Advanced Copper-Catalyzed Synthesis of Polysubstituted Beta-Lactams for Commercial Scale-Up
Introduction to Novel Beta-Lactam Synthesis Technology
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly beta-lactams, which serve as the core structural motif for a vast array of life-saving antibiotics including penicillins and cephalosporins. Patent CN113801050A, published in late 2021, introduces a groundbreaking advancement in this domain by disclosing a highly efficient preparation method for polysubstituted beta-lactam compounds. This technology leverages a copper-catalyzed interrupted Kinugasa reaction, a sophisticated transformation that overcomes the historical limitations of traditional cycloaddition strategies. By enabling the one-pot串联 (tandem) synthesis of highly functionalized products from simple starting materials, this innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to diversify their antibiotic pipelines. The method not only streamlines the synthetic route but also ensures excellent yield and enantioselectivity, addressing critical quality attributes required for modern drug development.
Furthermore, the versatility of this approach allows for the generation of three distinct classes of polysubstituted beta-lactams (General Formulas I, II, and III) by simply varying the third reaction component. This flexibility is paramount for medicinal chemists exploring structure-activity relationships (SAR) in the search for novel antimicrobial agents to combat rising drug resistance. The technical depth of this patent provides a solid foundation for scaling up production while maintaining rigorous purity standards. As we delve deeper into the mechanistic and commercial implications, it becomes evident that this process offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing, bridging the gap between academic methodology and industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-lactam rings has relied heavily on the classic Kinugasa reaction, first reported in 1972, which involves the copper-catalyzed cycloaddition of terminal alkynes and nitrones. While this reaction is a staple in organic synthesis, it suffers from a significant structural limitation: the reaction pathway invariably proceeds through a copper enolate intermediate that undergoes rapid protonation. Consequently, traditional methods are restricted to producing merely 3,4-disubstituted beta-lactam products. This lack of functional diversity severely constrains the chemical space available for drug discovery, often necessitating multi-step post-functionalization sequences to introduce additional substituents. These extra steps not only increase the overall cost and time of production but also lead to cumulative yield losses and increased waste generation, posing challenges for both economic efficiency and environmental compliance in large-scale operations.
The Novel Approach
In stark contrast, the methodology described in patent CN113801050A revolutionizes this landscape by implementing an "interrupted" Kinugasa reaction strategy. Instead of allowing the copper enolate intermediate to be quenched by a proton source, this novel approach introduces a third electrophilic component—such as benzaldehydes, imines, or activated olefins—to trap the intermediate via nucleophilic addition. This strategic interruption prevents premature protonation and facilitates the formation of highly functionalized polysubstituted beta-lactams in a single operational step. 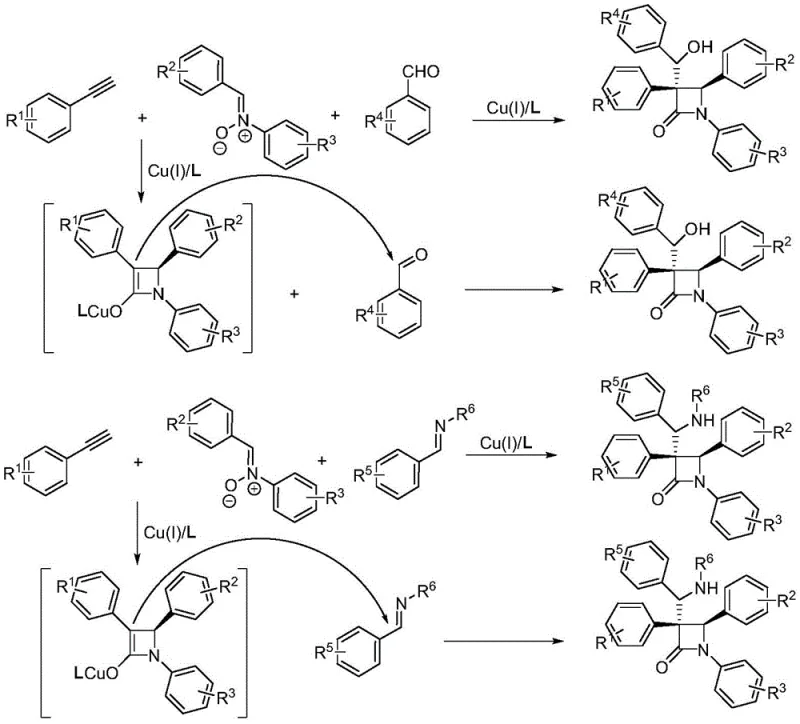 This one-pot tandem process dramatically simplifies the synthetic workflow, eliminating the need for isolation of unstable intermediates and reducing the total number of unit operations. The ability to access complex molecular architectures directly from simple precursors like phenylacetylenes and nitrones underscores the transformative potential of this technology for commercial scale-up of complex pharmaceutical intermediates.
This one-pot tandem process dramatically simplifies the synthetic workflow, eliminating the need for isolation of unstable intermediates and reducing the total number of unit operations. The ability to access complex molecular architectures directly from simple precursors like phenylacetylenes and nitrones underscores the transformative potential of this technology for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Interrupted Kinugasa Reaction
The success of this synthesis hinges on the precise orchestration of a chiral copper catalytic system. The reaction initiates with the activation of the terminal alkyne by a monovalent copper species, followed by cycloaddition with the nitrone to form a key four-membered copper enolate intermediate. In traditional scenarios, this species is fleeting; however, in this patented process, the presence of a specialized chiral ligand stabilizes the transition state and dictates the stereochemical outcome. The ligand of choice is an indenyl-substituted chiral bisoxazoline, a sophisticated structure designed to create a rigid chiral environment around the metal center. 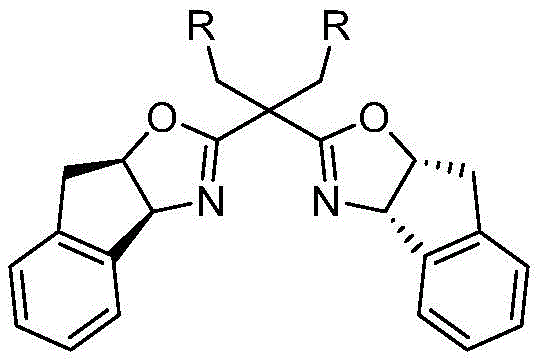 This specific ligand architecture is crucial for achieving the high enantiomeric ratios (often exceeding 95:5 er) reported in the experimental examples, ensuring that the resulting beta-lactams possess the correct optical purity required for biological efficacy.
This specific ligand architecture is crucial for achieving the high enantiomeric ratios (often exceeding 95:5 er) reported in the experimental examples, ensuring that the resulting beta-lactams possess the correct optical purity required for biological efficacy.
Moreover, the mechanism effectively blocks the protonation pathway by introducing a competitive nucleophilic attack from the third component. Whether reacting with an aldehyde to form a hydroxyl-substituted product, an imine to yield an amino-substituted derivative, or an olefin to create an ester-functionalized scaffold, the copper enolate acts as a potent nucleophile. This mechanistic divergence allows for the modular construction of diverse beta-lactam libraries. From a process chemistry perspective, understanding this interruption is vital for impurity control. By optimizing the molar ratios of the third component relative to the alkyne and nitrone, manufacturers can minimize side reactions such as self-dimerization of the alkyne or hydrolysis of the nitrone, thereby ensuring a clean impurity profile that simplifies downstream purification and enhances the overall robustness of the high-purity pharmaceutical intermediate supply chain.
How to Synthesize Polysubstituted Beta-Lactams Efficiently
The practical implementation of this synthesis is remarkably straightforward, utilizing standard laboratory equipment and readily available reagents. The process typically begins by charging a reaction vessel with the phenylacetylene derivative, the nitrone, and the chosen third component (aldehyde, imine, or olefin) in a polar aprotic solvent such as acetonitrile or DMF. A mild inorganic base, preferably potassium carbonate, is added to facilitate the deprotonation steps, while the catalytic system comprising Cu(I) salts and the chiral ligand is introduced under an inert nitrogen or argon atmosphere. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining phenylacetylene compounds, nitrone compounds, and a third component (benzaldehyde, imine, or olefin) in a solvent such as acetonitrile under an inert atmosphere.
- Add a monovalent copper catalyst (e.g., Cu(CH3CN)4PF6) and a specific chiral indenyl-substituted bisoxazoline ligand along with a base like potassium carbonate.
- Stir the mixture at low temperatures (0°C to room temperature) for 12 to 48 hours, then filter and purify the crude product via silica gel column chromatography to isolate the target polysubstituted beta-lactam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this interrupted Kinugasa reaction technology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the simplicity and accessibility of the raw materials. Phenylacetylenes, nitrones, and various aldehydes or imines are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and ensuring supply continuity. Furthermore, the reaction conditions are exceptionally mild, operating effectively between 0°C and room temperature. This eliminates the need for expensive cryogenic cooling systems or high-energy heating protocols, leading to substantial cost savings in utility consumption and reactor maintenance. The use of common solvents like acetonitrile also simplifies solvent recovery and recycling processes, aligning with green chemistry principles and reducing waste disposal costs.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis significantly reduces the operational expenditure associated with multi-step sequences. By consolidating the ring formation and functionalization into a single vessel, manufacturers save on labor, equipment usage time, and intermediate handling costs. Additionally, the high yields and selectivity reported in the patent examples mean less raw material is wasted on byproducts, directly improving the atom economy and lowering the cost of goods sold (COGS) for the final active pharmaceutical ingredient. The elimination of complex protection-deprotection strategies further streamlines the process, making it economically viable for large-volume production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable manufacturing timeline. Unlike sensitive organometallic reactions that require stringent exclusion of moisture or oxygen at extreme temperatures, this copper-catalyzed process tolerates standard inert atmosphere techniques. This reliability reduces the likelihood of batch failures due to environmental fluctuations, ensuring consistent delivery schedules for downstream API producers. The modular nature of the third component also allows for rapid adaptation to market demands; if a specific substituent is required, the feedstock can be swapped without overhauling the entire process infrastructure, enhancing agility in the supply chain.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the use of heterogeneous bases like potassium carbonate, which are easy to filter and handle in large reactors. The absence of toxic heavy metals other than copper, which can be effectively scavenged using standard resins, simplifies the meeting of strict residual metal specifications for pharmaceutical products. Moreover, the high efficiency of the reaction minimizes the generation of hazardous waste streams, supporting corporate sustainability goals and ensuring compliance with increasingly stringent environmental regulations governing chemical manufacturing facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in patent CN113801050A, providing clarity for R&D teams evaluating this technology for integration into their existing portfolios. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What distinguishes this interrupted Kinugasa reaction from traditional methods?
A: Traditional Kinugasa reactions typically yield only 3,4-disubstituted beta-lactams via protonation of the copper enolate intermediate. This patented method introduces a third electrophilic component to intercept the intermediate, enabling the synthesis of highly functionalized polysubstituted beta-lactams in a single pot.
Q: What are the key advantages for large-scale pharmaceutical manufacturing?
A: The process utilizes simple, commercially available starting materials like phenylacetylenes and nitrones. It operates under mild conditions (0°C to room temperature) with common solvents like acetonitrile, significantly simplifying process control and reducing energy consumption compared to cryogenic or high-pressure alternatives.
Q: How does the chiral ligand impact the product quality?
A: The use of a specialized indenyl-substituted chiral bisoxazoline ligand is critical for achieving high enantioselectivity. This ensures the production of optically pure intermediates, which is essential for the biological activity and regulatory compliance of downstream antibiotic APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Beta-Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has thoroughly analyzed the interrupted Kinugasa reaction technology and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. Our state-of-the-art facilities are designed to handle sensitive copper-catalyzed reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the highest international pharmacopoeia standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next-generation antibiotic programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain for efficiency and reliability. Contact us today to discuss how our expertise in polysubstituted beta-lactam synthesis can drive value and innovation in your pharmaceutical projects.
