipophilicity. A groundbreaking preparation method disclosed in patent CN112125856A offers a robust solution for synthesizing these valuable intermediates. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing cheap and readily available starting materials to achieve high efficiency and broad substrate applicability.
The introduction of a trifluoromethyl group at the 2-position of the quinazolinone ring significantly enhances these pharmacological properties by modulating electronegativity and lipophilicity. A groundbreaking preparation method disclosed in patent CN112125856A offers a robust solution for synthesizing these valuable intermediates. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing cheap and readily available starting materials to achieve high efficiency and broad substrate applicability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone derivatives has been fraught with significant technical and operational challenges that hinder large-scale production. Conventional literature reports typically rely on the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, often requiring harsh reaction conditions that compromise safety and energy efficiency. Alternative routes involving the cyclization of anthranilic esters with unstable trifluoroacetamides or the use of isatoic anhydride with trifluoroacetic anhydride frequently suffer from narrow substrate scopes and the necessity for pre-activated, expensive reagents. Furthermore, methods promoted by coupling reagents like T3P often result in lower yields and generate substantial waste streams, complicating downstream purification and environmental compliance. These limitations collectively increase the cost of goods sold (COGS) and create bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology described in CN112125856A introduces a paradigm shift by employing a palladium-catalyzed carbonylation strategy that circumvents the drawbacks of traditional cyclization. This innovative route utilizes o-iodoaniline and trifluoroacetimidoyl chloride as primary building blocks, reacting them in the presence of a solid carbon monoxide surrogate known as TFBen (phenyl 1,3,5-tricarboxylate). By replacing toxic, gaseous carbon monoxide with a stable solid surrogate, the process dramatically improves operational safety and eliminates the need for specialized high-pressure gas handling infrastructure. The reaction proceeds smoothly in organic solvents like tetrahydrofuran (THF) at a moderate temperature of 90°C, demonstrating exceptional compatibility with diverse functional groups including halogens, alkyls, and nitro groups. This versatility allows for the rapid generation of compound libraries for drug discovery while maintaining a streamlined path to commercial manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The core of this synthetic breakthrough lies in a sophisticated catalytic cycle that orchestrates multiple bond-forming events in a single pot. The reaction initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, facilitated by potassium tert-butoxide, to generate a trifluoroacetamidine intermediate in situ. Subsequently, the palladium catalyst, specifically bis(triphenylphosphine)palladium(II) dichloride coordinated with the bidentate ligand 1,3-bis(diphenylphosphino)propane (dppp), undergoes oxidative addition into the carbon-iodine bond of the aromatic ring. 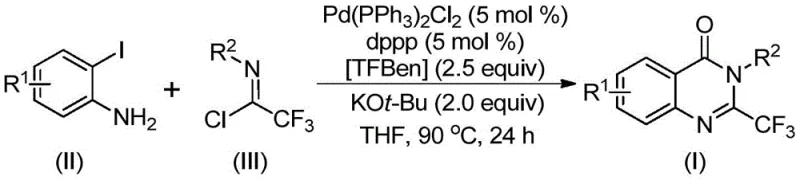 Under heating conditions, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a crucial acyl-palladium intermediate. The presence of the base further promotes the formation of a palladium-nitrogen species, leading to a seven-membered ring palladium intermediate before final reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active catalyst.
Under heating conditions, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a crucial acyl-palladium intermediate. The presence of the base further promotes the formation of a palladium-nitrogen species, leading to a seven-membered ring palladium intermediate before final reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active catalyst.
From an impurity control perspective, the choice of ligand and base plays a pivotal role in ensuring high purity profiles essential for pharmaceutical applications. The use of dppp as a ligand stabilizes the palladium center, minimizing the formation of homocoupling byproducts that often plague cross-coupling reactions. Additionally, the controlled release of carbon monoxide from the solid surrogate prevents local concentration spikes that could lead to side reactions or incomplete conversions. The reaction conditions are optimized to run for 16 to 30 hours, ensuring that the equilibrium favors the formation of the fused heterocyclic ring system over potential open-chain intermediates. This mechanistic precision results in crude products that are easier to purify via standard column chromatography, reducing the overall solvent consumption and processing time required to meet stringent quality specifications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The patent outlines a standardized protocol where the molar ratio of o-iodoaniline to trifluoroacetimidoyl chloride is maintained at approximately 1:2, with the palladium catalyst and ligand loaded at 5 mol % each. The use of THF as the solvent is preferred due to its ability to fully dissolve all reactants and facilitate the homogeneous catalytic cycle. While the general procedure is robust, slight adjustments in reaction time may be necessary depending on the electronic nature of the substituents on the aromatic rings, with electron-withdrawing groups sometimes requiring the full 30-hour duration to reach completion. For detailed step-by-step instructions on reagent addition, workup procedures, and purification techniques, please refer to the standardized guide below.
- Charge a reaction vessel with Pd(PPh3)2Cl2 catalyst, dppp ligand, KOt-Bu base, TFBen solid CO source, trifluoroacetimidoyl chloride, and o-iodoaniline in THF solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages regarding cost structure and logistical reliability. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas chemistry. This flexibility expands the pool of potential contract manufacturing organizations (CMOs) capable of executing the synthesis, thereby enhancing supply chain resilience and reducing dependency on single-source vendors with specialized infrastructure. Furthermore, the use of commercially available starting materials like o-iodoaniline derivatives ensures a steady supply of raw inputs without the volatility associated with custom-synthesized precursors.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and dangerous carbon monoxide gas handling systems, which significantly lowers capital expenditure (CAPEX) and operational expenditure (OPEX). By utilizing cheap and readily available trifluoroacetimidoyl chlorides and avoiding the need for cryogenic conditions or high-pressure autoclaves, the overall production cost per kilogram is substantially reduced. The high atom economy of the tandem reaction minimizes waste generation, leading to lower costs associated with waste disposal and environmental compliance. Additionally, the high yields observed for many substrates, reaching up to 99% in optimized cases, mean less raw material is wasted on failed batches or extensive recycling loops.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as TFBen and solid palladium catalysts mitigates the risk of supply disruptions caused by the transportation hazards of compressed gases. Since the starting materials are widely sourced from the global chemical market, procurement teams can easily qualify multiple suppliers to prevent bottlenecks. The robustness of the reaction conditions, tolerating a wide range of temperatures and times within the specified window, ensures consistent batch-to-batch quality even when scaling up from laboratory to pilot plant volumes. This consistency is critical for maintaining uninterrupted supply lines to downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the use of common organic solvents like THF, which are well-understood in industrial recovery and recycling systems. The absence of toxic gas emissions simplifies the permitting process for new manufacturing lines and reduces the burden on scrubber systems and ventilation infrastructure. The simplified post-treatment process, involving filtration and standard chromatography, aligns well with existing purification trains in multipurpose plants, avoiding the need for dedicated unit operations. This ease of scale-up ensures that the technology can rapidly transition from R&D curiosity to a commercial reality, supporting the fast-paced timelines of modern drug development programs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these nuances is essential for R&D teams evaluating route selection and for procurement professionals assessing vendor qualifications. We encourage stakeholders to review these points carefully to align their expectations with the proven performance of this catalytic system.
Q: What is the primary advantage of using TFBen over gaseous CO in this synthesis?
A: TFBen (phenyl 1,3,5-tricarboxylate) acts as a solid carbon monoxide surrogate, eliminating the severe safety hazards and specialized high-pressure equipment required for handling toxic carbon monoxide gas, thereby simplifying operational protocols.
Q: What is the substrate scope for the R1 and R2 positions in this reaction?
A: The method demonstrates excellent compatibility with various substituents; R1 can be H, alkyl, halogen, or trifluoromethyl, while R2 accommodates substituted or unsubstituted aryl groups including phenyl, naphthyl, and nitro-phenyl derivatives.
Q: How does this method improve yield compared to traditional cyclization routes?
A: Unlike traditional methods requiring harsh conditions or unstable intermediates which often result in low yields, this Pd-catalyzed approach achieves yields up to 99% for optimized substrates under relatively mild thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipeline. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop to marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards. We are committed to leveraging advanced catalytic technologies like the one described in CN112125856A to drive innovation and efficiency for our global partners.
We invite you to engage with our technical team to explore how this efficient synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this palladium-catalyzed method for your supply chain. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique molecular targets. Let us collaborate to build a more resilient and cost-effective supply chain for the next generation of therapeutic agents.
