Advanced Synthesis of Polysubstituted Beta-Lactams for Next-Generation Antibiotic Development
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those serving as core structures for antibiotics. Patent CN113801050A introduces a significant advancement in this domain by disclosing a novel preparation method for polysubstituted beta-lactam compounds. These compounds, characterized by general formulas I, II, and III, represent a critical class of intermediates with potential applications in developing new antibiotics to combat drug-resistant bacteria. The innovation lies in the utilization of a copper-catalyzed interrupted Kinugasa reaction, which effectively transforms simple starting materials into highly functionalized four-membered ring systems. This approach addresses the long-standing challenge of accessing diverse substitution patterns on the beta-lactam core, which is often difficult to achieve through conventional cyclization strategies.
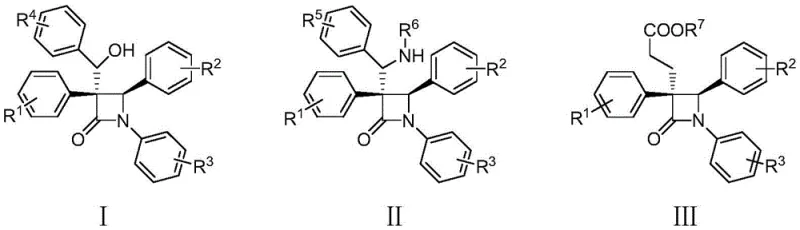
As a reliable pharmaceutical intermediate supplier, understanding the structural versatility offered by this patent is crucial. The ability to introduce various substituents at multiple positions on the beta-lactam ring opens new avenues for medicinal chemistry optimization. The patent highlights that these compounds are not merely theoretical constructs but are accessible through a streamlined synthetic route that avoids the need for complex, multi-step protection and deprotection sequences often associated with beta-lactam synthesis. This technological breakthrough provides a solid foundation for the cost reduction in API manufacturing, as it simplifies the supply chain for key antibiotic precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-lactams has relied heavily on the classic Kinugasa reaction, first reported in 1972. While effective for generating 3,4-disubstituted beta-lactams, this traditional method suffers from inherent limitations regarding structural diversity. The mechanism involves the formation of a copper enolate intermediate which inevitably undergoes protonation to yield the final product. This protonation step acts as a terminal point, preventing further functionalization at the alpha-position relative to the carbonyl group. Consequently, chemists are restricted to a narrow subset of beta-lactam derivatives, limiting the exploration of structure-activity relationships in drug discovery. Furthermore, alternative methods often require harsh reaction conditions, expensive reagents, or tedious multi-step sequences that negatively impact overall yield and environmental sustainability.
The Novel Approach
The methodology described in CN113801050A overcomes these barriers by employing an interrupted Kinugasa strategy. By introducing a third component—such as a benzaldehyde, imine, or olefin—into the reaction mixture, the protonation of the key copper enolate intermediate is blocked. Instead, the intermediate undergoes a nucleophilic addition with the third component, resulting in a tandem cyclization-functionalization sequence. This one-pot process allows for the direct assembly of polysubstituted beta-lactams with high complexity from simple precursors. The reaction conditions are notably mild, typically proceeding at temperatures between -20°C and room temperature, which preserves sensitive functional groups and reduces energy consumption. This shift from a two-component to a three-component coupling represents a paradigm shift in heterocyclic synthesis, offering unparalleled efficiency for generating chemical libraries.
Mechanistic Insights into Copper-Catalyzed Interrupted Kinugasa Reaction
The success of this transformation hinges on the precise interplay between the copper catalyst and a specialized chiral ligand. The reaction initiates with the activation of the terminal alkyne by the copper(I) species, forming a copper acetylide. This species then undergoes a cycloaddition with the nitrone to generate a five-membered isoxazoline intermediate, which subsequently rearranges to form the critical four-membered beta-lactam ring fused with a copper enolate. In traditional scenarios, this enolate would be quenched by a proton source. However, in this interrupted variant, the presence of a highly electrophilic third component intercepts the enolate. The chiral environment provided by the indenyl-substituted bisoxazoline ligand ensures that this addition occurs with high stereoselectivity, controlling the configuration of the newly formed stereocenters. This level of control is essential for producing high-purity beta-lactam intermediates required for pharmaceutical applications.
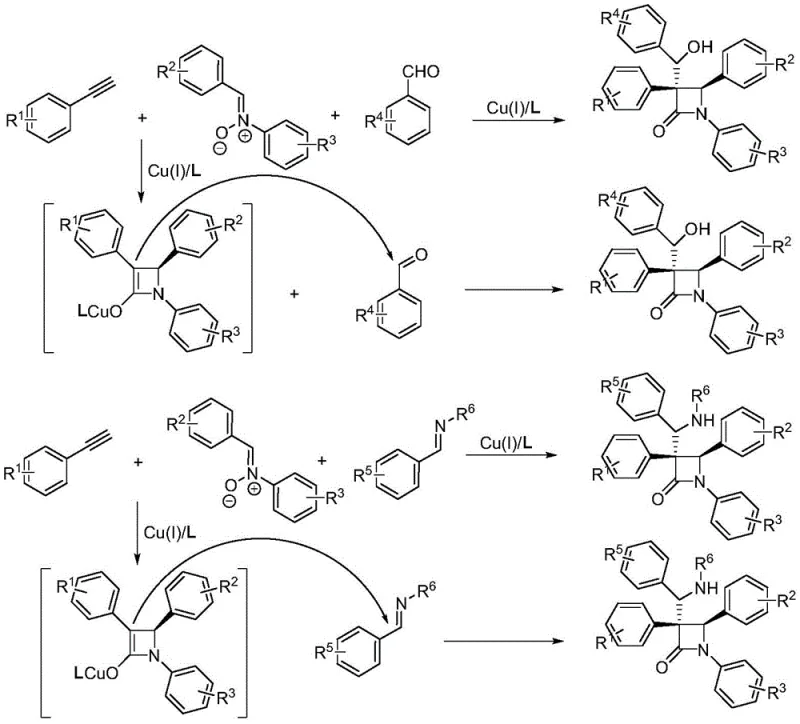
Impurity control is another critical aspect managed by this catalytic system. The use of specific ligands and mild bases like potassium carbonate minimizes side reactions such as polymerization of the alkyne or decomposition of the nitrone. The patent data indicates that the reaction proceeds cleanly to afford the desired diastereomers, which can be separated via standard silica gel chromatography. The high enantiomeric ratios observed in the examples suggest that the catalyst system effectively suppresses the formation of unwanted enantiomers, thereby reducing the burden on downstream purification processes. For R&D teams, this means a more predictable impurity profile and a clearer path to regulatory compliance for the final drug substance.
How to Synthesize Polysubstituted Beta-Lactams Efficiently
The experimental procedures outlined in the patent provide a clear roadmap for executing this synthesis. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent oxidation of the copper catalyst. The stoichiometry is carefully balanced, typically using a slight excess of the nitrone and the third component relative to the phenylacetylene to drive the reaction to completion. The choice of solvent is pivotal, with acetonitrile demonstrating superior performance compared to non-polar alternatives. Following the reaction period, the workup involves simple filtration through celite to remove metal residues, followed by solvent evaporation. The crude product is then purified using column chromatography with ethyl acetate and petroleum ether mixtures. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining phenylacetylene compound, nitrone compound, and a third component (aldehyde, imine, or olefin) in a solvent such as acetonitrile.
- Add a monovalent copper catalyst (e.g., Cu(CH3CN)4PF6), a specific chiral indenyl-substituted bisoxazoline ligand, and a base like potassium carbonate under an inert atmosphere.
- Stir the mixture at temperatures ranging from -20°C to room temperature for 12 to 48 hours, followed by filtration and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits. The primary advantage lies in the accessibility of the starting materials. Phenylacetylenes, nitrones, and aldehydes are commodity chemicals available from multiple global suppliers, ensuring a stable and competitive supply chain. This contrasts sharply with methods requiring bespoke, hard-to-source reagents that can create bottlenecks in production. The simplicity of the one-pot procedure also translates to significant operational efficiencies. By combining multiple bond-forming events into a single reactor charge, manufacturers can drastically reduce the number of unit operations, solvent usage, and labor hours required per kilogram of product. This consolidation of steps is a key driver for cost reduction in API manufacturing, allowing for better margin management in volatile markets.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium significantly lower the raw material costs. Furthermore, the mild reaction conditions reduce the energy load associated with heating or cryogenic cooling, contributing to a lower carbon footprint and reduced utility expenses. The high yields reported in the patent examples indicate efficient atom economy, meaning less waste is generated per unit of product, which lowers waste disposal costs.
- Enhanced Supply Chain Reliability: Because the reaction tolerates a wide range of functional groups on the aromatic rings of the substrates, manufacturers are not locked into a single specific raw material specification. This flexibility allows procurement teams to source alternative grades or suppliers of phenylacetylenes and nitrones without compromising the reaction outcome. Such adaptability is crucial for maintaining continuity of supply in the face of market fluctuations or geopolitical disruptions affecting specific chemical feedstocks.
- Scalability and Environmental Compliance: The protocol avoids the use of hazardous reagents or extreme pressures, making it inherently safer for scale-up to commercial production volumes. The solvent system employed is standard for the industry, facilitating easy recovery and recycling. The ability to produce complex chiral intermediates with high selectivity reduces the need for resource-intensive resolution steps later in the synthesis, aligning with green chemistry principles and increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this technology. They are derived from the specific embodiments and comparative examples found within the patent documentation, providing clarity on the scope and limitations of the method. Understanding these nuances is vital for process chemists evaluating the feasibility of this route for their specific targets.
Q: What distinguishes this interrupted Kinugasa reaction from traditional methods?
A: Traditional Kinugasa reactions typically yield only 3,4-disubstituted beta-lactams via protonation of the copper enolate intermediate. This patented method interrupts that protonation by introducing a third electrophilic component, allowing for the synthesis of highly functionalized polysubstituted beta-lactams in a single pot.
Q: What are the critical reaction conditions for optimal enantioselectivity?
A: The process requires a specific chiral indenyl-substituted bisoxazoline ligand paired with a monovalent copper catalyst. The reaction is best performed in polar aprotic solvents like acetonitrile at mild temperatures (0°C to room temperature) to maintain high stereocontrol.
Q: Can this methodology be adapted for large-scale manufacturing?
A: Yes, the methodology utilizes simple, commercially available starting materials such as phenylacetylenes and nitrones. The one-pot nature of the reaction simplifies processing and reduces waste, making it highly suitable for scaling up to industrial production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Beta-Lactam Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to optimize this interrupted Kinugasa reaction for your specific needs. Our rigorous QC labs and stringent purity specifications ensure that every batch of polysubstituted beta-lactam intermediate meets the highest standards required for pharmaceutical development. We understand the critical nature of antibiotic supply chains and are committed to delivering consistent quality and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can benefit your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and accelerate your project timelines.
