Advanced Synthesis of Polysubstituted Beta-Lactams for Next-Generation Antibiotics
Introduction to Patent CN113801050A
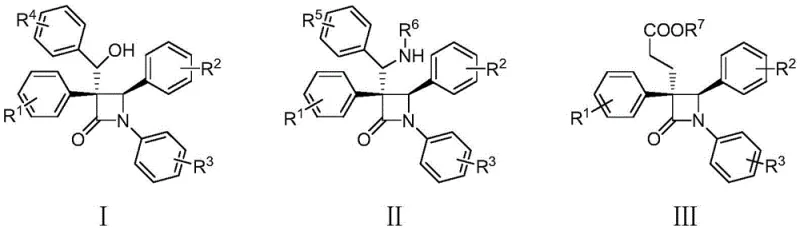
The pharmaceutical industry continuously seeks robust synthetic routes for beta-lactam scaffolds, which serve as the critical core structure for a vast array of antibiotics, including penicillins and cephalosporins. Patent CN113801050A introduces a groundbreaking methodology for the preparation of polysubstituted beta-lactam compounds, specifically targeting structures defined by general formulas I, II, and III. This innovation addresses a long-standing challenge in organic synthesis by leveraging a copper-catalyzed interrupted Kinugasa reaction. Unlike conventional approaches that often struggle with limited functionalization, this technique enables the construction of highly complex heterocyclic systems through a streamlined one-pot process. The significance of this development lies in its potential to accelerate the discovery of novel antibiotics, particularly in the face of growing antimicrobial resistance. By providing access to diverse chemical space with high enantioselectivity, this patent offers a valuable toolkit for R&D teams focused on next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-lactam rings has relied heavily on the classic Kinugasa reaction, first reported in 1972. While effective for generating basic 3,4-disubstituted beta-lactams from terminal alkynes and nitrones, this traditional pathway suffers from a significant mechanistic bottleneck. The reaction proceeds through a copper enolate intermediate which invariably undergoes protonation to terminate the cycle. This inherent limitation restricts the structural diversity of the final products, preventing the direct installation of additional functional groups at the C3 or C4 positions without requiring tedious post-synthetic modifications. Furthermore, many existing methods for creating more complex beta-lactams demand harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that drastically reduce overall atom economy and increase waste generation. These factors collectively hinder the efficient production of advanced antibiotic candidates needed for modern medicine.
The Novel Approach
The methodology disclosed in CN113801050A fundamentally alters the reaction trajectory by intercepting the reactive copper enolate intermediate before protonation can occur. By introducing a third component—such as a benzaldehyde, imine, or olefin—the process effectively "interrupts" the standard Kinugasa cycle. This strategic modification allows for a tandem cascade where the initial cycloaddition is immediately followed by a nucleophilic addition to the external electrophile. The result is a direct, one-pot synthesis of highly functionalized polysubstituted beta-lactams. This approach not only bypasses the need for separate functionalization steps but also operates under remarkably mild conditions, typically between -20°C and room temperature. The ability to seamlessly integrate three distinct building blocks into a single chiral scaffold represents a substantial leap forward in synthetic efficiency and versatility.
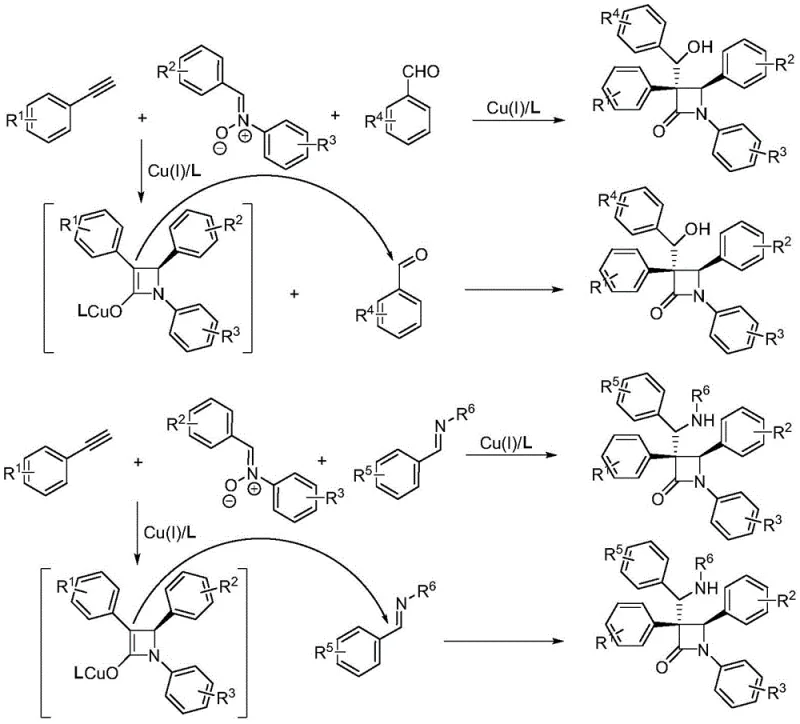
Mechanistic Insights into Copper-Catalyzed Interrupted Kinugasa Reaction
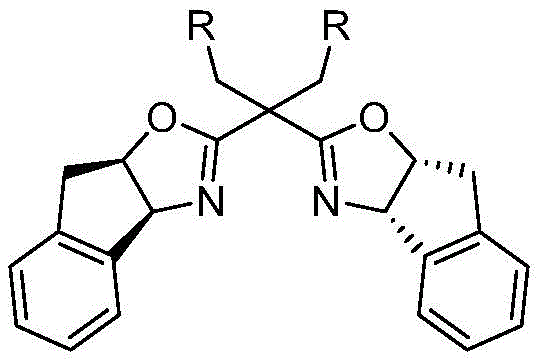
The success of this transformation hinges on the precise coordination between the copper catalyst and a specialized chiral ligand system. The reaction initiates with the activation of the terminal alkyne by a monovalent copper species, forming a copper acetylide. This species then undergoes a [3+2] cycloaddition with the nitrone dipole to generate a five-membered isoxazolidine intermediate, which subsequently rearranges to form the crucial four-membered beta-lactam ring bearing a copper enolate moiety. In traditional scenarios, this enolate would simply abstract a proton from the solvent or additives. However, in this interrupted protocol, the presence of a strong electrophile (the third component) competes effectively for the nucleophilic enolate carbon. The stereochemical outcome is rigorously controlled by the chiral environment provided by the ligand, ensuring high enantiomeric excess (ee) values, often exceeding 95% as demonstrated in the patent examples.
Central to this stereocontrol is the use of an indenyl-substituted chiral bisoxazoline ligand, as depicted in the structural data. This specific ligand architecture creates a rigid chiral pocket around the copper center, directing the approach of the reactants and stabilizing the transition state leading to the desired enantiomer. The patent highlights that replacing this specific ligand with alternatives results in a failure to obtain the target products, underscoring the critical nature of the catalyst system design. Furthermore, the choice of base, such as potassium carbonate, and solvent, preferably acetonitrile, plays a vital role in maintaining the reactivity of the copper enolate while suppressing side reactions. This delicate balance allows for the efficient trapping of the intermediate, yielding products with excellent diastereoselectivity and structural integrity suitable for downstream pharmaceutical applications.
How to Synthesize Polysubstituted Beta-Lactams Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The general procedure involves combining the phenylacetylene derivative, the nitrone, and the chosen third component in a dry solvent under an inert atmosphere. The specific molar ratios are critical, with the patent recommending a slight excess of the nitrone and third component relative to the alkyne to drive the reaction to completion. The addition of the copper catalyst and chiral ligand must be performed at low temperatures to control the exothermic nature of the initial cycloaddition. Following the reaction period, standard workup procedures involving filtration through celite and silica gel chromatography allow for the isolation of the pure diastereomers. For detailed operational parameters and specific stoichiometric calculations tailored to your specific substrate, please refer to the standardized guide below.
- Prepare the reaction mixture by combining phenylacetylene, nitrone, and a third component (aldehyde, imine, or olefin) in acetonitrile under inert atmosphere.
- Add copper(I) catalyst (e.g., Cu(CH3CN)4PF6), chiral bisoxazoline ligand, and potassium carbonate base to the mixture at 0°C.
- Stir the reaction from 0°C to room temperature for 12-48 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial manufacturing perspective, the adoption of this interrupted Kinugasa reaction offers compelling advantages over legacy synthetic routes. The primary benefit stems from the drastic simplification of the process flow. By consolidating what would traditionally be a multi-step sequence into a single one-pot operation, manufacturers can significantly reduce the consumption of solvents, reagents, and energy. This consolidation directly translates to lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry principles. Moreover, the reliance on commercially available starting materials such as substituted phenylacetylenes and nitrones ensures a stable and reliable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps between the ring formation and functionalization phases leads to substantial cost savings. Traditional methods often require protecting group strategies and separate reaction vessels for each transformation, which inflates both material costs and labor hours. By contrast, this tandem process minimizes unit operations, thereby enhancing overall process efficiency. Additionally, the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium further optimizes the cost structure, making the production of high-value antibiotic intermediates more economically viable on a large scale.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater supply chain resilience. Operating at near-ambient temperatures (-20°C to room temperature) reduces the need for specialized cryogenic equipment or high-energy heating systems, simplifying facility requirements. The tolerance of the system to various functional groups on the aromatic rings allows for the use of diverse feedstock sources without compromising reaction performance. This flexibility ensures that procurement teams can source raw materials from multiple vendors without fearing batch-to-batch variability, thus securing continuous production schedules and reducing lead times for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated effectively in laboratory settings with straightforward workup procedures. The use of acetonitrile, a common industrial solvent, facilitates easy recovery and recycling, supporting sustainable manufacturing practices. Furthermore, the high atom economy of the three-component coupling means less chemical waste is generated per kilogram of product. This efficiency aids in meeting stringent environmental regulations regarding waste disposal and emissions, positioning manufacturers as responsible partners in the global pharmaceutical supply network while maintaining high throughput capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. Understanding these details is essential for process chemists and project managers evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: What distinguishes the Interrupted Kinugasa Reaction from the traditional method?
A: Traditional Kinugasa reactions typically terminate via protonation to yield 3,4-disubstituted beta-lactams. The interrupted variant traps the copper enolate intermediate with a third electrophilic component, enabling the synthesis of highly functionalized polysubstituted derivatives.
Q: What represents the key advantage of this synthesis for supply chain stability?
A: The process utilizes readily available starting materials like phenylacetylenes and nitrones in a one-pot cascade, significantly simplifying the operational workflow and reducing the dependency on complex, multi-step precursor synthesis.
Q: Is this method scalable for commercial production of API intermediates?
A: Yes, the reaction operates under mild conditions (0°C to room temperature) using standard solvents like acetonitrile and common bases, making it highly amenable to scale-up from laboratory to industrial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Beta-Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the copper-catalyzed interrupted Kinugasa reaction in accelerating drug discovery. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts can be seamlessly translated into industrial reality. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards. We understand that the development of novel antibiotics requires not just chemical expertise but also a partner who can navigate the complexities of regulatory compliance and supply chain logistics with precision.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of complex beta-lactam intermediates or process optimization for existing routes, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions and secure a competitive advantage in the global marketplace.
