Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113307778A introduces a significant breakthrough in this domain by disclosing a preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are ubiquitous in modern medicinal chemistry, serving as critical scaffolds in blockbuster drugs such as Sitagliptin and various antifungal agents. The ability to efficiently install a trifluoromethyl group at the 3-position of the triazole ring opens new avenues for drug discovery and process optimization. This report analyzes the technical merits of this invention, highlighting its potential to serve as a reliable pharmaceutical intermediate supplier solution for global R&D teams seeking high-purity building blocks.
The strategic value of this technology lies not only in the chemical novelty but also in its practical applicability to complex molecule synthesis. As shown in the following illustration, the 1,2,4-triazole core is a privileged structure found in diverse therapeutic areas ranging from antidiabetics to antiviral reagents. The introduction of the trifluoromethyl group further modulates the electronic properties of the molecule, often leading to improved lipophilicity and binding affinity. By mastering the synthesis of these specific derivatives, manufacturers can offer a competitive edge in the development of next-generation active pharmaceutical ingredients (APIs) and advanced intermediates.
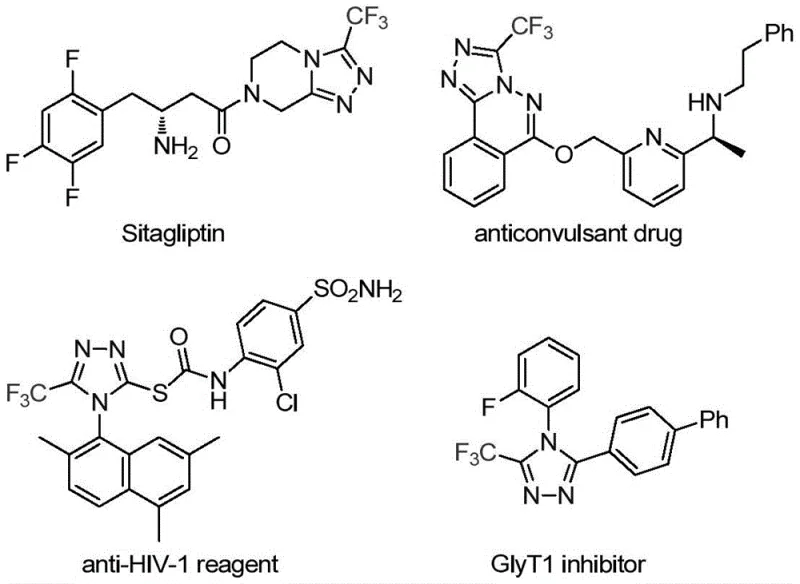
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder efficient commercial production. Traditional literature reports primarily describe cyclization reactions involving trifluoroacetyl hydrazine and amidine compounds, which often require stringent reaction conditions and generate significant amounts of hazardous waste. Another common pathway involves the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone, a process that can be sensitive to moisture and requires careful control of stoichiometry to avoid side reactions. Furthermore, copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose serious safety risks due to the explosive nature of diazo compounds, making them less desirable for large-scale manufacturing environments where safety protocols are paramount.
Beyond safety concerns, the substrate scope of these legacy methods is often narrow, limiting the diversity of derivatives that can be produced without extensive re-optimization. Many existing protocols struggle to tolerate sensitive functional groups, leading to low yields or complex purification burdens that drive up the cost of goods sold. The reliance on expensive or difficult-to-handle reagents further exacerbates the supply chain vulnerabilities for procurement managers who need consistent access to raw materials. Consequently, there is a pressing demand for a safer, more versatile, and operationally simple synthetic route that can deliver high-purity products without compromising on scalability or environmental compliance standards.
The Novel Approach
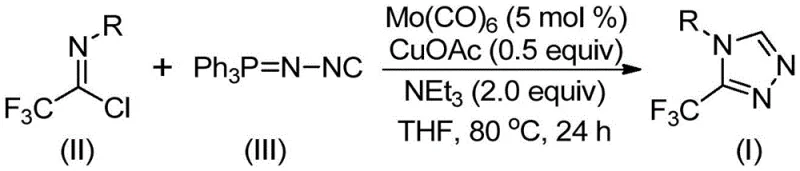
The methodology disclosed in CN113307778A represents a paradigm shift by employing a molybdenum and copper co-catalyzed cycloaddition reaction. This innovative strategy utilizes trifluoroethylimidoyl chloride and functionalized isonitrile as key starting materials, both of which are commercially available and relatively inexpensive compared to specialized diazo reagents. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran (THF) at moderate temperatures ranging from 70°C to 90°C. This mild thermal profile reduces energy consumption and minimizes the risk of thermal runaway, addressing key concerns for supply chain heads focused on plant safety and operational continuity.
Moreover, this novel approach demonstrates exceptional functional group tolerance, allowing for the synthesis of a wide array of 3,4-disubstituted 1,2,4-triazole derivatives that were previously difficult to access. The use of molecular sieves in the reaction mixture helps to scavenge water, driving the equilibrium towards product formation and ensuring high conversion rates. The post-treatment process is remarkably straightforward, involving simple filtration followed by column chromatography, which facilitates rapid isolation of the target compounds. This simplicity translates directly into reduced processing time and lower labor costs, providing a compelling argument for cost reduction in fine chemical manufacturing for prospective partners evaluating this technology for adoption.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cyclization
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to optimize this process for specific substrates. The reaction initiates with the interaction between molybdenum hexacarbonyl and the functionalized isonitrile, forming a reactive metal-isocyanide complex. This activation step is critical as it enhances the nucleophilicity of the isonitrile carbon, preparing it for the subsequent cycloaddition event. Simultaneously, the cuprous acetate acts as a promoter, facilitating the [3+2] cycloaddition between the activated isonitrile species and the trifluoroethylimidoyl chloride. This dual-metal synergy ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 1,2,4-triazole ring over potential isomeric byproducts.
Following the ring closure, the intermediate undergoes a transformation where triphenylphosphine oxide is eliminated, likely assisted by trace water present in the system or generated during the reaction, to yield the final aromatic triazole product. This elimination step restores aromaticity to the heterocyclic ring, providing the thermodynamic driving force for the reaction completion. The presence of triethylamine as a base serves to neutralize the hydrochloric acid byproduct generated from the imidoyl chloride, preventing acid-catalyzed decomposition of the sensitive triazole ring. This detailed mechanistic understanding allows chemists to fine-tune reaction parameters such as catalyst loading and base equivalents to maximize yield and purity for specific commercial batches.
Impurity control is another critical aspect managed by this catalytic system. The high selectivity of the Mo/Cu combination minimizes the formation of oligomeric byproducts or unreacted starting materials that often plague non-catalytic thermal cyclizations. By maintaining strict control over the molar ratios of trifluoroethylimidoyl chloride to functionalized isonitrile, typically around 1:1.5, the process ensures that the limiting reagent is fully consumed while suppressing side reactions. The use of aprotic solvents like THF further enhances the solubility of the polar intermediates, ensuring a homogeneous reaction mixture that promotes consistent heat transfer and mass transport. These factors collectively contribute to a robust impurity profile, simplifying the downstream purification requirements and ensuring the final product meets stringent pharmaceutical specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls as outlined in the patent disclosure. The process begins with the careful weighing of molybdenum hexacarbonyl, cuprous acetate, and triethylamine, which are then introduced into a reaction vessel containing the organic solvent. It is imperative to ensure that the molecular sieves are activated prior to use to maximize their water-scavenging capacity, as moisture control is vital for the stability of the imidoyl chloride reactant. Once the catalysts and base are dispersed, the trifluoroethylimidoyl chloride and functionalized isonitrile are added sequentially to initiate the exothermic complexation phase.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical curiosity. The primary advantage lies in the substantial cost savings achieved through the utilization of earth-abundant metal catalysts. Unlike processes relying on precious metals such as palladium or rhodium, the Mo/Cu system utilizes copper acetate, which is significantly cheaper and widely available in the global market. This shift reduces the raw material cost burden and mitigates the risk of price volatility associated with precious metal markets, ensuring more stable pricing models for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous diazo reagents directly lowers the bill of materials for each production batch. Furthermore, the mild reaction conditions reduce energy consumption required for heating and cooling cycles, contributing to lower utility costs. The simplified workup procedure minimizes the volume of solvents and silica gel required for purification, leading to reduced waste disposal fees. These cumulative efficiencies result in a leaner manufacturing process that enhances overall profit margins without sacrificing product quality or yield performance.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including trifluoroethylimidoyl chloride and various isonitriles, are commodity chemicals with established supply chains. This availability reduces the lead time for raw material procurement and decreases the risk of production stoppages due to material shortages. The robustness of the reaction against minor variations in reagent quality further ensures consistent output, allowing supply chain planners to forecast production volumes with greater accuracy. This reliability is essential for maintaining just-in-time inventory levels and meeting the demanding delivery schedules of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been demonstrated to work effectively from milligram to gram scales with potential for tonnage production. The absence of explosive diazo intermediates simplifies the safety assessment for plant expansion, accelerating the timeline for technology transfer to commercial facilities. Additionally, the use of THF, a solvent with well-established recovery and recycling protocols, supports green chemistry initiatives. The reduced generation of heavy metal waste aligns with increasingly strict environmental regulations, positioning manufacturers as responsible partners in the sustainable development of the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this route for their specific projects. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What are the primary limitations of conventional trifluoromethyl triazole synthesis methods?
A: Traditional methods often rely on harsh cyclization conditions involving trifluoroacetyl hydrazine or require complex multi-component reactions with diazonium salts. These legacy processes frequently suffer from limited substrate tolerance, difficult post-treatment procedures, and challenges in scaling up due to safety concerns with unstable intermediates.
Q: How does the Mo/Cu co-catalytic system improve reaction efficiency?
A: The novel approach utilizes molybdenum hexacarbonyl to activate the functionalized isonitrile while cuprous acetate promotes the [3+2] cycloaddition. This dual-catalyst system operates under mild thermal conditions (70-90°C) and demonstrates high conversion rates with readily available starting materials, significantly simplifying the operational workflow compared to single-metal or metal-free alternatives.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the method can be extended to gram-level reactions and beyond. The use of inexpensive catalysts like cuprous acetate and common solvents like THF, combined with a straightforward workup procedure involving filtration and chromatography, makes the process highly amenable to industrial scale-up for pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex heterocyclic synthesis positions us as a strategic partner capable of meeting the evolving needs of the global pharmaceutical market.
We invite you to collaborate with us to leverage this advanced catalytic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market. Let us help you secure a stable supply of high-purity 3-trifluoromethyl-1,2,4-triazoles while optimizing your overall production costs.
