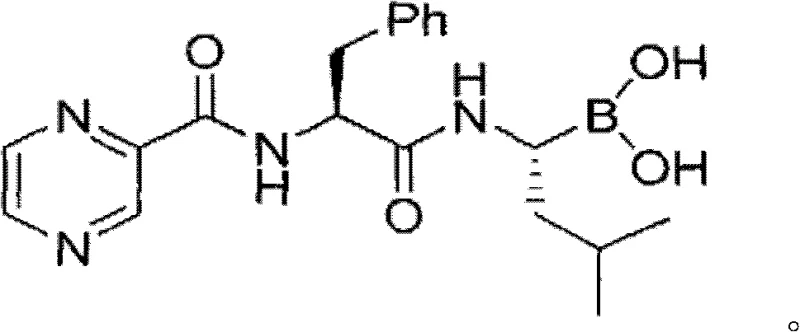
Scalable Synthesis of Bortezomib Intermediates: Overcoming Cryogenic and Purification Bottlenecks
Scalable Synthesis of Bortezomib Intermediates: Overcoming Cryogenic and Purification Bottlenecks
The global demand for high-purity oncology therapeutics continues to drive innovation in process chemistry, particularly for complex proteasome inhibitors like Bortezomib. Patent CN103044467A introduces a transformative methodology for synthesizing the critical intermediate R-(1-amino-3-methyl)butyl pinacol borate, addressing longstanding inefficiencies in supply chain stability and production costs. This technical insight analyzes the patented route, which replaces expensive chiral auxiliaries and cryogenic operations with a robust copper-catalyzed borylation and resolution strategy. By leveraging commodity feedstocks such as 3-methylbutyraldehyde and bis-pinacol diboron, the disclosed process offers a viable pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity for downstream API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Bortezomib intermediates has been plagued by significant operational and economic barriers that hinder efficient industrial production. Prior art routes, such as those disclosed in WO200509789, rely heavily on (1S,2S,3R,5S)-(+)-2,3-pinanediol as a chiral ligand, a reagent that is not only prohibitively expensive but also lacks large-scale domestic manufacturing sources in key regions. Furthermore, these legacy processes mandate extremely harsh reaction conditions, specifically requiring chloromethylene insertion reactions to be conducted at cryogenic temperatures of -80°C, which imposes severe energy burdens and infrastructure limitations on production facilities. Additionally, the reliance on lithium hexamethyldisilazide (LiHMDS) as a base further escalates costs due to its high price and the necessity for strict moisture-free environments, rendering these methods economically unfeasible for high-volume generic drug manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN103044467A presents a paradigm shift by utilizing a copper-catalyzed addition reaction between 3-methylbutyraldehyde and bis-pinacol diboron under mild reflux conditions. This innovative approach completely eliminates the need for cryogenic cooling and expensive chiral boron reagents, substituting them with readily available commodity chemicals that ensure reliable supply chain reliability. The process flow is streamlined to avoid the formation of double bond shifts and subsequent racemization often seen in imine-based condensation routes, thereby securing high optical purity without the need for resource-intensive silica gel column chromatography. By integrating a strategic resolution step using tartaric acid derivatives, the novel route achieves superior stereochemical control while drastically simplifying post-reaction workup procedures, making it ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Borylation and Chiral Resolution
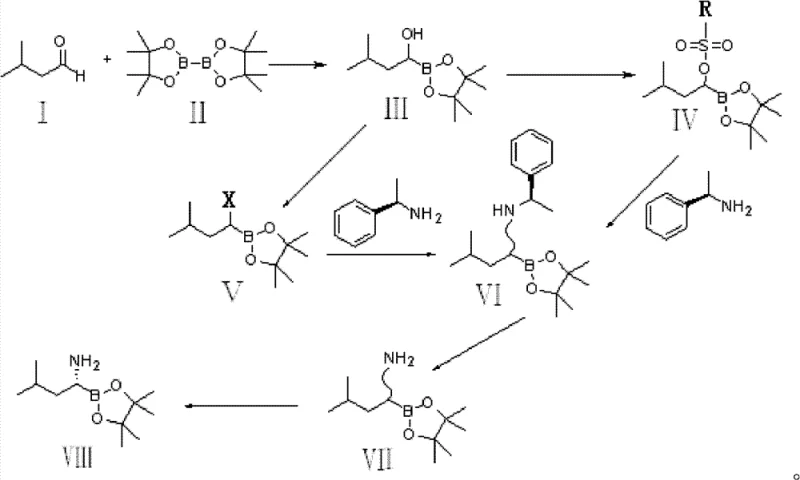
The core of this technological advancement lies in the initial copper-catalyzed borylation step, where 3-methylbutyraldehyde reacts with bis-pinacol diboron in the presence of catalysts such as cuprous chloride or copper acetate. This transformation proceeds through a coordinated mechanism that facilitates the addition of the boron species across the carbonyl group, generating the (1-hydroxy-3-methyl)butyl-pinacol borate intermediate with high molar yields ranging from 88% to 94%. The selection of solvents like benzene or dichloromethane and the optimization of catalyst loading (1:0.05 molar ratio) are critical parameters that ensure the reaction proceeds efficiently at reflux temperatures, avoiding the kinetic traps associated with lower temperature protocols. This robust catalytic cycle not only maximizes atom economy but also minimizes the formation of side products that typically complicate downstream purification efforts in traditional syntheses.
Following the activation of the hydroxyl group via sulfonylation or halogenation, the process employs a nucleophilic substitution with R-(+)-1-phenylethylamine to introduce the nitrogen functionality, followed by a catalytic hydrogenation step to remove the benzyl protecting group. The resulting racemic amine mixture is then subjected to a precise chiral resolution using D-(-)-tartaric acid or D-(+)-dibenzoyl tartaric acid in methanol or ethanol. This resolution strategy is pivotal for impurity control, as it effectively separates the desired R-enantiomer from the S-isomer through differential solubility and crystallization kinetics, achieving chiral purities exceeding 98.5%. The ability to attain such high optical purity through crystallization rather than chromatographic separation is a decisive factor in meeting the stringent purity specifications required for oncology API production.
How to Synthesize R-(1-Amino-3-methyl)butyl Pinacol Borate Efficiently
The synthesis of this critical Bortezomib intermediate is achieved through a sequential five-step protocol that prioritizes operational simplicity and high yield. The process begins with the copper-catalyzed borylation of 3-methylbutyraldehyde, followed by activation of the resulting alcohol, amination, hydrogenolytic deprotection, and final chiral resolution. Each step has been optimized to function under mild conditions, utilizing standard laboratory equipment and commercially available reagents to ensure reproducibility. For detailed standardized synthesis steps and specific reaction parameters, please refer to the guide below.
- Perform copper-catalyzed addition of 3-methylbutyraldehyde with bis-pinacol diboron to form the hydroxy-borate intermediate.
- Activate the hydroxyl group via sulfonylation or halogenation to prepare for nucleophilic substitution.
- Conduct amination with R-(+)-1-phenylethylamine followed by catalytic hydrogenation to remove the benzyl protecting group.
- Resolve the resulting amine mixture using D-(-)-tartaric acid or D-(+)-dibenzoyl tartaric acid to isolate the R-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic advantages by fundamentally altering the cost structure and risk profile of Bortezomib intermediate manufacturing. The transition from specialized, low-availability reagents like pinanediol to commodity aldehydes and boron esters significantly mitigates supply chain volatility, ensuring that production schedules are not disrupted by raw material shortages. Furthermore, the elimination of cryogenic requirements reduces capital expenditure on specialized cooling infrastructure and lowers ongoing energy consumption, contributing to a more sustainable and cost-effective manufacturing footprint. These operational improvements collectively enhance the overall reliability of the supply chain, allowing for more predictable lead times and inventory management for downstream API producers.
- Cost Reduction in Manufacturing: The replacement of expensive chiral ligands and bases with cost-effective alternatives drives significant cost reduction in pharmaceutical intermediate manufacturing. By removing the dependency on LiHMDS and pinanediol, the process eliminates major cost centers associated with reagent procurement and hazardous waste disposal. Additionally, the avoidance of column chromatography reduces solvent consumption and processing time, leading to substantial cost savings in utilities and labor without compromising the quality of the final product.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 3-methylbutyraldehyde ensures enhanced supply chain reliability and reduces the risk of production stoppages. Unlike specialized chiral auxiliaries that may have limited suppliers, these commodity chemicals can be sourced from multiple vendors globally, providing procurement teams with greater negotiating power and supply security. This diversification of the supply base is critical for maintaining continuous production flows in the highly regulated pharmaceutical sector.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures facilitate easy scalability and environmental compliance for industrial production. The absence of extreme temperatures and the reduction in solvent usage align with green chemistry principles, minimizing the environmental impact of the manufacturing process. This scalability ensures that the technology can be seamlessly transferred from pilot plants to multi-ton commercial production facilities, meeting the growing global demand for Bortezomib.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the specific advantages and data provided in the patent documentation. These insights are designed to clarify the operational benefits and quality assurances associated with this novel methodology for stakeholders evaluating technology transfer opportunities.
Q: How does this route improve upon traditional pinanediol-based synthesis?
A: Traditional routes rely on expensive (1S,2S,3R,5S)-(+)-2,3-pinanediol and require harsh cryogenic conditions (-80°C). This patented method utilizes commodity 3-methylbutyraldehyde and operates under mild reflux conditions, significantly reducing raw material costs and energy consumption while avoiding complex low-temperature infrastructure.
Q: Is column chromatography required for purification?
A: No. A critical advantage of this process is the elimination of silica gel column chromatography. The final product achieves high optical purity (up to 99.2%) through crystallization and resolution steps, which is essential for reducing solvent waste and processing time in industrial-scale manufacturing.
Q: What represents the key cost-saving mechanism in this technology?
A: The primary cost drivers removed are the chiral ligand pinanediol and the expensive base LiHMDS. By shifting the chirality introduction to a resolution step using common tartaric acid derivatives and using robust copper catalysis, the process achieves substantial cost reduction in pharmaceutical intermediate manufacturing without compromising optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust process chemistry in delivering high-quality oncology therapeutics to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN103044467A are executed with precision and efficiency. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions and chiral resolutions, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis technology can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume and requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
