Advanced Asymmetric Allylic Amination for Commercial Scale Chiral Nucleoside Production
The pharmaceutical industry's relentless pursuit of potent antiviral therapies has placed chiral carbocyclic nucleosides at the forefront of medicinal chemistry research. Patent CN110590781B discloses a groundbreaking methodology for the synthesis of these critical scaffolds via asymmetric allylic amination. This technology addresses the longstanding challenge of constructing chiral five-membered carbocyclic purine nucleosides with high stereochemical fidelity. By leveraging a palladium-catalyzed coupling between purine bases and alicyclic Morita-Baylis-Hillman (MBH) carbonates, this invention offers a streamlined pathway to compounds analogous to blockbuster drugs like Abacavir and Entecavir. For R&D directors and procurement strategists, this represents a pivotal shift from laborious multi-step syntheses to a concise, catalytic approach that promises enhanced efficiency and reduced environmental footprint in the manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral carbocyclic nucleosides has been plagued by inefficiencies that hinder commercial viability. Traditional strategies often rely on introducing an amino group onto a pre-formed chiral carbocyclic ring, followed by the complex construction of the purine or pyrimidine base. Alternatively, chemists have attempted to couple a fully functionalized chiral carbocycle with a nucleobase, but these methods frequently suffer from poor regioselectivity and low yields. Furthermore, existing asymmetric catalytic routes, such as cycloadditions, often require expensive, specialized catalysts and harsh reaction conditions that are difficult to maintain on a large scale. These legacy processes result in extended production timelines, excessive solvent waste, and significant challenges in controlling the impurity profile, ultimately driving up the cost of goods for the final active pharmaceutical ingredient.
The Novel Approach
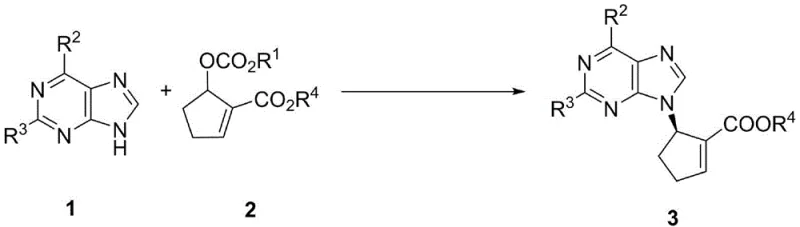
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient palladium-catalyzed asymmetric allylic amination reaction. This method directly couples readily available purine derivatives with alicyclic MBH carbonates in the presence of a chiral ferrocene-derived nitrogen-phosphine ligand. As illustrated in the reaction scheme below, this one-step transformation constructs the critical C-N bond while simultaneously establishing the chiral center with exceptional precision. The process operates under mild conditions, typically between -20°C and 25°C, and achieves diastereoselectivity greater than 95/5 and enantioselectivity up to 99.6%. This dramatic improvement in selectivity minimizes the formation of unwanted isomers, simplifying downstream purification and significantly boosting the overall throughput of the manufacturing process.
Mechanistic Insights into Pd-Catalyzed Asymmetric Allylic Amination
The success of this synthetic route hinges on the sophisticated design of the chiral ligand system. The patent highlights the superiority of ferrocene-derived phosferrox ligands, specifically ligand L6, which features a unique combination of planar chirality from the ferrocene backbone and central chirality from the oxazoline ring. This dual-chirality environment creates a rigid coordination sphere around the palladium center, effectively discriminating between the enantiotopic faces of the allylic intermediate. The mechanism likely proceeds through the formation of a cationic pi-allyl palladium complex, where the bulky substituents on the ligand (such as the isopropyl group in L6) sterically direct the nucleophilic attack of the purine nitrogen. This precise spatial control ensures that the reaction proceeds predominantly at the N9 position of the purine, avoiding the formation of the N7 regioisomer which is a common side reaction in nucleoside chemistry.
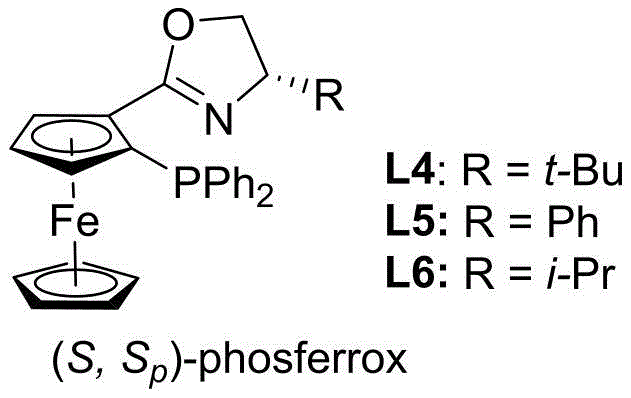
Furthermore, the inclusion of specific additives plays a crucial role in optimizing the reaction kinetics and selectivity. The patent data indicates that phenolic additives, particularly 1-naphthol, significantly enhance both the reaction rate and the yield. It is hypothesized that these additives may assist in the ionization of the MBH carbonate leaving group or stabilize the transition state through hydrogen bonding interactions. This synergistic effect between the palladium catalyst, the chiral ligand, and the acidic additive allows the reaction to proceed with high turnover numbers even at relatively low catalyst loadings (2.5 to 5 mol%). For process chemists, understanding this mechanistic nuance is vital for troubleshooting and scaling the reaction, as it provides levers to adjust selectivity and conversion without changing the core reagents.
How to Synthesize Chiral Five-membered Carbocyclic Purine Nucleosides Efficiently
The practical implementation of this technology involves a straightforward protocol that balances high performance with operational simplicity. The synthesis begins with the preparation of the catalytic system in an inert atmosphere, followed by the sequential addition of substrates and additives. The reaction is monitored until completion, after which standard workup procedures yield the crude product. Due to the high selectivity of the catalyst system, the crude material often requires minimal purification to achieve pharmaceutical-grade purity. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the reaction mixture by combining purine substrate, alicyclic MBH carbonate, palladium catalyst (Pd2(dba)3), chiral ligand (L6), potassium carbonate, and 1-naphthol additive in toluene under nitrogen.
- Stir the reaction mixture at controlled temperatures ranging from -20°C to 25°C for approximately 3 days to ensure complete conversion and high stereoselectivity.
- Upon completion, concentrate the organic phase under vacuum and purify the crude product via column chromatography to isolate the target chiral nucleoside with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric allylic amination technology translates into tangible strategic advantages. The primary benefit lies in the drastic simplification of the supply chain for key antiviral intermediates. By reducing the number of synthetic steps required to access the chiral core, manufacturers can significantly reduce the inventory of raw materials and the associated holding costs. Moreover, the use of commodity chemicals such as toluene as the solvent, rather than exotic or highly regulated solvents, streamlines logistics and reduces the regulatory burden associated with solvent recovery and disposal. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment limitations or stringent temperature control requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly resolution steps and the high atom economy of the coupling reaction. Traditional methods often involve resolving racemic mixtures, which inherently wastes half of the material; this catalytic asymmetric synthesis avoids that loss entirely. Additionally, the high yields and selectivity mean that less raw material is consumed per kilogram of final product, directly lowering the variable cost of production. The ability to use lower catalyst loadings while maintaining performance further contributes to cost efficiency, making the process economically viable for large-scale commercial manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as substituted purines and simple alicyclic carbonates, mitigates the risk of supply disruptions. Unlike specialized chiral pool starting materials that may have limited suppliers, the precursors for this reaction are commoditized and can be sourced from multiple vendors globally. This diversification of the supply base ensures continuity of supply even in volatile market conditions. Furthermore, the scalability of the reaction from gram to multi-ton scales has been demonstrated, providing confidence that the supply chain can expand seamlessly to meet surging demand for antiviral therapies without requiring new process development.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles. The reaction generates fewer byproducts compared to multi-step linear syntheses, reducing the volume of waste that requires treatment. The use of toluene, a solvent with well-established recovery protocols, facilitates efficient recycling, minimizing the environmental footprint. The mild reaction temperatures reduce energy consumption for heating or cooling, contributing to a lower carbon footprint for the manufacturing site. These factors collectively simplify the environmental permitting process and enhance the sustainability profile of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and licensees.
Q: What represents the primary advantage of this asymmetric allylic amination method over traditional nucleoside synthesis?
A: The primary advantage is the direct construction of the chiral center in a single catalytic step with exceptional stereocontrol (up to 99.6% ee), eliminating the need for multiple protection/deprotection steps and resolution processes common in conventional routes.
Q: Is this catalytic system suitable for large-scale industrial production?
A: Yes, the process utilizes robust conditions (0°C to room temperature), commercially available solvents like toluene, and stable palladium/ligand systems, making it highly amenable to scale-up from kilogram to multi-ton manufacturing without requiring cryogenic infrastructure.
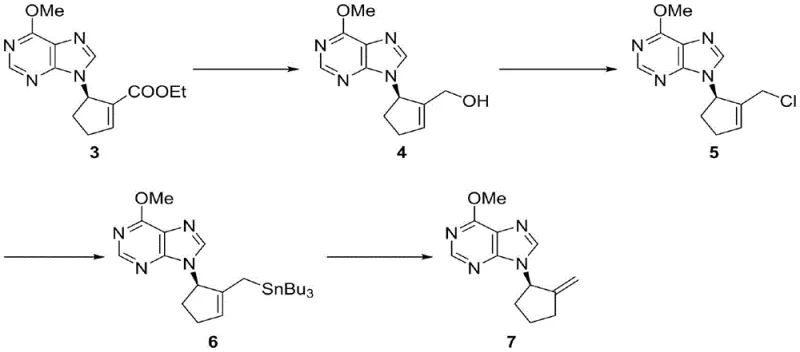
Q: Can the resulting nucleoside products be further functionalized for diverse drug candidates?
A: Absolutely. The synthesized five-membered carbocyclic purine nucleosides contain reactive functionalities, such as the exocyclic double bond or ester groups, which allow for extensive downstream derivatization to create libraries of antiviral and anticancer analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Carbocyclic Purine Nucleosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric allylic amination technology for the global antiviral market. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from pilot plant to full-scale manufacturing. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify enantiomeric excess and structural integrity.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next-generation nucleoside analogs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you secure a competitive advantage in the rapidly evolving landscape of antiviral drug development through superior chemistry and reliable supply chain execution.
