Revolutionizing Chiral Indoxazinone Production High-Purity Synthesis for Scalable Pharmaceutical Intermediate Manufacturing
The patent CN112209947A represents a significant advancement in asymmetric synthesis methodology through its disclosure of a novel chiral indoxazinone compound and its highly efficient production process. This innovation directly addresses critical challenges in pharmaceutical intermediate manufacturing by introducing a catalytic system that achieves exceptional stereochemical control under remarkably mild conditions. The methodology leverages commercially accessible starting materials—specifically 2-indolylmethanol and nitrone—to construct complex molecular architectures essential for next-generation anti-tumor therapeutics. Crucially, the process demonstrates robust scalability from laboratory benchtop to industrial production environments while maintaining stringent quality parameters required for pharmaceutical applications. This breakthrough not only expands synthetic chemists' toolbox but also establishes a new benchmark for sustainable manufacturing practices within the fine chemical industry through its atom-economical design and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing chiral indole-fused ring compounds have been plagued by severe operational constraints that compromise both safety and economic viability. These methods typically require extreme reaction conditions including high temperatures exceeding 80°C or cryogenic environments below -40°C, creating significant safety hazards through potential thermal runaway or hazardous reagent handling. The resulting processes suffer from inconsistent enantioselectivity with typical ee values below 75%, necessitating costly chiral separation techniques that dramatically increase production expenses while reducing overall yield. Furthermore, conventional catalytic systems often employ transition metals that introduce challenging purification requirements to meet pharmaceutical regulatory standards for heavy metal residues. The inherent instability of intermediates under harsh conditions also leads to complex impurity profiles that require extensive analytical monitoring and additional processing steps, ultimately rendering these methods unsuitable for commercial-scale manufacturing despite their academic interest.
The Novel Approach
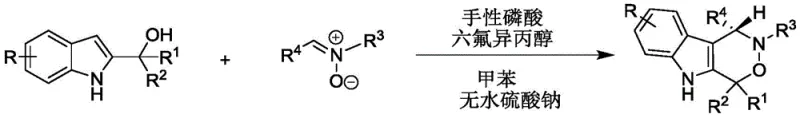
The patented methodology fundamentally reimagines the synthetic pathway through an innovative dual-catalyst system operating under exceptionally mild conditions at ambient temperatures between 20°C and 30°C. By employing chiral phosphoric acid in conjunction with hexafluoroisopropanol as co-catalyst, the process achieves unprecedented stereochemical control with enantiomeric excess consistently exceeding 90% across diverse substrate combinations. This breakthrough eliminates the need for hazardous reagents or extreme temperature regimes while simultaneously improving atom economy through a streamlined one-pot reaction sequence. The use of standard solvents like toluene and simple additives such as anhydrous sodium sulfate ensures compatibility with existing manufacturing infrastructure without requiring specialized equipment modifications. Most significantly, the method demonstrates remarkable versatility across a wide range of functionalized substrates while maintaining high yields and purity levels essential for pharmaceutical applications.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Cyclization
The catalytic cycle operates through a sophisticated dual activation mechanism where chiral phosphoric acid simultaneously activates both reaction partners through hydrogen bonding interactions. The phosphate moiety coordinates with the hydroxyl group of 2-indolylmethanol while the acidic proton engages the nitrone oxygen, creating a well-defined chiral environment that directs stereoselective C-N bond formation. This precise spatial arrangement enables the cyclization step to proceed through a highly ordered transition state that minimizes competing racemic pathways while maximizing enantioselectivity. The hexafluoroisopropanol co-catalyst plays a critical role in stabilizing key intermediates through secondary interactions that further enhance stereochemical control without participating directly in bond formation events. This synergistic catalytic system represents a significant conceptual advance over traditional single-catalyst approaches by creating a cooperative environment where multiple weak interactions collectively achieve superior selectivity outcomes.
Impurity control is inherently engineered into this catalytic system through multiple self-regulating mechanisms that prevent common side reactions observed in conventional syntheses. The mild reaction conditions suppress thermal decomposition pathways that typically generate colored impurities requiring additional purification steps in traditional processes. The precise stereochemical control minimizes diastereomeric byproducts that would otherwise complicate final product isolation while maintaining high chemical purity throughout the reaction sequence. Furthermore, the absence of transition metals eliminates potential contamination sources that would necessitate expensive metal scavenging procedures during downstream processing. This built-in quality assurance mechanism significantly reduces analytical burden during manufacturing while ensuring consistent product quality that meets stringent pharmaceutical specifications without requiring additional quality control interventions.
How to Synthesize Chiral Indoxazinone Efficiently
This patented synthetic route represents a paradigm shift in manufacturing chiral indoxazinone compounds by combining operational simplicity with exceptional stereochemical control. The methodology has been specifically designed for seamless integration into existing pharmaceutical intermediate production facilities through its use of standard equipment and commercially available reagents. Detailed standardized operating procedures have been developed to ensure consistent results across different manufacturing scales while maintaining strict adherence to quality parameters required for pharmaceutical applications. The following section provides essential guidance for implementing this innovative process within your organization's production environment.
- Combine stoichiometric quantities of 2-indolylmethanol and nitrone in toluene solvent with anhydrous sodium sulfate as moisture scavenger under inert atmosphere.
- Introduce chiral phosphoric acid catalyst and hexafluoroisopropanol co-catalyst while maintaining reaction temperature between 20°C and 30°C with continuous stirring.
- Monitor reaction progression via thin-layer chromatography until complete consumption of starting materials is confirmed by visual inspection.
- Execute workup by filtration through celite to remove inorganic salts followed by solvent concentration under reduced pressure prior to purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points inherent in traditional manufacturing approaches for complex chiral intermediates. The process eliminates dependency on specialized equipment or hazardous materials that typically create supply chain vulnerabilities while simultaneously reducing cost drivers associated with complex purification requirements. By operating within standard temperature ranges using common solvents and catalysts, the method significantly enhances manufacturing flexibility and resilience against raw material shortages or price volatility in specialty chemical markets. These operational improvements translate directly into tangible business benefits that strengthen competitive positioning while supporting sustainable growth objectives within pharmaceutical supply networks.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive purification steps required for heavy metal removal while reducing waste treatment costs associated with hazardous byproducts; simplified workup procedures using standard filtration techniques minimize solvent consumption and processing time; consistent high yields across diverse substrates maximize raw material utilization efficiency without requiring costly reprocessing cycles; reduced energy requirements from ambient temperature operation further contribute to operational cost savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Utilization of widely available starting materials from multiple global suppliers mitigates single-source dependency risks; straightforward reaction monitoring via standard TLC eliminates specialized analytical requirements that could cause production delays; compatibility with existing manufacturing infrastructure enables rapid technology transfer without capital equipment investments; robust process performance across varying batch sizes ensures consistent delivery timelines regardless of order volume fluctuations.
- Scalability and Environmental Compliance: The one-pot reaction design minimizes intermediate handling requirements while maintaining high selectivity at commercial scales; simple aqueous workup procedures generate minimal hazardous waste streams compared to traditional methods; elimination of cryogenic or high-temperature operations reduces energy consumption and associated carbon footprint; compatibility with standard purification techniques ensures consistent quality during scale-up from laboratory validation to multi-ton production volumes.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent specifications and experimental data provided in CN112209947A. These responses reflect practical implementation insights derived from thorough examination of the disclosed methodology and its operational parameters.
Q: How does this method overcome traditional limitations in chiral indole-fused ring synthesis?
A: The process eliminates violent reaction conditions through mild temperature control at 20-30°C while achieving high enantioselectivity via dual catalysis with chiral phosphoric acid and hexafluoroisopropanol, directly addressing prior art safety hazards and low yield issues.
Q: What ensures consistent high purity for pharmaceutical applications?
A: The stereoselective catalytic cycle provides precise spatial control during cyclization, yielding products with up to 98% yield and >95% ee without requiring additional purification steps beyond standard silica gel chromatography.
Q: Is this process suitable for commercial scale-up?
A: Yes - the use of readily available reagents like toluene solvent and anhydrous sodium sulfate combined with simple operational procedures enables seamless transition from laboratory to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications. With rigorous QC labs implementing advanced analytical protocols including HPLC, LC-MS, and NMR verification at multiple production stages, we ensure consistent delivery of high-purity chiral indoxazinone compounds meeting all regulatory requirements. Our technical team possesses deep expertise in optimizing catalytic processes like this patented methodology to achieve maximum efficiency while maintaining exceptional product quality standards essential for anti-tumor drug development pipelines.
Leverage our specialized knowledge by requesting a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements; our technical procurement team stands ready to provide detailed COA data and comprehensive route feasibility assessments upon inquiry to support your next-generation therapeutic development initiatives.
