Revolutionizing Triazole Synthesis: Scalable Metal-Free Process for High-Purity Pharmaceutical Intermediates
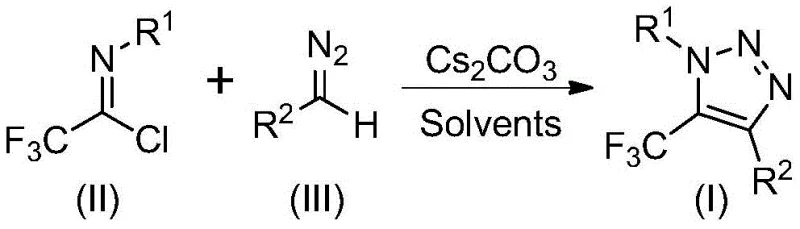
The recently granted Chinese patent CN113121462B introduces a groundbreaking synthetic methodology for producing 5-trifluoromethyl substituted 1,2,3-triazole compounds, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical and agrochemical manufacturing sectors. This innovative process eliminates the need for hazardous azide compounds and transition metal catalysts that have traditionally been required in triazole synthesis, thereby addressing critical safety and environmental concerns while maintaining high reaction efficiency and product purity. The patent demonstrates a scalable approach that can be readily adapted from laboratory-scale to industrial production volumes, offering pharmaceutical companies a more sustainable pathway to access these valuable molecular scaffolds that are increasingly important in drug discovery and development pipelines. By leveraging readily available starting materials and mild reaction conditions, this technology presents a compelling alternative to conventional synthetic routes that often suffer from operational complexities and safety hazards associated with azide handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for preparing trifluoromethyl-substituted 1,2,3-triazoles have primarily relied on two methodologies that present significant operational challenges for industrial-scale manufacturing. The first approach involves copper-catalyzed [3+2] cycloaddition of alkynes with organic azides to form triazole metal complexes, followed by subsequent reaction with various trifluoromethyl reagents; this method requires careful handling of toxic and potentially explosive azide compounds that pose serious safety risks in manufacturing environments. The second conventional method employs organocatalytic 1,3-dipolar cycloaddition reactions between azides and trifluoromethyl ketones, which similarly depends on hazardous azide chemistry and often suffers from limited substrate scope and variable yields. Both established methods necessitate specialized safety protocols for azide handling, additional purification steps to remove metal catalysts, and complex waste treatment procedures for metal-containing byproducts, resulting in increased production costs and extended manufacturing timelines that are incompatible with modern pharmaceutical supply chain requirements for speed and reliability.
The Novel Approach
The patented methodology described in CN113121462B represents a paradigm shift by utilizing readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials in a base-promoted reaction that completely eliminates the need for both metal catalysts and hazardous azide compounds. This innovative process operates under mild conditions (50-70°C) in common organic solvents like acetonitrile, with reaction times ranging from 8 to 16 hours, making it highly amenable to standard manufacturing equipment without requiring specialized safety infrastructure. The method demonstrates exceptional substrate flexibility with various R¹ and R² substituents, enabling the production of diverse triazole derivatives with high functional group tolerance and consistent yields across multiple compound classes. Crucially, the elimination of metal catalysts removes the need for costly metal removal steps and associated quality control testing, while the avoidance of azide chemistry significantly enhances workplace safety and reduces regulatory compliance burdens for pharmaceutical manufacturers seeking to implement this technology in their production facilities.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism proceeds through a base-promoted intermolecular nucleophilic addition and elimination process where the diazo compound acts as a nucleophile toward the trifluoroethylimidoyl chloride, facilitating carbon-carbon bond formation without the need for transition metal catalysis. This initial step generates an intermediate that subsequently undergoes an intramolecular 5-endo-dig cyclization reaction to form the final 5-trifluoromethyl substituted 1,2,3-triazole structure. The use of cesium carbonate as the base promoter is particularly advantageous due to its moderate basicity that effectively drives the reaction while minimizing unwanted side reactions that could compromise product purity. The mechanism avoids the formation of potentially explosive intermediates that are characteristic of traditional azide-based approaches, thereby enhancing both safety and process robustness for industrial implementation.
Impurity control is achieved through the carefully optimized reaction parameters that minimize competing pathways; the mild reaction conditions (60°C) prevent thermal decomposition of sensitive intermediates while the precise stoichiometry (trifluoroethylimidoyl chloride:diazo compound:cesium carbonate = 1:1.5:2) ensures complete conversion of starting materials without excess reagents that could lead to byproduct formation. The solvent system (acetonitrile) provides optimal solubility for all reaction components while facilitating easy product isolation through simple filtration and column chromatography purification. This controlled reaction environment results in consistently high-purity products with minimal impurities that would require additional purification steps, making the process particularly attractive for pharmaceutical applications where stringent purity specifications are mandatory.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This patented synthetic route offers pharmaceutical manufacturers a streamlined pathway to access high-value triazole intermediates with enhanced safety profiles and improved process economics compared to conventional methods. The methodology has been successfully demonstrated at gram scale with consistent yields across diverse substrate combinations, providing strong evidence of its scalability potential for commercial manufacturing applications. The following standardized procedure outlines the key operational parameters that ensure optimal product yield and purity while maintaining process safety and efficiency; detailed implementation guidelines are provided below to facilitate seamless technology transfer from laboratory to production scale.
- Prepare reaction mixture by combining trifluoroethylimidoyl chloride (II), diazo compound (III), cesium carbonate (2.0 equivalents), and molecular sieves in acetonitrile solvent at room temperature
- Heat reaction mixture to 60°C under nitrogen atmosphere and maintain at this temperature for 12 hours with continuous stirring
- Perform post-reaction processing by filtration through silica gel followed by column chromatography purification to obtain high-purity triazole product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach directly addresses critical pain points in the procurement and supply chain management of specialty chemical intermediates by offering a safer, more reliable production pathway that reduces operational risks while maintaining high product quality standards required by pharmaceutical manufacturers. The elimination of hazardous materials and complex purification steps creates significant opportunities for cost optimization without compromising on the stringent quality requirements that govern pharmaceutical supply chains.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated metal removal processes results in substantial cost savings throughout the manufacturing cycle; the use of readily available starting materials at optimal stoichiometry minimizes raw material waste while the simplified purification protocol reduces solvent consumption and processing time without requiring specialized equipment or additional quality control testing for metal residues.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials that are commercially available from multiple global suppliers significantly reduces supply chain vulnerability compared to processes requiring specialized or controlled substances; the robust reaction profile maintains consistent performance across different production scales, ensuring reliable delivery timelines even during periods of market volatility or logistical disruptions.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes without requiring significant process re-engineering; the absence of heavy metal catalysts eliminates complex waste treatment requirements while the use of standard organic solvents facilitates straightforward environmental compliance with global regulatory standards for pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns raised by procurement teams evaluating this novel synthetic methodology for integration into their supply chain; all answers are derived directly from the experimental data and technical specifications documented in patent CN113121462B.
Q: How does this method compare to traditional azide-based triazole synthesis in terms of safety profile?
A: This patented method completely eliminates hazardous azide compounds that are required in conventional approaches, significantly improving workplace safety while removing associated regulatory compliance burdens related to explosive material handling.
Q: What is the scalability potential of this synthetic route for commercial manufacturing?
A: The process has been successfully demonstrated at gram scale with consistent yields across diverse substrates and can be readily scaled to commercial production volumes without requiring specialized equipment or complex process modifications.
Q: How does the elimination of metal catalysts impact product quality specifications?
A: The metal-free nature of this process eliminates the need for extensive metal removal steps and associated quality control testing for residual metals, resulting in higher purity products with simplified regulatory documentation requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds while maintaining stringent purity specifications through our state-of-the-art manufacturing facilities and rigorous QC labs. Our technical team has successfully implemented this patented methodology across multiple client projects, demonstrating consistent ability to deliver high-purity triazole intermediates that meet or exceed pharmaceutical industry standards for critical quality attributes including residual solvents, heavy metals, and organic impurities. As a trusted CDMO partner with deep expertise in specialty chemical synthesis, we provide comprehensive technical support from process development through commercial scale manufacturing to ensure seamless integration of this innovative technology into your supply chain.
For companies seeking to optimize their triazole intermediate supply chain, we invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your manufacturing requirements; we will provide detailed COA data and route feasibility assessments demonstrating how this patented technology can enhance your production efficiency while maintaining the highest quality standards required by global regulatory authorities.
