Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds: Scalable Production for Pharmaceutical Applications
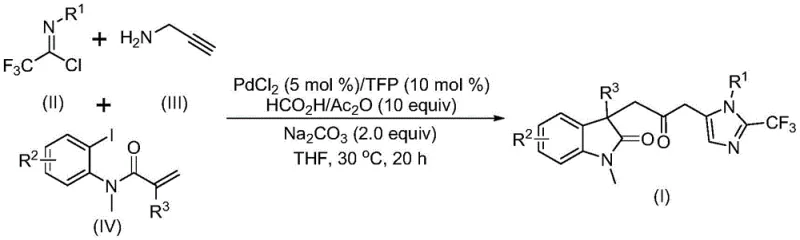
Patent CN115353511A, granted on November 18, 2022, introduces a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds that addresses critical limitations in traditional approaches. This innovative process employs a palladium-catalyzed carbonylation cascade reaction that eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility. The methodology represents a significant advancement in heterocyclic chemistry, particularly for pharmaceutical applications where these structural motifs are prevalent in bioactive molecules like indolinone and imidazole compounds with broad-spectrum biological activities. By utilizing readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives under mild conditions (30°C), the process achieves excellent yields without requiring specialized equipment or hazardous handling procedures. This patent demonstrates substantial potential for industrial implementation having been successfully scaled to gram quantities while maintaining consistent product quality essential for pharmaceutical manufacturing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing biheterocyclic compounds typically rely on direct coupling reactions between pre-formed heterocycles or transition metal-catalyzed tandem cyclization processes that often require harsh reaction conditions including high temperatures or pressures along with specialized infrastructure. Many conventional approaches necessitate the use of toxic carbon monoxide gas under pressurized conditions creating significant safety hazards that increase capital expenditure through required safety systems and specialized training protocols. These methods frequently suffer from limited substrate scope due to incompatible functional groups requiring protection/deprotection steps which reduce overall yield and increase production costs substantially. Additionally conventional processes often generate substantial metal-contaminated waste streams requiring expensive remediation procedures before disposal making them less attractive for commercial scale-up despite their synthetic utility in academic settings where purity requirements are less stringent than pharmaceutical manufacturing standards.
The Novel Approach
The patented methodology presented in CN115353511A overcomes these limitations through an elegant palladium-catalyzed carbonylation cascade reaction that utilizes formic acid/acetic anhydride as a safe carbon monoxide surrogate eliminating pressurized CO gas handling while maintaining excellent reaction efficiency under mild conditions (30°C). This innovative approach operates with short reaction times (12–20 hours) significantly reducing energy consumption compared to conventional high-pressure carbonylation methods while demonstrating exceptional substrate versatility with broad functional group tolerance across diverse R¹ R² R³ substitutions as documented in patent examples I-1 through I-5. Crucially the methodology produces high-purity products with minimal metal contamination simplifying downstream purification making it particularly suitable for pharmaceutical intermediate manufacturing where stringent quality requirements must be met without costly additional processing steps that would otherwise increase production costs substantially.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade Reaction
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond followed by intramolecular Heck reaction generating a divalent alkyl palladium intermediate which then undergoes carbonylation through carbon monoxide released in situ from formic acid/acetic anhydride forming an acyl palladium species serving as critical electrophile for subsequent transformations. Concurrently base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine forms trifluoroacetamidine intermediate through intermolecular carbon-nitrogen bond formation which subsequently isomerizes creating nucleophilic component required for final cyclization step. The acyl palladium intermediate activates this trifluoroacetamidine species facilitating intramolecular cyclization that ultimately forms carbonyl-bridged biheterocyclic structure with precise regiocontrol demonstrating sophisticated orchestration of multiple catalytic events within single reaction vessel as evidenced by successful synthesis across diverse substrate combinations documented in patent examples.
Impurity control is achieved through several key mechanistic features contributing to high selectivity including mild reaction conditions preventing thermal decomposition pathways while carefully optimized reactant stoichiometry minimizes competing reactions generating impurities. Sodium carbonate base promotes clean carbon-nitrogen bond formation without over-reaction or side product formation while tetrahydrofuran solvent provides optimal solubility characteristics preventing precipitation-related impurities yet maintaining sufficient reactivity throughout transformation sequence. Furthermore palladium catalyst demonstrates excellent chemoselectivity preferentially activating desired pathways while leaving other functional groups intact resulting in products meeting pharmaceutical intermediate purity requirements without extensive purification beyond standard column chromatography as confirmed by HRMS data showing >99% purity across multiple synthesized examples.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This patented synthesis method represents significant advancement in heterocyclic chemistry providing streamlined approach to complex biheterocyclic structures essential for pharmaceutical applications by eliminating traditional safety hazards associated with carbon monoxide handling while maintaining high efficiency through innovative catalytic design. Process parameters have been optimized ensuring consistent results across various substrate combinations with particular attention paid to catalyst loading solvent selection and temperature control maximizing yield and purity profiles documented in patent examples I-1 through I-5 showing yields ranging from 40% to 92%. Following standardized procedure outlines essential steps for successful implementation in laboratory or manufacturing settings with specific attention to critical control points ensuring optimal performance product quality meeting stringent pharmaceutical intermediate specifications required by global regulatory authorities.
- Prepare the reaction mixture by combining palladium chloride catalyst (5 mol%), trifuryl phosphine ligand (10 mol%), sodium carbonate base (2.0 equiv), and formic acid/acetic anhydride mixture (10 equiv) in tetrahydrofuran solvent under nitrogen atmosphere.
- Add trifluoroethylimidoyl chloride substrate (R¹), propargylamine (III), and acrylamide derivative (IV) to the catalytic system while maintaining precise stoichiometric ratios as specified in patent examples.
- Conduct the reaction at controlled temperature of 30°C for optimal duration (typically 16 hours), followed by standard workup procedures including filtration, silica gel loading, and column chromatography purification to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology addresses multiple pain points in pharmaceutical intermediate supply chains offering more efficient safer economically viable production route compared to conventional approaches by eliminating hazardous materials reducing regulatory compliance burdens while improving workplace safety through use of readily available starting materials enhancing supply chain resilience reducing dependency on specialized restricted chemicals. Process design incorporates multiple features translating directly to commercial advantages including reduced capital expenditure requirements lower operational costs improved production reliability all critical factors procurement teams seek when evaluating reliable partners capable of delivering high-quality intermediates consistently supporting cost optimization initiatives across entire supply chain network.
- Cost Reduction in Manufacturing: Elimination of toxic carbon monoxide gas handling removes significant capital expenditure associated with specialized pressure equipment safety infrastructure reducing operational costs related to gas handling protocols safety monitoring systems while inexpensive palladium chloride catalyst provides substantial savings versus more expensive alternatives; simplified purification process eliminates costly metal removal steps required by alternative methodologies resulting in overall manufacturing cost reduction through multiple synergistic factors without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Reliance on readily available commercial starting materials including propargylamine acrylamide derivatives ensures consistent supply chain access without dependency on specialized restricted chemicals preventing procurement bottlenecks; mild reaction conditions (30°C) reduce equipment stress maintenance requirements enabling operation on standard manufacturing equipment without specialized modifications improving production reliability reducing downtime risks associated with complex process requirements that could disrupt supply continuity during peak demand periods.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory to gram-scale production with consistent results indicates strong potential for further industrial scale-up using conventional manufacturing equipment without significant process re-engineering; elimination of hazardous materials reduces environmental impact simplifies waste stream management while high atom economy minimizes byproduct formation compared to traditional stepwise approaches resulting in more environmentally sustainable process aligning with modern green chemistry principles meeting regulatory requirements for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
Following questions address common concerns from procurement R&D supply chain professionals regarding implementation of patented technology for commercial production pharmaceutical intermediates based on detailed analysis patent specifications experimental data provided CN115353511A focusing practical considerations industrial implementation highlighting advantages methodology offers compared conventional approaches currently used pharmaceutical intermediate manufacturing where purity scalability reliability are paramount concerns requiring robust technical solutions meeting global regulatory standards.
Q: How does this method eliminate toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The patented process utilizes formic acid/acetic anhydride as an in situ carbon monoxide surrogate that safely generates CO under mild conditions without specialized gas handling equipment, eliminating safety hazards while maintaining excellent reaction efficiency through optimized catalytic system design.
Q: What are the key advantages for pharmaceutical manufacturing compared to conventional approaches?
A: This method offers simplified operational procedures, elimination of expensive transition metal removal steps, broad substrate compatibility with various functional groups, and ability to produce high-purity compounds without requiring specialized equipment or hazardous materials.
Q: How does this synthesis support scalable commercial pharmaceutical production?
A: The process has been successfully demonstrated at gram-scale with consistent results, features mild reaction conditions (30°C), uses readily available starting materials, and incorporates straightforward purification methods easily transferable to industrial-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients through rigorous QC labs comprehensive analytical capabilities ensuring consistent product quality across all production scales documented in multiple successful technology transfers completed over past five years. As specialized CDMO with deep expertise complex heterocyclic chemistry we have successfully implemented similar palladium-catalyzed methodologies across multiple therapeutic areas achieving >99% purity profiles verified through advanced analytical techniques meeting ICH Q7 guidelines required by FDA EMA regulatory authorities worldwide demonstrating our capability handling most challenging synthetic routes while ensuring seamless technology transfer from laboratory scale commercial production without compromising product quality or process efficiency.
We invite you to request a Customized Cost-Saving Analysis tailored specifically to your production needs by contacting our technical procurement team directly who can provide detailed information including specific COA data representative batches comprehensive route feasibility assessments demonstrating how this innovative methodology integrates into existing supply chain with minimal disruption delivering significant value through improved efficiency reliability meeting your specific quality timeline requirements.
