Scalable Lewis Acid Catalyzed Synthesis of Dihydrofuran Derivatives for Pharmaceutical Applications
Introduction to Advanced Dihydrofuran Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for greener, safer, and more cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN110938048A, which discloses a novel method for the efficient construction of dihydrofuran derivatives. This technology leverages a Lewis acid-catalyzed insertion reaction, utilizing sulfur ylides as nucleophilic reagents and ionic liquids as the reaction medium. For R&D directors and process chemists, this represents a significant departure from traditional methodologies that often rely on hazardous reagents and energy-intensive conditions. The ability to form C-C bonds and cyclize complex heterocyclic structures in a single step under mild conditions addresses critical bottlenecks in the synthesis of bioactive molecules. As a leading entity in the fine chemical sector, understanding and adopting such innovations is crucial for maintaining a competitive edge in the supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan and its derivatives has relied on classical transformations such as the Paal-Knorr synthesis or various metal-catalyzed cyclizations involving diazo compounds. While effective in academic settings, these traditional methods present substantial challenges when translated to industrial scale. The use of diazo compounds, for instance, introduces severe safety hazards due to their inherent instability and potential explosiveness, necessitating specialized equipment and rigorous safety protocols that drive up operational costs. Furthermore, many conventional routes require harsh reaction conditions, including high temperatures or strong acidic/basic environments, which can lead to poor functional group tolerance and the formation of complex impurity profiles. The reliance on volatile organic solvents also exacerbates environmental concerns, creating significant waste disposal liabilities and complicating regulatory compliance for modern chemical manufacturing facilities striving for sustainability.
The Novel Approach
In stark contrast, the methodology described in patent CN110938048A offers a transformative solution by employing sulfur ylides as safe and stable carbene precursors. This approach eliminates the risks associated with diazo compounds while maintaining high reactivity for C-C bond formation. The reaction proceeds efficiently at room temperature, drastically reducing energy consumption compared to thermal cyclization methods. By utilizing ionic liquids as the solvent system, the process avoids the use of toxic volatile organic compounds, aligning perfectly with green chemistry initiatives. The versatility of this method is demonstrated by its compatibility with various substrates, allowing for the rapid construction of diverse dihydrofuran scaffolds essential for drug discovery. This shift towards milder, safer, and more environmentally benign conditions marks a significant step forward in the reliable supply of complex pharmaceutical intermediates.
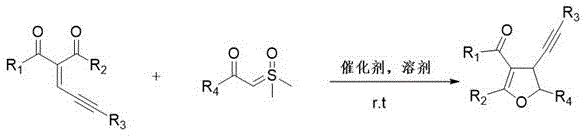
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise activation of the enynone substrate by the Lewis acid catalyst. In this mechanistic pathway, the Lewis acid, such as zinc chloride or copper salts, coordinates with the carbonyl oxygen or the alkyne moiety of the enynone, increasing its electrophilicity. This activation facilitates a Michael-type addition of the sulfur ylide, which acts as a soft nucleophile, to the electron-deficient alkene system. The subsequent intramolecular cyclization is driven by the nucleophilic attack of the resulting enolate or carbanion species onto the activated alkyne or carbonyl center, closing the five-membered furan ring. This concerted or stepwise cascade allows for the efficient formation of the dihydrofuran core with high regioselectivity. The mild nature of the Lewis acid catalysis ensures that sensitive functional groups on the substrate remain intact, thereby preserving the structural integrity required for downstream biological activity.
From an impurity control perspective, the use of room temperature conditions and stable sulfur ylides significantly minimizes the generation of side products. Traditional high-energy pathways often promote decomposition or polymerization of reactive intermediates, leading to difficult-to-remove impurities that compromise the purity of the final API intermediate. In this novel system, the controlled reactivity of the sulfur ylide prevents uncontrolled carbene insertion events that are common with diazo precursors. Furthermore, the ionic liquid solvent creates a unique microenvironment that can stabilize transition states and suppress unwanted side reactions. This results in a cleaner reaction profile, simplifying the purification process and ensuring that the final product meets the stringent purity specifications demanded by global regulatory bodies for pharmaceutical applications.
How to Synthesize Dihydrofuran Derivatives Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it highly attractive for process development teams. The procedure involves the sequential addition of the enynone starting material, the sulfur ylide reagent, the chosen Lewis acid catalyst, and the ionic liquid solvent into a standard reactor. The mixture is then subjected to stirring at ambient temperature, typically overnight, to allow the reaction to reach completion without the need for external heating or cooling. This straightforward protocol reduces the complexity of the manufacturing setup and lowers the barrier for adoption in existing facilities. Following the reaction, the product is isolated through standard purification techniques such as silica gel column chromatography, yielding the target dihydrofuran derivative in high purity. For detailed standardized synthesis steps and specific parameter optimization, please refer to the guide below.
- Charge the reactor with enynone compound, sulfur ylide compound, Lewis acid catalyst (e.g., ZnCl2), and ionic liquid solvent sequentially.
- Stir the reaction mixture at room temperature overnight to ensure complete conversion and cyclization.
- Purify the crude reaction mixture using silica gel column chromatography to isolate the high-purity dihydrofuran product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Lewis acid-catalyzed synthesis offers compelling economic and logistical benefits. The shift away from expensive and hazardous reagents translates directly into reduced raw material costs and lower insurance premiums associated with chemical storage and handling. The elimination of volatile organic solvents not only reduces waste disposal fees but also mitigates the risk of supply chain disruptions caused by tightening environmental regulations on solvent usage. Furthermore, the use of abundant base metal catalysts like zinc or copper, as opposed to scarce noble metals, insulates the production process from the price volatility often seen in the precious metal market. These factors combined create a more resilient and cost-efficient supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of costly noble metal catalysts and hazardous diazo reagents with inexpensive Lewis acids and stable sulfur ylides leads to substantial cost savings. The mild reaction conditions eliminate the need for energy-intensive heating or cryogenic cooling, further driving down utility expenses. Additionally, the simplified workup procedure reduces labor hours and solvent consumption during purification, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including enynones and sulfur ylides, are readily available from multiple global suppliers, reducing the risk of single-source dependency. The stability of these reagents allows for bulk purchasing and long-term storage without significant degradation, enabling strategic inventory management. This reliability ensures consistent production schedules and timely delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The green nature of this process, characterized by the use of recyclable ionic liquids and the absence of toxic VOCs, facilitates easier regulatory approval for new manufacturing sites. The scalability of the reaction from gram to kilogram scale is supported by the exothermic safety profile and the simplicity of the unit operations involved. This alignment with environmental, social, and governance (ESG) goals enhances the corporate reputation and marketability of the supplied chemical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent literature to provide clarity for potential partners. Understanding these aspects is vital for evaluating the feasibility of integrating this route into your existing production portfolio. The answers reflect the consensus on the safety, efficiency, and applicability of the method in an industrial context.
Q: Why are sulfur ylides preferred over diazo compounds in this synthesis?
A: Sulfur ylides offer superior safety and stability profiles compared to diazo compounds, which are often toxic, explosive, and difficult to handle. This substitution significantly reduces operational risks and storage costs in a manufacturing environment.
Q: What are the environmental benefits of using ionic liquids as solvents?
A: Ionic liquids replace volatile and toxic organic solvents, aligning with green chemistry principles. They minimize VOC emissions and simplify waste treatment, thereby reducing the environmental compliance burden for chemical manufacturers.
Q: Is this catalytic method suitable for large-scale production?
A: Yes, the process operates under mild room temperature conditions with readily available Lewis acid catalysts like zinc chloride. The simplicity of the workup and the avoidance of harsh conditions make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrofuran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Lewis acid-catalyzed synthesis of dihydrofuran derivatives for the pharmaceutical industry. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources of complex heterocyclic building blocks.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
