Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in recent intellectual property disclosures, specifically patent CN113307778A, a significant technological breakthrough has been achieved in the efficient construction of 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are not merely academic curiosities; they are critical pharmacophores found in blockbuster medications such as Sitagliptin and various antiviral agents, underscoring their immense commercial value. The disclosed methodology leverages a novel dual-catalytic system involving molybdenum and copper to facilitate a direct cycloaddition reaction, offering a robust alternative to traditional synthetic routes that often suffer from harsh conditions or limited substrate scope.
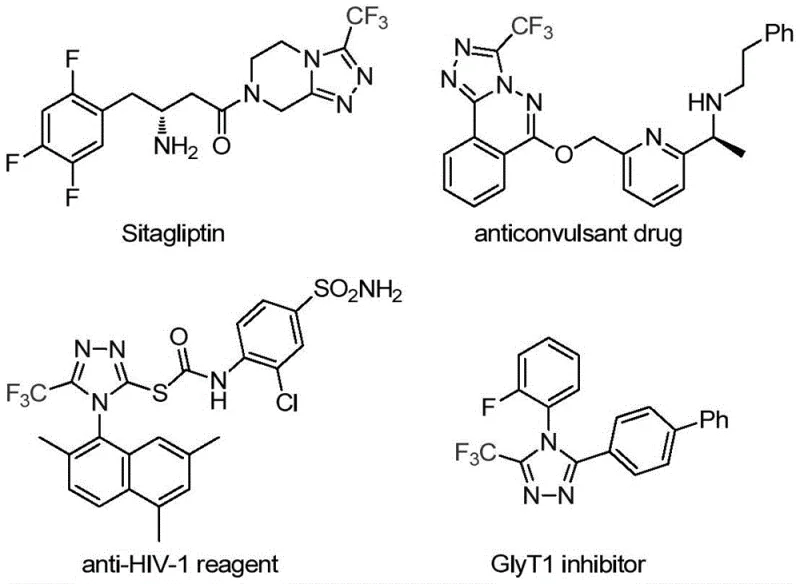
For R&D directors and process chemists, the ability to access these fluorinated heterocycles through a streamlined, one-pot transformation represents a substantial advancement in route design. The patent details a protocol that operates under relatively mild thermal conditions, utilizing readily accessible starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles. This approach not only simplifies the synthetic workflow but also enhances the safety profile of the manufacturing process by avoiding the generation of hazardous intermediates often associated with older diazonium-based methodologies. By establishing a reliable pathway to these high-value intermediates, manufacturers can significantly accelerate the development timeline for new therapeutic entities targeting metabolic disorders and infectious diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant operational challenges and safety concerns that hinder efficient commercial production. Traditional literature methods frequently rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that often require multiple synthetic steps and rigorous purification protocols. Furthermore, alternative strategies involving copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane introduce severe safety hazards due to the explosive nature of diazo compounds, necessitating specialized equipment and stringent safety controls that drive up capital expenditure. These conventional routes also frequently exhibit poor atom economy and limited functional group tolerance, leading to lower overall yields and increased waste generation, which are critical pain points for procurement managers focused on cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent utilizes a molybdenum-copper co-catalytic system to drive a direct [3+2] cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles. This novel approach eliminates the need for unstable diazonium salts and complex multi-step sequences, consolidating the synthesis into a single, operationally simple reaction vessel. The use of inexpensive metal catalysts like cuprous acetate and molybdenum hexacarbonyl, combined with common organic bases like triethylamine, drastically reduces the raw material costs compared to processes relying on precious metals like palladium or rhodium. Moreover, the reaction demonstrates exceptional versatility, accommodating a wide array of substituents on the aromatic ring, including electron-donating and electron-withdrawing groups, thereby providing a universal platform for generating diverse libraries of triazole derivatives for drug discovery programs.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the synergistic interaction between the molybdenum and copper species, which orchestrates the assembly of the five-membered triazole ring with high precision. The reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex that primes the carbon-nitrogen triple bond for nucleophilic attack. Subsequently, the copper catalyst promotes the [3+2] cycloaddition of this activated species onto the trifluoroethylimidoyl chloride, facilitating the formation of a key five-membered ring intermediate. This step is critical as it establishes the core heterocyclic framework while maintaining the integrity of the sensitive trifluoromethyl group, which is essential for the biological activity of the final product.
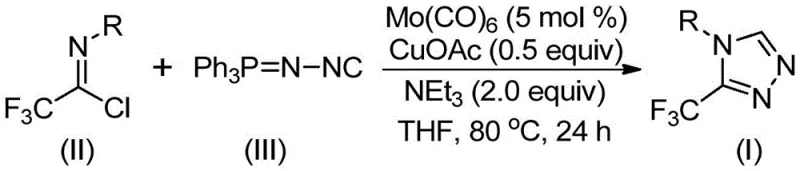
Following the cyclization event, the reaction proceeds through a hydrolysis-mediated elimination of triphenylphosphine oxide, driven by trace water present in the system or added during workup, to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole compound. This mechanistic pathway is particularly advantageous for impurity control, as the byproducts generated, such as triphenylphosphine oxide, are typically solid and easily removed via standard filtration or chromatographic techniques. The robustness of this catalytic cycle ensures consistent product quality and high purity profiles, addressing a primary concern for quality assurance teams who must validate that residual metal levels and organic impurities remain within strict regulatory limits for pharmaceutical ingredients. Understanding this mechanism allows process engineers to fine-tune parameters such as temperature and catalyst loading to maximize throughput while minimizing side reactions.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard laboratory glassware and commonly available reagents that simplify the technology transfer from bench scale to pilot plant operations. The protocol involves charging a reaction vessel with the catalysts, base, and solvent, followed by the sequential addition of the imidoyl chloride and isonitrile substrates under an inert atmosphere to prevent oxidation of the sensitive metal centers. The detailed standardized synthesis steps, including precise stoichiometric ratios and specific workup procedures to ensure maximum recovery, are outlined below for technical reference.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this molybdenum-copper catalyzed route offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing facilities. By shifting away from hazardous diazonium chemistry and precious metal catalysts, companies can mitigate significant safety risks and reduce dependency on volatile supply markets for expensive reagents. The use of commodity chemicals like triethylamine and cuprous acetate ensures a stable and predictable supply chain, reducing the likelihood of production delays caused by raw material shortages. Furthermore, the simplified post-processing requirements, which primarily involve filtration and standard chromatography, streamline the manufacturing workflow, allowing for faster batch turnover times and improved facility utilization rates without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the substitution of costly noble metal catalysts with abundant and inexpensive transition metals like copper and molybdenum. This strategic material selection eliminates the need for expensive metal scavenging resins and complex purification steps required to meet strict residual metal specifications in pharmaceutical products. Additionally, the high reaction efficiency and broad substrate scope minimize the loss of valuable starting materials, leading to superior overall process mass intensity and reduced waste disposal costs. The elimination of hazardous reagents also lowers the overhead associated with specialized safety infrastructure and regulatory compliance, contributing to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as trifluoroethylimidoyl chloride and functionalized isonitriles, ensures a robust supply chain that is less susceptible to geopolitical disruptions or market fluctuations. Unlike processes dependent on custom-synthesized or unstable intermediates that require immediate use, the reagents for this method can be sourced from multiple global suppliers and stored safely for extended periods. This flexibility empowers procurement teams to negotiate better pricing terms and maintain optimal inventory levels, ensuring continuous production schedules and reliable delivery commitments to downstream pharmaceutical clients who depend on just-in-time manufacturing models.
- Scalability and Environmental Compliance: The mild reaction conditions, operating effectively between 70°C and 90°C, make this process highly amenable to scale-up in standard stainless steel reactors without the need for extreme pressure or cryogenic cooling systems. The use of common organic solvents like THF, which can be efficiently recovered and recycled, aligns with green chemistry principles and helps facilities meet increasingly stringent environmental regulations regarding solvent emissions and waste generation. The straightforward workup procedure reduces the volume of aqueous waste streams and simplifies the treatment of effluent, lowering the environmental footprint of the manufacturing process and enhancing the company's sustainability profile in the eyes of stakeholders and regulators.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters, substrate compatibility, and scalability based on the experimental data provided in the patent documentation. These insights address critical considerations for process development and help clarify the operational boundaries of the method.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, using Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, nitro, and naphthyl substituents with yields ranging from moderate to excellent.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states the method can be expanded to gram-level reactions and utilizes cheap, commercially available starting materials, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics, and we are committed to delivering excellence in every batch we produce. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that our 3-trifluoromethyl-1,2,4-triazole derivatives meet the highest standards of quality and consistency required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your development timelines and enhance your competitive advantage in the marketplace.
