Advanced Recovery of High-Purity Diketone from Complex Steroid Mother Liquors for Commercial Scale-Up
The pharmaceutical and fine chemical industries are increasingly focused on sustainable manufacturing practices, particularly in the complex realm of steroid synthesis where raw material costs are high and waste streams are chemically intricate. Patent CN115322239A introduces a groundbreaking methodology for the recovery of high-value diketone intermediates from the mother liquor generated during the production of double-alkyne carbon-lost esters. This innovation addresses a critical bottleneck in steroid manufacturing, where traditional purification methods often fail to recover active ingredients from complex mixtures containing various chiral isomers and by-products. By converting waste mother liquor back into a usable starting material, this technology not only enhances the overall atom economy of the synthesis route but also provides a robust solution for supply chain stability. The process leverages specific solvent systems and base-catalyzed reactions to achieve high recovery rates without compromising the structural integrity of the steroid backbone.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
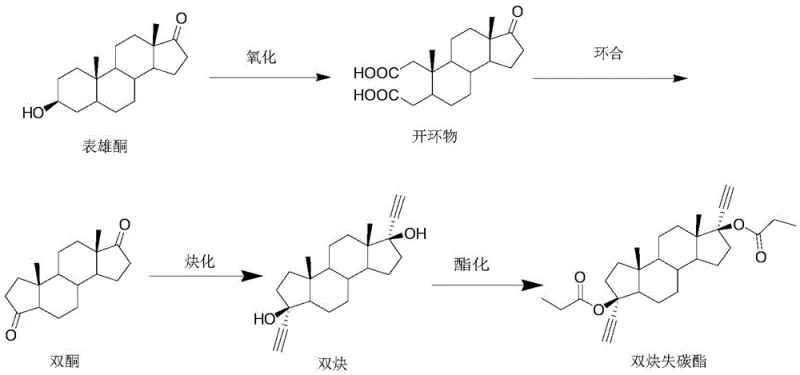
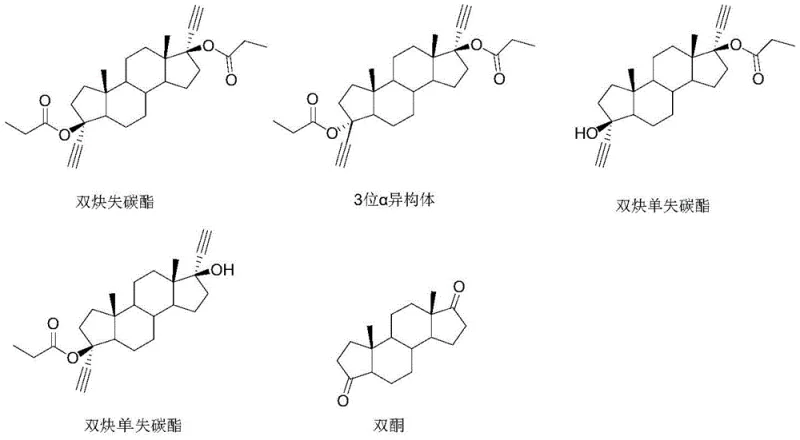
Historically, the management of mother liquor from the synthesis of diacetylene carbon-lost esters has been a significant challenge for process chemists and plant managers alike. The mother liquor typically contains a complex mixture comprising approximately 50-60% of the desired diacetylene carbon-lost ester, 20-30% of the 3-position alpha isomer, and 10-30% of other impurities including mono-carbon-lost esters. Conventional approaches often involve repeated solvent refining in an attempt to isolate the target ester, but this is frequently inefficient due to the similar physicochemical properties of the 3-alpha chiral isomer and the target molecule. In many cases, the difficulty in separating these components leads manufacturers to treat the mother liquor as hazardous waste, resulting in substantial financial loss and environmental burden. Furthermore, attempts to chemically revert these materials using harsh aqueous conditions, such as refluxing with potassium hydroxide in water, have historically yielded poor results, with product purity dropping as low as 23% due to the poor solubility of steroid compounds in aqueous media.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing a two-stage chemical conversion strategy that effectively resets the molecular clock of the waste stream. Instead of attempting the difficult physical separation of isomers, the new process chemically transforms the entire mixture of diacetylene esters and isomers back into the precursor diketone. This is achieved through a mild hydrolysis step followed by a highly selective de-ethynylation reaction using potassium tert-butoxide in anhydrous tetrahydrofuran. This approach bypasses the solubility limitations of aqueous bases, ensuring that the steroid skeleton remains intact while the unwanted alkyne and ester functional groups are cleanly removed. The result is a streamlined pathway that turns a low-value waste stream into a high-purity raw material, effectively closing the loop in the manufacturing cycle and drastically reducing the reliance on fresh starting materials like epiandrosterone.
Mechanistic Insights into Base-Catalyzed De-ethynylation and Hydrolysis
The core of this recovery process lies in the precise control of reaction conditions during the de-ethynylation step, which is critical for preserving the stereochemistry of the steroid nucleus. The use of potassium tert-butoxide (t-BuOK) in anhydrous tetrahydrofuran (THF) is a masterstroke of solvent engineering; THF provides excellent solubility for the lipophilic steroid intermediates, ensuring a homogeneous reaction phase that is impossible to achieve with water. The tert-butoxide anion acts as a strong, non-nucleophilic base that facilitates the elimination of the acetylene groups at the 2-alpha and 17-alpha positions without attacking the sensitive ketone functionalities elsewhere in the molecule. This selectivity is paramount, as harsher nucleophilic conditions could lead to aldol condensations or epimerization, which would ruin the batch. The reflux conditions in THF allow for sufficient thermal energy to drive the reaction to completion over a 7 to 8-hour period, ensuring that even the sterically hindered isomers are fully converted.
Following the de-ethynylation, the purification mechanism relies on the differential solubility of the product versus remaining impurities in hot ethanol. The addition of activated carbon during the reflux in 95% ethanol serves a dual purpose: it adsorbs colored polymeric by-products and trace metal residues, while the subsequent cooling crystallization allows the pure diketone to precipitate out of the solution. The patent specifies a crystallization temperature of 0 to 5 degrees Celsius, which maximizes the yield by minimizing the solubility of the product in the mother liquor. This rigorous refining protocol ensures that the final recovered diketone meets stringent purity specifications of greater than 95%, making it chemically equivalent to virgin material and suitable for re-entry into the main synthesis line for the production of contraceptive intermediates.
How to Synthesize Diketone Efficiently
The synthesis of diketone from waste mother liquor is a sophisticated three-step operation that requires precise control over stoichiometry and temperature to ensure optimal recovery. The process begins with the hydrolysis of the ester groups, followed by the critical de-ethynylation step, and concludes with a standard recrystallization. Detailed operational parameters, including specific mass ratios of solvents to reactants and exact monitoring intervals via HPLC, are essential for replicating the high yields reported in the patent data. For process engineers looking to implement this recycling loop, adhering to the specific solvent ratios and base concentrations is vital to prevent side reactions.
- Hydrolyze the mother liquor using a THF/Ethanol/NaOH mixture at room temperature to remove ester groups.
- Perform de-ethynylation by refluxing the crude product with potassium tert-butoxide in anhydrous tetrahydrofuran.
- Refine the resulting solid via ethanol recrystallization with activated carbon to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this recovery technology offers a compelling value proposition centered on cost avoidance and resource security. By reclaiming valuable steroid intermediates from what was previously considered waste, manufacturing facilities can significantly reduce their consumption of expensive upstream raw materials such as epiandrosterone. This internal recycling loop acts as a hedge against market volatility in the pricing of natural steroid precursors, providing a more predictable cost structure for long-term production planning. Furthermore, the ability to convert complex isomeric mixtures into a single, usable intermediate simplifies inventory management and reduces the volume of hazardous chemical waste that requires costly disposal, thereby lowering the overall environmental compliance burden of the facility.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic reduction in raw material costs associated with the synthesis of diacetylene carbon-lost esters. By recovering the diketone intermediate with a total yield exceeding 70%, the effective cost per kilogram of the final active pharmaceutical ingredient is lowered substantially. This efficiency gain is achieved without the need for expensive exotic catalysts or high-pressure equipment, relying instead on conventional reagents like sodium hydroxide and potassium tert-butoxide which are readily available and inexpensive. The elimination of the need to purchase fresh starting material for every batch translates directly into improved gross margins for the manufacturing operation.
- Enhanced Supply Chain Reliability: Integrating this recovery process creates a more resilient supply chain by diversifying the sources of critical intermediates. Instead of relying solely on external suppliers for fresh diketone or epiandrosterone, manufacturers can generate a significant portion of their feedstock internally from their own process waste. This self-sufficiency reduces the risk of supply disruptions caused by external logistics issues or raw material shortages. Additionally, the robustness of the chemical process, which tolerates a wide range of impurity profiles in the input mother liquor, ensures consistent output quality even when the composition of the waste stream fluctuates, guaranteeing a steady flow of materials for downstream synthesis.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as stirring, filtration, and distillation that are easily transferable from pilot scale to multi-ton commercial production. The use of tetrahydrofuran and ethanol, both of which can be recovered and reused through distillation, minimizes solvent waste and aligns with green chemistry principles. By converting hazardous steroid waste into a useful product, the facility reduces its environmental footprint and potential liability associated with waste disposal. This alignment with sustainability goals not only satisfies regulatory requirements but also enhances the corporate reputation of the manufacturer in an increasingly eco-conscious global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recovery technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this recycling loop into existing manufacturing lines.
Q: Why is potassium tert-butoxide preferred over potassium hydroxide for de-ethynylation?
A: Potassium tert-butoxide offers superior solubility in organic solvents like tetrahydrofuran compared to inorganic bases in water, leading to significantly higher reaction yields and product purity for steroid compounds.
Q: What is the purity level of the recovered diketone?
A: The patented method consistently achieves a purity of greater than 95%, with specific examples demonstrating up to 96% purity, making it suitable for reuse in pharmaceutical synthesis.
Q: Can this method handle isomeric impurities?
A: Yes, the process effectively converts difficult-to-separate isomers, such as the 3-alpha isomer found in the mother liquor, back into the target diketone structure, maximizing material utilization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate recovery in the competitive landscape of steroid manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recovery routes like the one described in CN115322239A can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of recovered diketone meets the highest standards required for pharmaceutical applications. Our commitment to technical excellence allows us to offer customized solutions that maximize material utilization and minimize waste for our global partners.
We invite you to explore how our advanced recovery technologies can optimize your production costs and enhance your supply chain resilience. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with a leader in fine chemical innovation. Let us help you turn your process challenges into profitable opportunities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
