Advanced Asymmetric Hydrogen Transfer for Commercial Scale Chiral Purine Nucleosides
Introduction to Breakthrough Asymmetric Synthesis Technology
The pharmaceutical industry's relentless pursuit of potent antiviral agents has placed chiral carbocyclic purine nucleosides at the forefront of medicinal chemistry research. Compounds such as Abacavir, Entecavir, and Carbovir, which are critical for treating HIV and HBV, rely heavily on the precise stereochemical configuration of their carbocyclic rings. A pivotal advancement in this domain is documented in patent CN109761984B, which discloses a highly efficient method for synthesizing chiral five-membered carbocyclic purine nucleosides via asymmetric hydrogen transfer. This technology represents a paradigm shift from traditional high-pressure hydrogenation, offering a safer, more cost-effective route to these high-value pharmaceutical intermediates. By employing a dynamic kinetic resolution (DKR) strategy with a specialized chiral ruthenium catalyst, the process converts inexpensive racemic starting materials directly into optically pure products with two chiral centers, addressing long-standing challenges in purity and process safety.
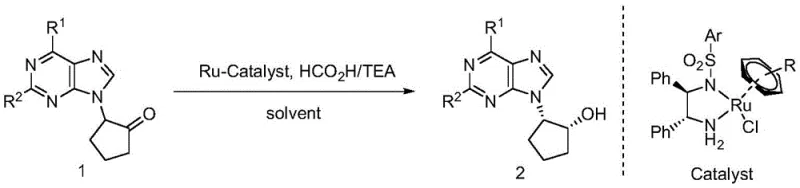
The core innovation lies in the ability to generate complex chiral architectures in a single step under remarkably mild conditions. Traditional routes often suffer from low atom economy or require hazardous reagents, but this patented approach utilizes a formic acid and triethylamine azeotrope as a benign hydrogen source. For R&D directors and process chemists, this methodology offers a robust platform for generating diverse libraries of nucleoside analogs, facilitating the rapid discovery of next-generation therapeutics while ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory standards for chirality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral carbocyclic nucleosides has been plagued by significant technical and economic hurdles. Conventional hydrogenation methods typically rely on high-pressure hydrogen gas (H2) in the presence of metal catalysts to reduce racemic alpha-aminocycloalkanones. While effective in some contexts, this approach necessitates specialized high-pressure reactors, rigorous safety protocols, and substantial capital investment, creating bottlenecks in commercial scale-up of complex pharmaceutical intermediates. Furthermore, alternative asymmetric hydrogen transfer methods using earlier generation catalysts have demonstrated suboptimal catalytic efficiency and poor enantioselectivity. These deficiencies often result in complex purification workflows to remove unwanted stereoisomers, drastically increasing production costs and reducing overall yield. The inability to effectively control the stereochemistry at multiple centers simultaneously has limited the practical application of these older technologies for large-scale manufacturing.
The Novel Approach
The methodology described in CN109761984B overcomes these barriers through a sophisticated dynamic kinetic resolution (DKR) process. By utilizing a specifically designed chiral ruthenium catalyst, the reaction achieves simultaneous racemization of the substrate and asymmetric reduction. This means that even though the starting material is a racemic mixture, the catalyst selectively converts both enantiomers into the desired single chiral product, theoretically allowing for 100% yield from a racemic start. The process operates at ambient temperatures (around 27°C) using formic acid as the hydrogen donor, eliminating the need for dangerous high-pressure infrastructure. This novel approach not only simplifies the operational workflow but also dramatically improves the stereochemical outcome, delivering products with exceptional enantiomeric excess (ee) values reaching up to 99%. Such precision is vital for ensuring the biological efficacy and safety profile of the final antiviral drugs.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogen Transfer
At the heart of this transformative synthesis is the chiral ruthenium catalyst, specifically the TsDPEN-derived complexes illustrated in the patent data. The mechanism involves a concerted outer-sphere hydrogen transfer where the ruthenium hydride species delivers a hydride ion to the carbonyl carbon of the cyclopentanone substrate while a proton is simultaneously transferred from the amine ligand to the carbonyl oxygen. The chiral environment created by the diamine ligand dictates the facial selectivity of this attack, ensuring that only one specific enantiomer of the alcohol product is formed. Crucially, the basic conditions provided by the triethylamine facilitate the rapid racemization of the unreacted ketone substrate via enolization. This dynamic equilibrium ensures that the 'wrong' enantiomer of the starting material is constantly converted into the 'right' one for the catalyst to act upon, driving the reaction to completion with high optical purity.
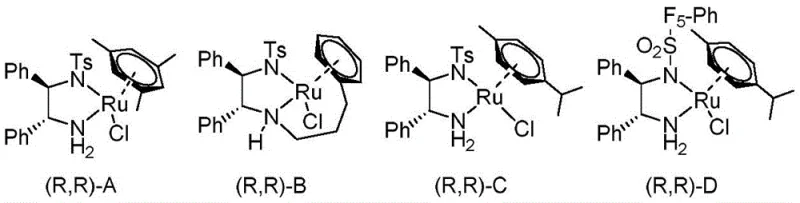
Impurity control is inherently built into this mechanistic design. The high diastereoselectivity (>99:1 dr) observed in the reaction minimizes the formation of cis/trans isomers, which are notoriously difficult to separate chromatographically on a large scale. The specific electronic and steric properties of Catalyst D, featuring a p-cymene ligand and a sulfonylated diamine backbone, have been optimized to maximize this selectivity. For quality assurance teams, this means the crude product profile is significantly cleaner, reducing the burden on downstream purification steps. The robustness of the catalyst system allows it to tolerate various substituents on the purine ring (such as methoxy, ethoxy, or thio groups) without compromising stereocontrol, making it a versatile tool for synthesizing a broad spectrum of nucleoside analogs required for modern antiviral therapy.
How to Synthesize Chiral Carbocyclic Purine Nucleosides Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and solvent selection to maximize efficiency. The patent outlines a streamlined protocol where the racemic alpha-purine substituted cyclopentanone is dissolved in a solvent like dioxane, followed by the addition of the chiral ruthenium catalyst and the formic acid/triethylamine hydrogen source. The reaction proceeds smoothly at mild temperatures, typically completing within 24 hours. Detailed standardized operating procedures regarding stoichiometry, workup, and purification are essential for reproducibility. For a comprehensive guide on executing this transformation in a GMP environment, please refer to the technical steps outlined below.
- Prepare the reaction mixture by combining racemic alpha-purine substituted cyclopentanone, chiral ruthenium catalyst (specifically Catalyst D), and an organic solvent such as dioxane.
- Add the hydrogen source mixture, consisting of formic acid and triethylamine (typically in a 1: 1 molar ratio), to the reaction vessel under inert atmosphere.
- Maintain the reaction temperature between 0°C and 30°C for approximately 24 hours to achieve dynamic kinetic resolution, yielding the chiral alcohol product with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift from high-pressure hydrogenation to ambient pressure hydrogen transfer fundamentally alters the cost structure of manufacturing these critical intermediates. By removing the requirement for expensive high-pressure reactors and the associated safety infrastructure, the capital expenditure (CAPEX) for setting up production lines is significantly reduced. Furthermore, the use of formic acid as a liquid hydrogen source simplifies logistics and storage compared to managing high-pressure gas cylinders, leading to substantial cost savings in operational expenditures (OPEX). The ability to use cheap, readily available racemic starting materials instead of expensive chiral pools further drives down the raw material costs, enhancing the overall economic viability of the process.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of inexpensive hydrogen donors drastically lower the barrier to entry for production. The high catalytic efficiency means lower catalyst loading is required to achieve full conversion, reducing the cost per kilogram of the final product. Additionally, the high selectivity minimizes waste generation and the need for extensive chromatographic purification, which is often the most expensive step in pharmaceutical manufacturing. This streamlined process flow translates directly into a more competitive pricing structure for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on racemic starting materials mitigates the supply risks associated with sourcing scarce chiral building blocks. Since the dynamic kinetic resolution creates chirality during the synthesis, the supply chain is no longer dependent on the availability of specific enantiomers from natural sources or resolution processes. This independence ensures a more stable and continuous supply of raw materials, reducing lead time for high-purity pharmaceutical intermediates and protecting against market volatility. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by equipment maintenance or safety shutdowns.
- Scalability and Environmental Compliance: The mild reaction conditions (0-30°C) and the absence of hazardous high-pressure gases make this process inherently safer and easier to scale from laboratory to industrial tonnage. The simplified safety profile reduces the regulatory burden and insurance costs associated with chemical manufacturing. Moreover, the atom economy of the hydrogen transfer reaction is superior to many traditional reduction methods, resulting in less chemical waste. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, ensuring long-term sustainability and cost reduction in API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogen transfer technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the process capabilities and limitations for potential partners and stakeholders.
Q: What are the advantages of this asymmetric hydrogen transfer method over traditional high-pressure hydrogenation?
A: Unlike traditional methods requiring dangerous high-pressure hydrogen gas and complex equipment, this patented process utilizes a safe formic acid/triethylamine system at ambient temperatures (27°C), significantly reducing operational risks and capital expenditure while achieving superior enantioselectivity (up to 99% ee).
Q: Can this synthesis method be scaled for industrial production of antiviral intermediates?
A: Yes, the process is highly scalable due to its mild reaction conditions and the use of readily available racemic starting materials. The dynamic kinetic resolution mechanism ensures high efficiency, making it suitable for commercial scale-up of complex pharmaceutical intermediates like those used in HIV and HBV treatments.
Q: How does the catalyst selection impact the stereochemical outcome of the nucleoside synthesis?
A: The selection of the specific chiral ruthenium catalyst, particularly the TsDPEN-derived Catalyst D, is critical. It facilitates dynamic kinetic resolution by rapidly racemizing the starting ketone while selectively reducing one enantiomer, thereby converting the entire racemic pool into a single desired chiral product with excellent diastereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Carbocyclic Purine Nucleosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient chiral synthesis in the development of life-saving antiviral medications. Our team of expert process chemists has extensively evaluated the technology disclosed in CN109761984B and possesses the technical expertise to implement this advanced asymmetric hydrogen transfer route. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from pilot scale to full commercial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise chiral HPLC analysis to guarantee the enantiomeric excess required for regulatory approval.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your nucleoside projects. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and detailed route feasibility assessments. Let us help you secure a reliable supply of high-quality chiral intermediates while optimizing your production costs and accelerating your time to market.
