Innovative Metal-Free Synthesis of 5-Trifluoromethyl Triazoles: Scalable Production for Pharmaceutical Intermediates
Patent CN113121462B introduces a groundbreaking synthetic methodology for producing 5-trifluoromethyl substituted 1,2,3-triazole compounds, representing a significant advancement in the field of heterocyclic chemistry with direct applications in pharmaceutical intermediate manufacturing. This innovative process addresses critical limitations in existing synthetic routes by eliminating hazardous azide chemistry while maintaining high efficiency and selectivity. The technology enables the production of structurally diverse triazole derivatives that serve as essential building blocks for bioactive molecules, particularly those requiring enhanced metabolic stability and lipophilicity characteristics imparted by the trifluoromethyl group. Unlike conventional approaches that rely on copper-catalyzed cycloadditions or organocatalytic methods, this patent describes a metal-free, azide-free synthetic pathway that significantly improves process safety and operational simplicity without compromising product quality or yield. The methodology has been validated through extensive experimental data demonstrating its robustness across various substrate combinations and its potential for industrial implementation in pharmaceutical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethyl-substituted triazoles suffer from multiple critical limitations that hinder their industrial adoption. The first major method involves copper-catalyzed [3+2] cycloaddition between alkynes and organic azides to form triazole metal complexes, followed by subsequent reaction with trifluoromethyl reagents. This two-step process requires handling highly toxic and potentially explosive azide compounds, creating significant safety hazards in manufacturing environments. Additionally, the copper catalyst necessitates complex purification steps to remove trace metal residues that would otherwise compromise the purity specifications required for pharmaceutical applications. The second conventional approach employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly relies on hazardous azide chemistry while often suffering from limited substrate scope and variable regioselectivity. Both methods exhibit poor functional group tolerance, require specialized equipment for safe azide handling, and generate complex waste streams that increase environmental compliance costs. These limitations collectively restrict scalability and increase production costs while introducing unacceptable safety risks in commercial manufacturing settings.
The Novel Approach
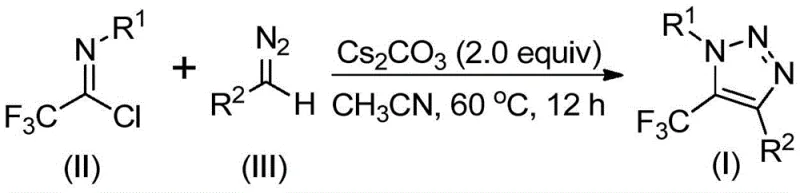
The patented methodology overcomes these fundamental limitations through an elegant base-promoted reaction between readily available diazo compounds and trifluoroethylimidoyl chloride precursors. This innovative process completely eliminates the need for toxic azides, transition metal catalysts, and specialized trifluoromethyl reagents while maintaining high reaction efficiency under mild conditions (60°C). The mechanism proceeds through a base-mediated nucleophilic addition/elimination pathway followed by intramolecular cyclization, avoiding hazardous intermediates while providing excellent regiocontrol. Crucially, the reaction demonstrates broad substrate compatibility across diverse aromatic and aliphatic systems with functional groups including halogens, alkyl chains, and phosphorus-containing moieties. The simplified workup procedure involving filtration and column chromatography significantly reduces processing time compared to conventional methods requiring multiple purification steps to remove metal catalysts. This streamlined approach not only enhances operational safety but also substantially lowers production costs while maintaining the high purity standards essential for pharmaceutical intermediates.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism begins with cesium carbonate-mediated deprotonation of the diazo compound (III), generating a nucleophilic species that attacks the electrophilic carbon of trifluoroethylimidoyl chloride (II). This initial addition forms a key intermediate that undergoes spontaneous intramolecular cyclization through a 5-endo-dig pathway to produce the final triazole product (I). The base plays a dual role in both facilitating the initial nucleophilic attack and promoting the cyclization step while maintaining optimal reaction kinetics at moderate temperatures (50-70°C). The absence of transition metals eliminates potential coordination complexes that could lead to regioisomeric impurities, while the carefully controlled stoichiometry (trifluoroethylimidoyl chloride:diazo compound:cesium carbonate = 1:1.5:2) ensures complete conversion without side reactions. This mechanistic pathway provides exceptional regioselectivity for the desired 1,4-disubstituted triazole isomer without requiring additional directing groups or catalysts.
Impurity control is achieved through multiple built-in mechanisms within this synthetic pathway. The mild reaction conditions prevent thermal decomposition pathways common in high-temperature processes, while the absence of metal catalysts eliminates potential metal-mediated side reactions that could generate difficult-to-remove impurities. The precise stoichiometric control prevents excess reagent accumulation that might lead to byproduct formation, and the straightforward workup procedure efficiently separates any minor impurities through standard chromatographic techniques. This inherent process robustness ensures consistent production of high-purity triazole intermediates meeting pharmaceutical quality standards without requiring specialized purification equipment or additional processing steps that would increase production costs and complexity.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This innovative synthetic route represents a significant advancement in triazole chemistry by providing a safer, more efficient alternative to conventional methods that rely on hazardous materials. The process has been optimized through systematic experimentation to achieve maximum yield and purity while minimizing operational complexity. The following standardized procedure has been developed based on extensive laboratory validation and demonstrates excellent reproducibility across different substrate combinations. Detailed operational parameters have been established to ensure consistent results during scale-up from laboratory to commercial production environments. The subsequent section provides a step-by-step implementation guide for R&D teams looking to adopt this methodology in their synthetic workflows.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, diazo compounds, and molecular sieves in acetonitrile solvent under nitrogen atmosphere
- Maintain reaction temperature at 60°C for 12 hours with continuous stirring to ensure complete conversion
- Perform post-reaction processing through filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate supply chains. The elimination of hazardous materials reduces regulatory compliance burdens while improving workplace safety metrics. The simplified process flow decreases capital investment requirements for specialized equipment typically needed for handling explosive compounds. These advantages collectively enhance supply chain resilience by reducing dependency on complex multi-step processes with multiple failure points while improving overall manufacturing reliability through process intensification.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous azide handling protocols significantly reduces raw material costs while avoiding costly metal removal steps required in conventional processes. The use of readily available starting materials at optimal stoichiometry minimizes waste generation and improves atom economy, leading to substantial cost savings throughout the production cycle without compromising product quality or purity specifications.
- Enhanced Supply Chain Reliability: The simplified reaction pathway using stable, commercially available precursors reduces supply chain vulnerability by eliminating dependencies on specialized hazardous materials with complex handling requirements. The robust nature of the process ensures consistent product quality across different production batches and scales, providing procurement teams with greater confidence in supply continuity while reducing qualification timelines for new manufacturing sites.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedure enable seamless scale-up from laboratory to commercial production without requiring major process re-engineering. The elimination of heavy metal catalysts simplifies waste stream management and reduces environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical intermediate manufacturing. These insights have been developed through comprehensive evaluation of the reaction mechanism, process parameters, and scalability characteristics documented in the patent literature.
Q: How does this method eliminate safety concerns associated with traditional triazole synthesis?
A: The patented process completely avoids toxic and explosive azide compounds required in conventional methods by utilizing stable diazo compounds and trifluoroethylimidoyl chloride as starting materials, significantly enhancing laboratory and plant safety while maintaining high reaction efficiency.
Q: What are the scalability advantages of this metal-free synthesis approach?
A: The process demonstrates excellent scalability from gram-scale laboratory synthesis to industrial production due to its mild reaction conditions (60°C), simple workup procedure, and elimination of transition metal catalysts that require complex removal steps, ensuring consistent product quality across production scales.
Q: How does this method address impurity profile challenges in pharmaceutical intermediate production?
A: The base-promoted mechanism avoids metal contamination pathways and provides precise control over regioselectivity through optimized stoichiometry (1:1.5:2 molar ratio), resulting in cleaner reaction profiles with fewer byproducts that require extensive purification to meet stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our patented methodology represents a significant advancement in triazole chemistry with substantial implications for pharmaceutical intermediate manufacturing. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our technical team has successfully implemented similar base-promoted methodologies across multiple product lines, ensuring consistent delivery of high-quality intermediates that meet exacting pharmaceutical standards through rigorous process validation and quality control protocols.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your supply chain. Contact us today to obtain specific COA data and route feasibility assessments tailored to your manufacturing requirements.
