Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Introduction to Advanced Quinazolinone Synthesis
The pharmaceutical industry continuously seeks efficient pathways to access bioactive heterocyclic scaffolds, particularly those containing fluorine motifs which enhance metabolic stability and lipophilicity. Patent CN111675662A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in existing synthetic routes. This innovation leverages a tandem cyclization strategy catalyzed by inexpensive metallic iron, specifically ferric chloride, to construct the quinazolinone core with high precision. By utilizing readily available trifluoroethylimide chloride and isatin as starting materials, this methodology circumvents the need for complex, pre-functionalized synthons that often plague traditional approaches. The significance of this development lies not only in its chemical elegance but also in its potential to streamline the supply chain for reliable pharmaceutical intermediate suppliers who require robust, scalable, and cost-effective manufacturing processes for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on the cyclization of trifluoromethyl-containing synthons with substrates such as anthranilamide, anthranilic acid, or isatoic anhydride. While these methods are chemically feasible, they suffer from severe practical limitations that hinder their industrial applicability. Common trifluoromethylating agents like trifluoroacetic anhydride or ethyl trifluoroacetate often necessitate harsh reaction conditions, including extreme temperatures or strong acidic environments, which can degrade sensitive functional groups on the molecular scaffold. Furthermore, these traditional routes frequently exhibit narrow substrate scope, meaning they fail to accommodate diverse substituents without significant optimization, leading to low overall yields and poor atom economy. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable production timelines, making the sourcing of high-purity intermediates a challenging endeavor.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a tandem cyclization reaction that dramatically simplifies the synthetic landscape. By reacting trifluoroethylimide chloride with isatin in the presence of a base and an iron catalyst, the process achieves direct construction of the target heterocycle with exceptional efficiency. This method boasts excellent functional group tolerance, allowing for the introduction of various substituents such as alkyl, halogen, methoxy, and nitro groups at different positions on the aromatic rings without compromising reaction success. The versatility of this route is exemplified by the successful synthesis of multiple derivatives, demonstrating a broad applicability that is crucial for medicinal chemistry campaigns. 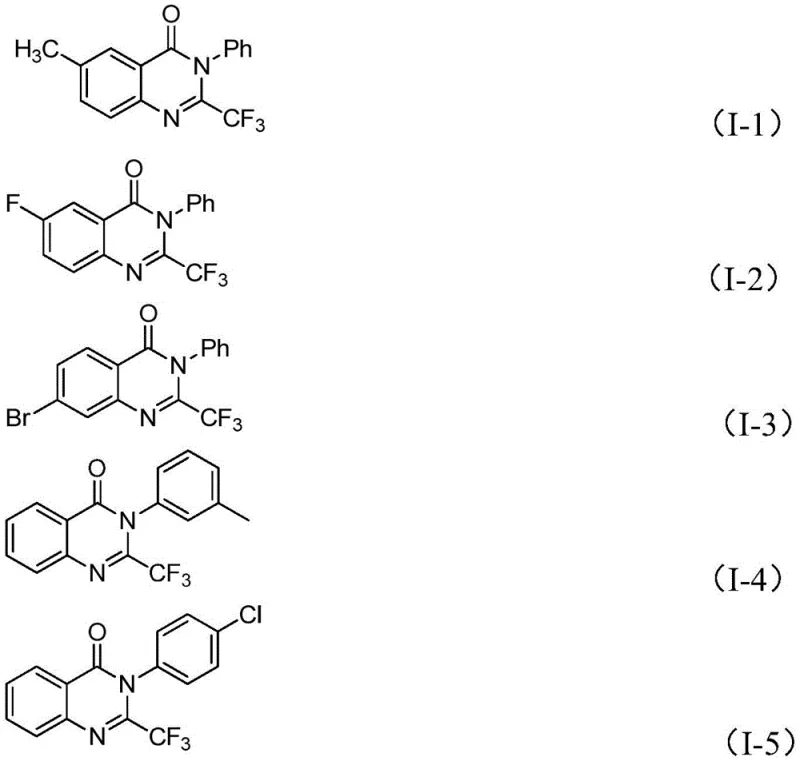 This expanded substrate scope ensures that research and development teams can rapidly generate diverse libraries of compounds for biological screening, accelerating the drug discovery process while maintaining high standards of chemical purity and structural integrity.
This expanded substrate scope ensures that research and development teams can rapidly generate diverse libraries of compounds for biological screening, accelerating the drug discovery process while maintaining high standards of chemical purity and structural integrity.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the mechanistic pathway facilitated by the ferric chloride catalyst. The reaction is proposed to proceed through an initial alkali-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimide chloride and the isatin substrate. This step generates a key trifluoroacetamidine intermediate, which subsequently undergoes an iron-catalyzed decarbonylation cyclization. The iron center likely coordinates with the carbonyl oxygen and the nitrogen atoms, lowering the activation energy for the ring-closing step and facilitating the loss of carbon monoxide. Following cyclization, an isomerization event occurs to yield the thermodynamically stable 2-trifluoromethyl substituted quinazolinone final product. 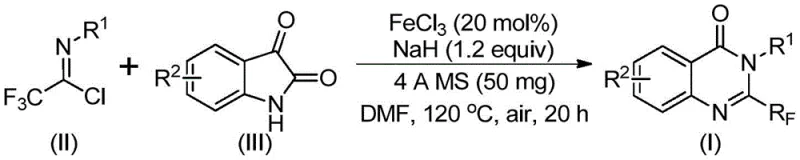 Understanding this mechanism is vital for R&D directors as it highlights the role of the catalyst in driving the reaction forward under relatively mild conditions compared to thermal cyclizations, ensuring that the process remains selective and minimizes the formation of unwanted by-products.
Understanding this mechanism is vital for R&D directors as it highlights the role of the catalyst in driving the reaction forward under relatively mild conditions compared to thermal cyclizations, ensuring that the process remains selective and minimizes the formation of unwanted by-products.
From an impurity control perspective, the use of 4A molecular sieves in the reaction mixture plays a pivotal role in maintaining high product quality. These sieves act as desiccants, scavenging trace amounts of water that could otherwise hydrolyze the sensitive imidoyl chloride starting material or interfere with the sodium hydride base. By maintaining anhydrous conditions throughout the reaction timeline, the process ensures that the primary reaction pathway dominates, significantly reducing the generation of hydrolysis by-products or oligomeric impurities. This level of control is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory specifications. The combination of a robust iron catalyst and effective moisture control creates a reaction environment that is both chemically efficient and operationally reliable, providing a solid foundation for consistent batch-to-batch reproducibility in a commercial manufacturing setting.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The protocol involves mixing the iron catalyst, sodium hydride base, and molecular sieves in a polar aprotic solvent such as DMF, followed by the sequential addition of the substrates. The reaction is typically initiated at a lower temperature of 40°C to allow for the initial coupling, followed by heating to 120°C to drive the cyclization and decarbonylation steps to completion. Detailed standardized synthetic steps for this transformation are provided in the guide below, ensuring that laboratory and production teams can replicate the results with high fidelity. Adhering to these optimized conditions allows for the efficient conversion of starting materials into the desired quinazolinone scaffold, minimizing waste and maximizing throughput for downstream applications.
- Mix ferric chloride catalyst, sodium hydride base, and 4A molecular sieves in an organic solvent such as DMF under inert atmosphere.
- Add trifluoroethylimidoyl chloride and isatin substrates to the reaction mixture and stir at 40°C for approximately 10 hours.
- Increase temperature to 120°C and continue reaction for 20 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this iron-catalyzed methodology represents a strategic opportunity to optimize costs and enhance supply reliability. The replacement of expensive precious metal catalysts with commodity-grade ferric chloride drastically reduces the raw material cost profile of the synthesis. Additionally, the use of widely available starting materials like isatin and easily synthesized imidoyl chlorides mitigates the risk of supply chain disruptions associated with specialized or proprietary reagents. This accessibility ensures a more stable supply of critical intermediates, allowing manufacturers to plan production schedules with greater confidence and reduce lead times for high-purity pharmaceutical intermediates. The operational simplicity of the process further contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing the need for complex equipment or specialized handling procedures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive iron salts lead to substantial cost savings in the overall production budget. By avoiding the need for costly metal scavenging steps required for precious metal residues, the downstream purification process is simplified, further reducing operational expenses. This economic efficiency makes the process highly attractive for large-scale production where margin optimization is critical for competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a consistent supply chain that is less vulnerable to geopolitical or logistical fluctuations. Since the reagents are common chemicals found in the bulk chemical market, sourcing is straightforward and can be diversified across multiple vendors. This redundancy strengthens the supply chain resilience, ensuring that production targets can be met even in volatile market conditions, thereby securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The use of iron, a non-toxic and environmentally benign metal, aligns with green chemistry principles and simplifies waste disposal compliance. This environmental compatibility reduces the regulatory burden associated with heavy metal waste management, facilitating smoother approval processes for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic benefits of adopting this technology for your specific manufacturing needs. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing production portfolios or R&D pipelines.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for this synthesis?
A: The use of ferric chloride (FeCl3) offers a significant cost advantage over precious metal catalysts while maintaining high catalytic efficiency. It eliminates the need for expensive transition metals, simplifying the purification process and reducing heavy metal residue concerns in the final pharmaceutical intermediate.
Q: Can this synthesis method accommodate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully accommodates various substituents including alkyl, halogen, methoxy, and nitro groups on both the aryl ring of the imidoyl chloride and the isatin scaffold, allowing for the synthesis of a wide library of derivatives.
Q: Is this process suitable for large-scale industrial production?
A: The protocol is designed for scalability, utilizing readily available starting materials and standard reaction conditions. The elimination of sensitive reagents and the use of robust iron catalysis make it highly amenable to commercial scale-up from gram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest quality standards required for drug development. We are committed to leveraging our technical expertise to deliver reliable solutions that accelerate your time to market while maintaining cost efficiency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-quality chemical solutions for the global healthcare market.
