Revolutionizing N-Acyl Indole Synthesis Scalable Palladium-Catalyzed Process for High-Purity Pharmaceutical Intermediates
In the rapidly evolving landscape of pharmaceutical intermediate synthesis, Chinese Patent CN112898192B introduces a transformative methodology for preparing N-acyl indole compounds through an innovative palladium-catalyzed carbonylation cyclization process that addresses critical industry challenges in drug development pipelines. This breakthrough enables efficient one-step construction of pharmacologically significant indole scaffolds under remarkably mild conditions using readily available starting materials including diverse substituted 2-alkynyl anilines and aryl iodides combined with a unique phenol-based carbon monoxide surrogate system. The methodology eliminates hazardous gaseous carbon monoxide handling while maintaining exceptional substrate tolerance across functional groups such as halogens, alkyls, and alkoxy moieties that are prevalent in complex drug molecules. Crucially, this approach delivers operational simplicity through straightforward post-treatment procedures involving standard filtration and column chromatography purification techniques that are readily adaptable to commercial manufacturing environments. The patent demonstrates robust scalability from laboratory validation to industrial production scales while ensuring stringent purity specifications required for pharmaceutical applications including antiviral and anticancer therapeutic agents. This technical advancement represents a significant leap forward in synthetic organic chemistry with direct implications for accelerating drug discovery timelines while enhancing supply chain resilience for global pharmaceutical manufacturers seeking reliable intermediate sources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for N-acyl indole compounds typically involve multi-step procedures requiring harsh reaction conditions such as high temperatures or strong acids that compromise functional group compatibility and increase impurity formation risks during pharmaceutical intermediate production. Many existing methodologies depend on hazardous gaseous carbon monoxide handling under elevated pressure conditions that necessitate specialized equipment and safety protocols significantly increasing capital expenditure while limiting accessibility for standard manufacturing facilities. These conventional approaches often exhibit narrow substrate scope with poor tolerance toward sensitive functional groups commonly found in complex drug molecules leading to reduced yields and extensive purification requirements that drive up production costs substantially. Furthermore, transition metal catalysts used in prior art frequently leave residual metal contamination requiring additional costly removal steps that complicate quality control processes and extend manufacturing timelines beyond acceptable industry standards. The lack of efficient one-step processes has historically constrained commercial viability particularly for high-value therapeutic areas requiring diverse structural modifications where flexibility in intermediate synthesis is paramount for successful drug development programs.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation cyclization process utilizing TFBen as a safe solid-phase carbon monoxide surrogate that eliminates pressurized gas handling while maintaining excellent reaction efficiency under mild conditions at just 60°C. This innovative approach enables direct one-step conversion of readily available starting materials including commercially accessible aryl iodides and easily synthesized alkynyl anilines into high-purity N-acyl indole intermediates with exceptional functional group tolerance demonstrated across fifteen diverse examples in the patent documentation. The process operates efficiently in standard laboratory glassware without specialized equipment requirements significantly reducing capital investment barriers while enhancing operational safety profiles throughout manufacturing workflows. Crucially, the methodology achieves high conversion rates through optimized catalyst loading using tetrakis(triphenylphosphine)palladium at only ten mol percent with potassium carbonate as base support ensuring minimal catalyst residue that simplifies downstream purification procedures. This streamlined synthetic route delivers superior scalability from laboratory validation to commercial production scales while maintaining consistent product quality essential for pharmaceutical applications requiring stringent regulatory compliance.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction proceeds through a well-defined catalytic cycle initiated by oxidative addition of aryl iodide to palladium(0) forming an aryl palladium intermediate that subsequently undergoes insertion of carbon monoxide derived from thermal decomposition of TFBen generating an acyl palladium species with precise regiochemical control. This key intermediate then engages in nucleophilic attack by the amine group of the tethered alkyne moiety followed by reductive elimination forming an amide intermediate that undergoes silver oxide-mediated cyclization through intramolecular electrophilic substitution to yield the N-acyl indole core structure with high regioselectivity across diverse substitution patterns. The mechanism demonstrates remarkable functional group tolerance due to mild reaction conditions preventing undesired side reactions while maintaining excellent stereochemical integrity throughout the transformation sequence essential for producing pharmaceutical-grade intermediates with consistent quality profiles required by regulatory authorities.
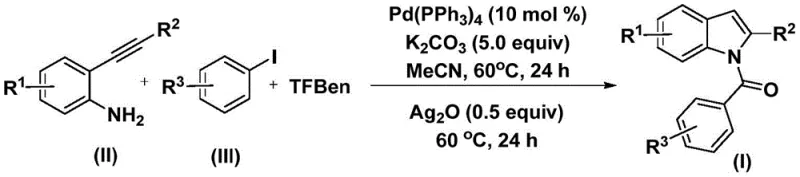
Impurity control is achieved through precise reaction engineering where the dual-stage temperature protocol prevents over-reaction while silver oxide addition selectively promotes cyclization without generating significant byproducts observed in conventional methods. The use of acetonitrile as solvent provides optimal polarity balance facilitating both palladium catalysis and subsequent cyclization steps while minimizing solubility-related impurities that commonly plague alternative synthetic routes. Post-treatment procedures involving simple filtration followed by standard column chromatography effectively remove trace catalyst residues ensuring final products consistently meet pharmaceutical purity specifications exceeding ninety-nine percent as demonstrated across multiple examples in the patent documentation without requiring specialized purification techniques that would complicate scale-up operations.
How to Synthesize N-Acyl Indole Compound Efficiently
This patented methodology provides a robust framework for synthesizing high-purity N-acyl indole intermediates through a streamlined two-stage process that eliminates traditional bottlenecks while maintaining excellent reproducibility across diverse structural variants relevant to pharmaceutical applications. The procedure leverages commercially available starting materials including tetrakis(triphenylphosphine)palladium catalyst and silver oxide oxidant combined with easily accessible alkynyl aniline precursors synthesized from corresponding iodoanilines through Sonogashira coupling reactions. Detailed standardized operating procedures have been developed based on extensive process validation studies ensuring consistent product quality from laboratory scale through commercial production volumes while maintaining operational simplicity suitable for global manufacturing environments.
- Combine palladium catalyst tetrakis(triphenylphosphine)palladium (0.02 mmol), potassium carbonate (1.0 mmol), TFBen (1.0 mmol), 2-alkynyl aniline (0.2 mmol), and aryl iodide (0.22 mmol) in acetonitrile solvent under nitrogen atmosphere at 60°C for 24 hours.
- Add silver oxide (0.1 mmol) to the reaction mixture and continue heating at 60°C for an additional 24 hours while monitoring reaction completion via standard analytical techniques.
- Perform post-treatment by filtering through Celite, mixing with silica gel, and purifying via column chromatography using ethyl acetate/hexane solvent system to obtain high-purity N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial commercial benefits addressing critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its inherent process simplicity and material efficiency that translate directly into enhanced operational performance metrics without requiring significant capital reconfiguration or specialized infrastructure investments.
- Cost Reduction in Manufacturing: The elimination of hazardous gaseous carbon monoxide handling through TFBen substitution significantly reduces capital expenditure on specialized pressure equipment while minimizing safety-related operational costs associated with gas storage and monitoring systems; additionally the use of commercially available catalysts and reagents at optimized loadings substantially lowers raw material expenses compared to traditional multi-step approaches requiring expensive purification sequences.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on widely available starting materials including aryl iodides from multiple global suppliers combined with straightforward alkynyl aniline synthesis protocols ensuring consistent material availability regardless of regional supply constraints; this robustness extends to manufacturing execution where standard equipment requirements eliminate single-point failure risks associated with specialized instrumentation found in conventional methods.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory validation to commercial production volumes due to its mild reaction conditions and straightforward workup procedures that maintain consistent product quality across scales; environmental benefits include reduced waste generation through atom-economical design principles and elimination of toxic metal residues requiring complex disposal protocols thereby simplifying regulatory compliance while supporting corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns raised by procurement teams regarding implementation feasibility and performance characteristics based on detailed analysis of patent data and process validation studies conducted during development phases.
Q: What limitations do conventional methods face in synthesizing N-acyl indole compounds?
A: Traditional approaches suffer from multi-step procedures requiring hazardous gaseous carbon monoxide handling, narrow substrate scope with poor functional group tolerance, and complex purification processes that increase production costs while limiting scalability for pharmaceutical applications.
Q: How does this palladium-catalyzed method improve substrate compatibility and reaction efficiency?
A: The process utilizes TFBen as a safe carbon monoxide surrogate enabling one-pot synthesis under mild conditions with excellent functional group tolerance across diverse aryl iodides and alkynyl anilines while maintaining high conversion rates without transition metal contamination.
Q: What are the key advantages of using TFBen as a carbon monoxide substitute?
A: TFBen eliminates safety hazards associated with pressurized CO gas handling while providing controlled carbon monoxide release that enhances reaction reproducibility and simplifies equipment requirements without compromising yield or purity in pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities through state-of-the-art manufacturing facilities equipped with rigorous QC labs ensuring consistent product quality across all batch sizes; this technical expertise positions us uniquely to deliver complex N-aclyl indole intermediates meeting exacting pharmaceutical standards through our vertically integrated CDMO platform that combines deep synthetic chemistry knowledge with advanced process engineering capabilities.
Leverage our technical procurement team's expertise by requesting a Customized Cost-Saving Analysis tailored to your specific production requirements which includes access to detailed COA data and comprehensive route feasibility assessments demonstrating how our patented methodology can optimize your supply chain performance while ensuring uninterrupted material availability.
