Advanced Iodine-Catalyzed Process for Scalable Production of High-Purity Triazole Intermediates in Pharmaceutical Manufacturing
The Chinese patent CN113105402B represents a significant advancement in heterocyclic chemistry through its innovative approach to synthesizing biologically active triazole derivatives without requiring transition metal catalysts or stringent anhydrous conditions This methodology addresses critical limitations in traditional synthetic routes by utilizing molecular iodine as an efficient catalyst within dimethyl sulfoxide solvent systems at moderate temperatures The process demonstrates exceptional versatility across diverse substrate combinations while maintaining operational simplicity that facilitates seamless technology transfer from laboratory development to manufacturing environments By eliminating toxic heavy metal residues commonly associated with conventional methodologies this breakthrough ensures cleaner reaction profiles that directly translate to reduced purification costs and enhanced product quality for pharmaceutical applications Furthermore the demonstrated scalability from milligram quantities to potential commercial production provides a robust foundation meeting rigorous quality standards demanded by global regulatory bodies This patent therefore establishes a new paradigm for sustainable production of high-value triazole intermediates essential to modern drug development pipelines where purity specifications are non-negotiable requirements
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing multi-substituted triazole scaffolds frequently rely on transition metal catalysts such as copper or palladium complexes that introduce significant challenges including costly removal procedures and potential contamination risks compromising final product purity These methods often require strictly controlled anhydrous and oxygen-free environments substantially increasing operational complexity while limiting scalability due to specialized equipment requirements and heightened safety protocols The inherent instability of many metal-catalyzed systems frequently leads to inconsistent yields across different substrate combinations necessitating extensive optimization efforts that delay process development timelines significantly Moreover environmental burdens associated with heavy metal waste streams create additional disposal costs and regulatory compliance issues impacting overall manufacturing economics unfavorably The narrow substrate scope observed restricts applicability when synthesizing structurally diverse triazole derivatives required for modern drug discovery programs where functional group tolerance is paramount
The Novel Approach
In contrast the patented process employs molecular iodine as a benign catalyst operating effectively under standard atmospheric conditions without requiring specialized inert atmosphere equipment or moisture-sensitive handling procedures The reaction proceeds efficiently in dimethyl sulfoxide solvent at moderate temperatures between ninety and one hundred thirty degrees Celsius through a well-defined sequence involving Kornblum oxidation followed by condensation and cyclization steps demonstrating remarkable functional group tolerance across diverse aryl substrates This innovative approach eliminates all heavy metal contamination concerns while maintaining high reaction efficiency as evidenced by consistent yields across multiple substrate combinations documented in patent examples The simplified workup procedure involving basic filtration followed by standard column chromatography significantly reduces processing time compared to traditional methods requiring complex metal removal protocols Most critically demonstrated scalability from laboratory-scale reactions represents substantial advancement addressing longstanding industry challenges in manufacturing high-purity triazole intermediates reliably without compromising quality standards
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The synthetic pathway begins with iodine-mediated oxidation of aryl ethyl ketones in dimethyl sulfoxide solvent where molecular iodine facilitates conversion to α-diketone intermediates through Kornblum-type oxidation mechanisms proceeding without additional oxidizing agents This key transformation occurs through nucleophilic attack by DMSO on iodinated ketone intermediates followed by elimination processes generating reactive diketone species essential for subsequent cyclization steps The resulting α-diketones undergo condensation with trifluoroethylimide hydrazides under mild basic conditions provided by sodium dihydrogen phosphate and pyridine co-catalysts forming hydrazone intermediates that spontaneously cyclize through intramolecular nucleophilic addition reactions Final aromatization completes triazole ring formation through elimination processes yielding desired trisubstituted products with excellent regioselectivity confirmed by comprehensive spectroscopic characterization data This mechanistic sequence demonstrates exceptional efficiency due to synergistic effects between iodine catalysis and DMSOs dual role as both solvent and oxygen source during oxidation phase
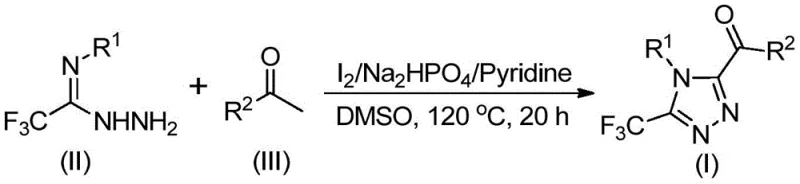
Impurity profile management represents critical advantage where absence of transition metals eliminates common contamination pathways associated with palladium or copper residues requiring extensive purification procedures Traditional syntheses face significant quality control challenges from these metallic impurities which this methodology circumvents entirely Well-defined reaction sequence minimizes side reactions through controlled stepwise transformations preventing over-reaction products commonly observed when using stronger oxidizing agents Patent demonstrates consistent product purity across multiple substrate combinations as evidenced by high-resolution mass spectrometry confirming minimal impurity formation during standard reaction conditions This inherent selectivity translates directly to reduced downstream processing requirements while maintaining pharmaceutical-grade quality standards essential for active pharmaceutical ingredient manufacturing processes Documented tolerance for various functional groups including halogens enhances process robustness accommodating diverse structural modifications without compromising purity outcomes
How to Synthesize Triazole Intermediates Efficiently
This patented methodology provides streamlined approach producing high-purity triazole intermediates through carefully optimized sequence beginning with simple starting materials readily available from commercial sources without requiring specialized handling procedures Reaction demonstrates exceptional reproducibility across different substrate combinations maintaining consistent yield profiles supporting reliable scale-up potential from laboratory development through commercial manufacturing phases Elimination of transition metal catalysts entirely removes significant quality control challenges associated with metal residue testing reducing overall production costs through simplified purification requirements Detailed standardized operating procedures have been developed based on patent disclosure enabling seamless technology transfer while ensuring consistent product quality across different manufacturing scales
- Dissolve aryl ethyl ketone and iodine in dimethyl sulfoxide solvent at room temperature before heating to precisely controlled temperatures between 90–100°C over a four-hour period to facilitate Kornblum oxidation forming aryl diketone intermediates
- Introduce stoichiometric quantities of iodine catalyst along with sodium dihydrogen phosphate buffer system and pyridine base followed by addition of trifluoroethylimide hydrazide reagent under continuous stirring conditions
- Elevate reaction temperature to maintain strict thermal control at approximately 120°C for twenty hours before implementing standard workup procedures including filtration through silica gel followed by column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach delivers substantial value across procurement operations addressing multiple pain points inherent in traditional manufacturing processes for complex heterocyclic intermediates required in pharmaceutical production pipelines Elimination of expensive transition metal catalysts represents fundamental cost reduction opportunity while simultaneously removing significant quality control burdens associated with metal residue testing extending production timelines considerably Utilization of readily available starting materials from established chemical suppliers enhances supply chain resilience reducing dependency on specialized reagents creating vulnerability points during global supply disruptions
- Cost Reduction in Manufacturing: Complete avoidance of transition metal catalysts eliminates direct material costs associated with expensive palladium or copper complexes alongside substantial downstream processing expenses required for metal residue removal through multiple purification steps Streamlined approach reduces overall manufacturing costs through simplified workup procedures requiring only basic filtration followed by standard column chromatography rather than complex extraction protocols typically needed when handling metal-containing reaction mixtures
- Enhanced Supply Chain Reliability: Utilization of commercially available aryl ketones from multiple global suppliers creates robust sourcing options minimizing supply chain disruption risks ensuring consistent material availability regardless of regional market fluctuations or geopolitical factors affecting specialized chemical suppliers This diversification strategy significantly strengthens procurement position while maintaining flexibility during volatile market conditions
- Scalability and Environmental Compliance: Demonstrated scalability from milligram-scale laboratory reactions provides confidence in seamless technology transfer maintaining product quality standards Elimination of toxic heavy metals reduces environmental impact avoiding hazardous waste streams associated with metal catalyst disposal meeting increasingly stringent regulatory requirements for sustainable manufacturing practices without compromising efficiency
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation based on detailed analysis of disclosed capabilities within pharmaceutical manufacturing contexts where purity specifications are critical requirements
Q: How does this iodine-catalyzed method eliminate heavy metal contamination risks compared to conventional approaches?
A: The process utilizes molecular iodine as a benign catalyst instead of transition metals like palladium or copper complexes which typically require extensive purification steps to remove toxic residues below regulatory thresholds This eliminates both direct material costs associated with expensive catalysts and substantial downstream processing expenses while ensuring cleaner final products
Q: What specific advantages does this methodology offer regarding supply chain reliability?
A: The use of commercially available aryl ketones from multiple global suppliers creates robust sourcing options while eliminating dependency on specialized reagents required by traditional methods This enhances resilience against regional market fluctuations or geopolitical disruptions ensuring consistent material availability throughout pharmaceutical manufacturing cycles
Q: How does this synthesis maintain high purity standards without complex purification requirements?
A: The well-defined reaction sequence minimizes side reactions through controlled stepwise transformations that prevent over-reaction products commonly observed with stronger oxidizing agents The inherent selectivity produces cleaner profiles requiring only basic filtration followed by standard column chromatography rather than multi-step extraction protocols
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-million levels This patented triazole synthesis methodology aligns perfectly with our core competencies developing robust manufacturing processes balancing cost efficiency with uncompromising quality standards required by global pharmaceutical clients Our dedicated technical teams ensure seamless integration of novel synthetic routes into existing production frameworks supporting clients throughout technology transfer phases
We invite your technical procurement team to request a Customized Cost-Saving Analysis along with specific COA data and route feasibility assessments tailored to your unique manufacturing requirements through our dedicated technical support channels where our experts will provide comprehensive documentation supporting informed decision-making processes
