Advanced Biocatalytic Synthesis of Chiral Duloxetine Intermediates for Commercial Scale
The pharmaceutical industry is continuously seeking more sustainable and efficient pathways for the production of chiral intermediates, particularly for high-volume antidepressants like Duloxetine. Patent CN103421854A introduces a groundbreaking biological preparation method for synthesizing (S)-3-(dimethylamino)-1-(thiophene-2-yl)-1-propyl alcohol, a critical chiral building block. This innovation addresses the longstanding challenges associated with traditional chemical reduction and earlier biocatalytic attempts by utilizing a dual-enzyme system comprising ketoreductase (KRED) and glucose dehydrogenase (GDH). By shifting the reaction paradigm from metal-catalyzed hydrogenation to an aqueous enzymatic reduction, this technology offers a compelling value proposition for R&D directors focused on impurity profiles and supply chain leaders concerned with process safety. The method operates under mild conditions, specifically at a temperature range of 25°C to 35°C and a controlled pH of 6.8 to 7.0, ensuring high stereo-selectivity while minimizing energy consumption. This report analyzes the technical merits of this patent and its implications for commercial manufacturing, highlighting how it enables the production of high-purity pharmaceutical intermediates with reduced environmental impact and enhanced operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the chiral alcohol intermediate for Duloxetine has relied heavily on chemical reduction methods utilizing precious metal catalysts such as ruthenium, which necessitate high-pressure hydrogenation equipment and pose significant safety risks in large-scale manufacturing. Alternatively, chemical resolution methods using chiral resolving agents have been employed, but these are inherently inefficient due to a maximum theoretical yield of 50% and the generation of substantial waste streams from the unwanted enantiomer. Even within the realm of biocatalysis, prior art methods disclosed in patents like US2010/0151534A1 often relied on isopropanol as a hydrogen donor, which results in the formation of acetone as a byproduct. The presence of acetone is problematic because it is a volatile, flammable, and toxic organic solvent that requires complex removal steps, often involving negative pressure distillation, to prevent interference with the reaction equilibrium and to meet strict residual solvent guidelines. These conventional approaches create bottlenecks in production capacity, increase the cost of goods sold due to expensive catalysts and solvent recovery, and complicate the regulatory approval process due to the potential for heavy metal or toxic solvent residues in the final active pharmaceutical ingredient.
The Novel Approach
The novel approach detailed in the patent data overcomes these deficiencies by implementing a coupled enzyme system that utilizes glucose as a clean, inexpensive, and safe hydrogen donor instead of isopropanol. In this system, the ketoreductase catalyzes the asymmetric reduction of the ketone substrate, while the glucose dehydrogenase continuously regenerates the necessary cofactor (NADPH) by oxidizing glucose to gluconic acid. This strategic shift eliminates the generation of acetone entirely, thereby removing the need for hazardous solvent removal steps and significantly simplifying the downstream processing workflow. The reaction proceeds in an aqueous buffer system, which not only enhances the stability of the biocatalysts but also aligns with the principles of green chemistry by reducing the reliance on organic solvents during the reaction phase. As illustrated in the reaction scheme below, the transformation is direct and highly selective, converting the ketone substrate directly into the desired (S)-alcohol with exceptional efficiency.
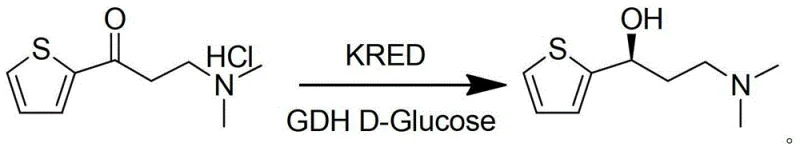
Furthermore, the process demonstrates remarkable robustness, maintaining high conversion rates exceeding 98% even when scaled up, which is a critical factor for commercial viability. The use of glucose as a reductant ensures that the byproduct is gluconic acid, which is water-soluble and easily separated from the organic product during the extraction phase, unlike acetone which can co-distill or form azeotropes. This fundamental change in the reaction chemistry provides a cleaner reaction profile, reduces the environmental footprint of the manufacturing process, and lowers the overall operational complexity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into KRED-GDH Coupled Asymmetric Reduction
The core of this technological advancement lies in the synergistic interaction between ketoreductase (KRED) and glucose dehydrogenase (GDH), which creates a self-sustaining catalytic cycle for cofactor regeneration. In asymmetric biocatalysis, the stoichiometric requirement for expensive cofactors like NADPH is a major cost driver; however, this patent describes a system where the cofactor is recycled in situ. The KRED enzyme facilitates the hydride transfer from the reduced cofactor to the prochiral ketone substrate, specifically targeting the re-face or si-face to ensure the formation of the (S)-enantiomer with high stereochemical fidelity. Simultaneously, the GDH enzyme oxidizes glucose, transferring the electrons to the oxidized cofactor (NADP+) to regenerate NADPH, thus allowing a catalytic amount of cofactor to drive the conversion of a large molar excess of substrate. This mechanism is highly efficient because it decouples the reduction potential from external chemical reductants, relying instead on the thermodynamic drive of glucose oxidation. The patent specifies a weight ratio of ketoreductase to glucose dehydrogenase preferably between 1:1 and 1:3, optimizing the balance between the primary reduction rate and the cofactor regeneration rate to prevent the accumulation of oxidized cofactor which could stall the reaction.
Impurity control is another critical aspect of this mechanism, particularly regarding the maintenance of pH and temperature to prevent non-enzymatic side reactions. The patent emphasizes conducting the reaction at a physiological pH range of 6.8 to 7.0, which is crucial for maintaining the ionization state of the enzyme active sites and the substrate. Deviations from this pH range could lead to enzyme denaturation or the promotion of racemization, which would degrade the optical purity of the product. Additionally, the reaction temperature is tightly controlled between 25°C and 35°C, preferably 28°C to 32°C, to maximize enzyme activity while minimizing thermal degradation. The use of a triethylamine buffer system provides the necessary buffering capacity to counteract the acidity generated by the formation of gluconic acid during the cofactor regeneration cycle. By continuously monitoring and adjusting the pH with sodium carbonate solution, the process ensures a stable reaction environment that supports high conversion rates greater than 98% and optical purity values reaching 99.9% ee. This level of control is essential for R&D directors who need to guarantee that the impurity profile of the intermediate remains within the strict limits required for downstream drug substance synthesis.
How to Synthesize (S)-3-(dimethylamino)-1-(thiophene-2-yl)-1-propyl alcohol Efficiently
The implementation of this biocatalytic route requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and high yield. The process begins with the preparation of the reaction mixture, where the ketone substrate and glucose are dissolved in a pre-prepared buffer solution, followed by the sequential addition of the enzyme cocktail and cofactor. Maintaining the correct stoichiometry of the biocatalysts and the hydrogen donor is vital for driving the reaction to completion without accumulating intermediate byproducts. The reaction is monitored via HPLC to track the conversion of the ketone to the alcohol, ensuring that the endpoint is reached before any potential product degradation occurs. Once the conversion exceeds 98%, the reaction is quenched, and the product is isolated through a series of pH adjustments and solvent extractions that leverage the solubility differences between the product and the aqueous byproducts. For a detailed breakdown of the specific operational parameters, reagent quantities, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction system by dissolving the ketone substrate and glucose hydrogen donor in a triethylamine buffer solution adjusted to pH 7.0.
- Introduce the biocatalyst system comprising ketoreductase (KRED), glucose dehydrogenase (GDH), and NADP cofactor into the reactor at 30°C.
- Maintain pH between 6.8 and 7.0 using sodium carbonate during the 22-24 hour reaction, followed by alkaline workup and toluene extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic method represents a strategic opportunity to optimize the cost structure and reliability of the Duloxetine intermediate supply chain. The elimination of precious metal catalysts such as ruthenium removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth metals. Furthermore, the removal of hazardous organic solvents like isopropanol and the avoidance of acetone byproducts simplifies the waste management process, leading to substantial cost savings in environmental compliance and solvent disposal. The aqueous nature of the reaction also enhances process safety by eliminating the need for high-pressure hydrogenation equipment, thereby reducing capital expenditure requirements for specialized reactor infrastructure and lowering insurance premiums associated with hazardous chemical processing. These factors collectively contribute to a more resilient and cost-effective manufacturing model that can better withstand market volatility and regulatory pressures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive chemical reagents with inexpensive biological catalysts and glucose. By avoiding the use of ruthenium catalysts, the raw material costs are significantly lowered, and the need for complex metal scavenging steps to meet heavy metal specifications is completely eliminated. Additionally, the simplified downstream processing, which does not require energy-intensive distillation to remove acetone, reduces utility costs and increases overall equipment effectiveness. The high conversion rate and optical purity mean that less material is wasted, and the need for costly chiral resolution or recrystallization steps to upgrade purity is minimized, leading to a more efficient use of raw materials and a lower cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on glucose as a hydrogen donor offers a distinct advantage due to its global availability and price stability compared to specialized chemical reductants. The robustness of the enzymatic process under mild conditions reduces the risk of batch failures caused by equipment malfunction or safety incidents, ensuring a more consistent and predictable production schedule. The ability to operate at ambient pressure and temperature also means that the process can be easily transferred between different manufacturing sites without requiring extensive requalification of high-pressure equipment, providing greater flexibility in sourcing and production planning. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who depend on just-in-time delivery models for their own production lines.
- Scalability and Environmental Compliance: The green chemistry profile of this method facilitates easier regulatory approval and scale-up, as the absence of toxic solvents and heavy metals simplifies the environmental impact assessment. The aqueous reaction system is inherently safer to scale, as it avoids the thermal runaway risks associated with exothermic chemical reductions, allowing for larger batch sizes and higher throughput. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, reducing the liability and administrative burden associated with waste disposal. This environmental compliance not only protects the company from regulatory fines but also enhances its corporate social responsibility profile, which is becoming an increasingly important factor for pharmaceutical companies when selecting suppliers for their active ingredient supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this route for commercial production and for assessing the quality standards that can be expected from suppliers utilizing this method. The information covers aspects of reaction efficiency, product quality, and process safety, providing a comprehensive overview of the technology's capabilities.
Q: How does this biocatalytic method improve upon traditional chemical reduction for Duloxetine intermediates?
A: Unlike traditional methods requiring expensive ruthenium catalysts and high-pressure hydrogen, this enzymatic route operates at ambient temperature and pressure, eliminating heavy metal contamination risks and significantly reducing safety hazards associated with hydrogenation.
Q: What are the purity and optical purity specifications achievable with this process?
A: The patented process consistently achieves a chemical purity exceeding 99.4% and an optical purity (ee value) of 99.9%, meeting the stringent requirements for chiral pharmaceutical intermediates without the need for complex chiral resolution steps.
Q: Does this process generate hazardous organic solvent byproducts like acetone?
A: No, by utilizing a glucose dehydrogenase (GDH) cofactor regeneration system with glucose as the hydrogen donor, the process avoids the use of isopropanol, thereby preventing the formation of toxic acetone byproducts common in older biocatalytic methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-DMAA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN103421854A to meet the evolving demands of the global pharmaceutical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which are equipped to verify the high optical purity and chemical integrity of chiral intermediates like (S)-3-(dimethylamino)-1-(thiophene-2-yl)-1-propyl alcohol. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to maintain the precise pH and temperature controls required for sensitive biocatalytic reactions, guaranteeing batch-to-batch reproducibility.
We invite procurement teams and R&D directors to collaborate with us to leverage this technology for your specific project needs. By partnering with us, you can access a Customized Cost-Saving Analysis that evaluates how switching to this enzymatic route can optimize your specific budget and timeline. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Our team is ready to provide the technical support and supply chain security necessary to bring your Duloxetine projects to market faster and more efficiently, ensuring that you have a reliable partner for your high-purity pharmaceutical intermediate needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
