Advanced Solid-Phase Synthesis of Bortezomib for Commercial Scale Manufacturing
The pharmaceutical landscape for oncology treatments has been profoundly shaped by the introduction of proteasome inhibitors, with Bortezomib standing as a cornerstone therapy for multiple myeloma and mantle cell lymphoma. As demand for this critical active pharmaceutical ingredient (API) continues to surge globally, the efficiency and scalability of its manufacturing process have become paramount concerns for supply chain stability. Patent CN104211758B introduces a transformative approach to Bortezomib production, shifting away from traditional liquid-phase methods toward a robust solid-phase synthesis strategy. This innovation addresses long-standing bottlenecks in purification and yield, offering a pathway to high-purity bortezomib that meets stringent regulatory standards while optimizing production costs. By leveraging Fmoc solid-phase peptide synthesis (SPPS) techniques, this method simplifies the construction of the complex peptide-boronate backbone, ensuring consistent quality for downstream drug formulation.
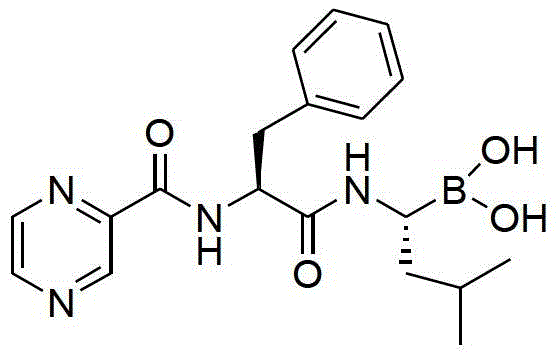
The structural complexity of Bortezomib, characterized by its dipeptide backbone and reactive boronic acid moiety, presents significant synthetic challenges. Traditional routes often struggle with the delicate balance required to install the boron group without degradation, alongside the need for precise stereochemical control. The methodology outlined in CN104211758B utilizes a strategic assembly on a solid support, allowing for the rigorous exclusion of impurities at each coupling step. This approach not only enhances the chemical integrity of the final product but also streamlines the workflow for commercial scale-up of complex pharmaceutical intermediates. For procurement and technical teams, understanding this shift from solution chemistry to solid-phase technology is crucial for evaluating long-term supply reliability and cost structures in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for Bortezomib, such as those disclosed in earlier patents like CN10235189 and CN1168633, predominantly rely on liquid-phase synthesis which entails significant operational complexities. These conventional methods typically involve multiple steps of condensation, debenzylation, and oxidation-deprotection that require harsh conditions, including high-pressure hydrogenation and the use of hazardous reagents. A major drawback of these liquid-phase protocols is the arduous post-treatment process; each reaction step necessitates labor-intensive two-phase extractions and extensive silica gel chromatographic purification to isolate the desired intermediate. These purification stages are not only time-consuming but also result in substantial product loss during transfer and extraction, inevitably depressing the overall yield. Furthermore, the generation of large volumes of organic solvent waste during chromatography poses severe environmental compliance challenges and increases the cost of waste disposal, making these legacy processes less sustainable for modern pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the solid-phase synthesis method described in CN104211758B revolutionizes the production workflow by anchoring the growing molecular chain to an insoluble resin carrier. This fundamental shift allows for the use of excess reagents to drive reactions to completion without complicating the isolation process, as unreacted materials and soluble by-products are simply washed away through filtration. The elimination of intermediate chromatographic purifications represents a paradigm shift in process efficiency, drastically reducing the time required between synthetic steps. By employing a Rink amide resin or similar solid supports, the synthesis proceeds through sequential coupling of protected fragments, maintaining stereochemical integrity throughout the assembly. This streamlined protocol minimizes equipment requirements to standard solid-phase reaction columns and avoids the risks associated with high-pressure hydrogenation, thereby enhancing operational safety. The result is a cleaner, more direct route to the target molecule that significantly mitigates the three-waste pollution issues inherent in liquid-phase chemistry.
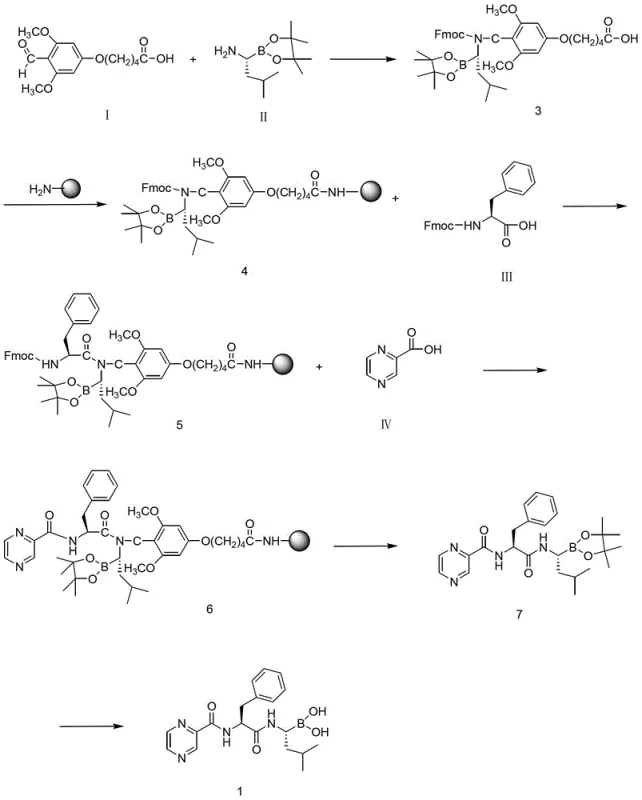
Mechanistic Insights into Fmoc Solid-Phase Coupling Strategy
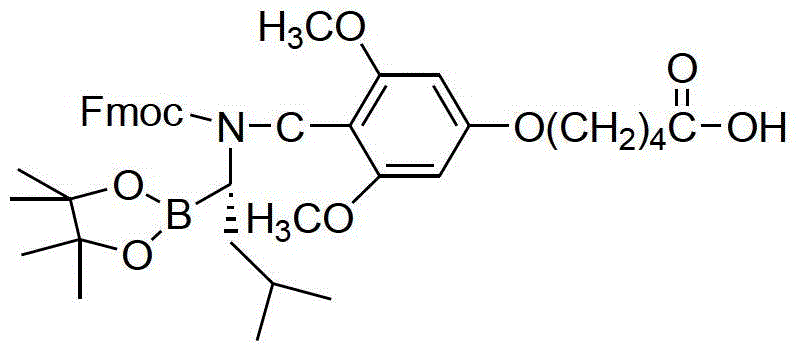
The core of this innovative synthesis lies in the meticulous application of Fmoc (9-fluorenylmethoxycarbonyl) solid-phase peptide synthesis principles to a non-peptidic boronic acid scaffold. The process initiates with the preparation of a specialized building block: an Fmoc-protected amino-boronic acid derivative. This is achieved by condensing pinacol-1-amido-3-methylbutane-1-boric acid ester with 5-(4-formyl-3,5-dimethoxyphenoxy)valeric acid, followed by reduction and Fmoc-protection. This specific intermediate serves as the anchor point on the resin, designed to withstand the subsequent acidic and basic conditions of the synthesis cycle. The choice of the pinacol ester protects the sensitive boronic acid functionality during the coupling phases, preventing premature hydrolysis or protodeboronation which could lead to inactive impurities. The stability of this protecting group is critical for maintaining the potency of the final API, ensuring that the boron atom remains intact until the final deprotection stage.
Once the resin is loaded, the elongation of the molecular chain proceeds through iterative cycles of deprotection and coupling, utilizing highly efficient activation chemistries. The patent highlights the use of advanced coupling reagents such as HATU (2-(7-Azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate) in combination with HOAt and DIPEA. This ternary system generates highly reactive O-acylisourea intermediates that facilitate rapid amide bond formation even on sterically hindered solid supports. The mechanism relies on the nucleophilic attack of the free amine on the resin by the activated carboxyl species, a reaction that is driven to near-quantitative conversion by the use of molar excesses of the soluble coupling partners. Crucially, the solid-phase format allows for real-time monitoring of reaction progress using colorimetric assays like the ninhydrin test, providing immediate feedback on coupling efficiency. This level of process control is difficult to achieve in liquid phase, where tracking reaction completion often requires TLC or HPLC analysis of aliquots, adding further delays to the production timeline.
How to Synthesize Bortezomib Efficiently
The execution of this solid-phase route requires precise control over resin swelling, reagent activation, and cleavage conditions to maximize yield and purity. The process begins with the swelling of the Rink amide resin in DMF to ensure full accessibility of the reactive sites, followed by the coupling of the Fmoc-protected boronic acid intermediate. Subsequent cycles involve the removal of the Fmoc group using piperidine, followed by the coupling of Fmoc-L-phenylalanine and finally pyrazine-2-carboxylic acid. Each step is punctuated by rigorous washing protocols to remove residual reagents, ensuring that the growing chain remains free of deletion sequences or truncated by-products.
- Synthesize the Fmoc-protected boronic acid intermediate via condensation and reduction of pinacol-1-amido-3-methylbutane-1-boric acid ester.
- Couple the intermediate to a Rink amide resin, followed by sequential coupling of Fmoc-L-phenylalanine and pyrazine-2-carboxylic acid.
- Cleave the resin-bound product using TFA and hydrolyze the boronic ester to obtain the final high-purity Bortezomib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid-phase methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic simplification of the purification train; by eliminating the need for repeated silica gel chromatography and multi-step extractions, the process reduces the consumption of expensive solvents and stationary phases. This reduction in material usage directly translates to cost reduction in pharmaceutical intermediates manufacturing, as the overhead associated with solvent recovery and waste treatment is substantially lowered. Furthermore, the ability to use excess reagents without penalty ensures that expensive chiral building blocks are utilized efficiently, minimizing the risk of batch failure due to incomplete reactions. The robustness of the solid-phase approach also means that the process is less sensitive to minor variations in reaction conditions, leading to more consistent batch-to-batch quality and reducing the rate of out-of-specification (OOS) results that can disrupt supply schedules.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes one of the most cost-intensive operations in small molecule synthesis. In liquid-phase synthesis, silica gel columns require large volumes of solvents and significant labor for packing and running, which scales poorly at commercial volumes. By replacing this with simple filtration and washing, the new method drastically cuts down on variable operating costs. Additionally, the mild reaction conditions (room temperature and atmospheric pressure) reduce energy consumption for heating and cooling, further contributing to a leaner cost structure. The avoidance of high-pressure hydrogenation equipment also lowers capital expenditure (CAPEX) requirements for facility upgrades, making the technology accessible for a wider range of manufacturing partners.
- Enhanced Supply Chain Reliability: Solid-phase synthesis is inherently modular and scalable, allowing for flexible production capacities that can be adjusted based on market demand. The use of commercially available resins and standard coupling reagents ensures that raw material sourcing is stable and not dependent on exotic or single-source suppliers. The simplified workflow reduces the total cycle time from raw materials to finished API, enabling faster response times to urgent procurement requests. Moreover, the reduced generation of hazardous waste simplifies regulatory compliance and logistics for waste disposal, removing a common bottleneck in chemical manufacturing that can lead to production stoppages. This operational resilience makes the supply of high-purity bortezomib more predictable and secure for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis route is significantly smaller than traditional methods, aligning with global trends towards green chemistry and sustainable manufacturing. The reduction in solvent waste and the absence of heavy metal catalysts or high-pressure hydrogen gas mitigate safety risks and environmental liabilities. Scaling this process from laboratory to pilot and eventually to commercial tonnage is straightforward, as it primarily involves increasing the size of the reaction columns rather than redesigning the entire process flow. This ease of scale-up ensures that the technology can meet the growing global demand for Bortezomib without compromising on quality or safety standards, providing a future-proof solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for assessing the quality profile of suppliers utilizing this method.
Q: What are the primary advantages of solid-phase synthesis over liquid-phase for Bortezomib?
A: Solid-phase synthesis eliminates complex post-treatment operations such as two-phase extraction and silica gel chromatography. By simply filtering and washing the resin, excess reagents and by-products are removed, significantly reducing three-waste pollution and improving total yield.
Q: Which coupling agents are preferred for this synthesis route?
A: The patent specifies the use of potent coupling systems such as HATU/HOAT/DIPEA or DIPCDI/HOBt combinations. These agents ensure efficient activation of carboxyl groups under mild conditions, driving the coupling reactions to completion on the solid support.
Q: How is the final boronic acid functionality revealed in this process?
A: After cleavage from the resin using TFA to obtain the boronic ester, the final step involves hydrolysis using sodium periodate or potassium periodate in a THF/water system. This converts the pinacol boronic ester into the active free boronic acid required for proteasome inhibition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthesis routes for life-saving oncology drugs like Bortezomib. Our technical team has extensively analyzed the solid-phase methodology described in CN104211758B and possesses the expertise to implement this advanced chemistry at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of API intermediates. Our state-of-the-art facilities are equipped with dedicated solid-phase synthesis reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-quality Bortezomib intermediates that support the uninterrupted manufacturing of finished dosage forms for patients worldwide.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for proteasome inhibitors. By leveraging our technical proficiency in solid-phase chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us partner with you to drive efficiency and reliability in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
