Scalable Manufacturing of 3-Trifluoromethyl-1,2,4-Triazoles via Iodine-Promoted Cyclization for Global Pharma Supply Chains
Introduction to Advanced Triazole Synthesis Technology
The development of efficient synthetic routes for fluorinated heterocycles remains a critical priority for the global pharmaceutical industry, particularly given the prevalence of the 1,2,4-triazole scaffold in modern therapeutics. As illustrated in the structural diversity of bioactive molecules such as Sitagliptin and various Factor IXa inhibitors, the incorporation of a trifluoromethyl group onto the triazole core significantly enhances metabolic stability and binding affinity. 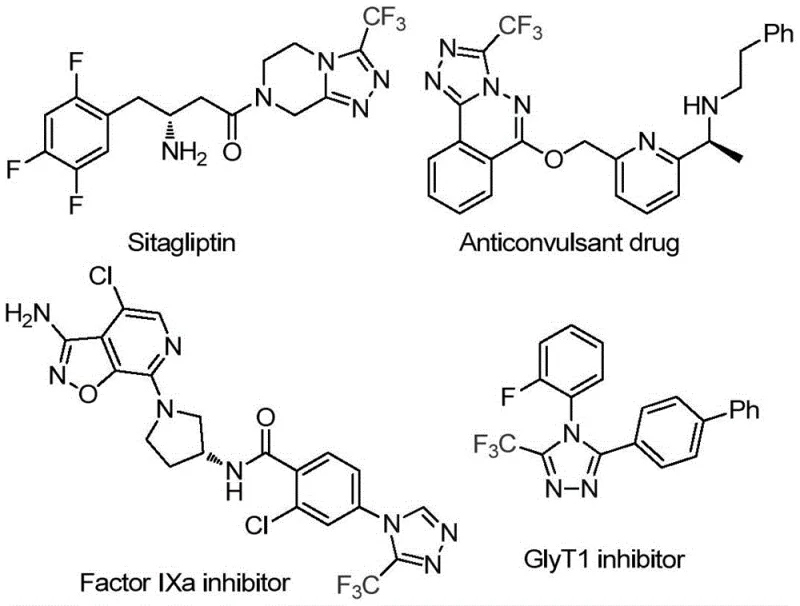 . Addressing the need for more sustainable and operationally simple manufacturing processes, the technology disclosed in patent CN114920707B introduces a groundbreaking approach that utilizes the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source. This innovation represents a paradigm shift in how complex nitrogen-containing heterocycles are constructed, moving away from multi-step sequences involving hazardous reagents toward a streamlined, one-pot tandem cyclization. For R&D directors and process chemists, this method offers a compelling solution to the challenges of scalability and cost-efficiency in API intermediate production.
. Addressing the need for more sustainable and operationally simple manufacturing processes, the technology disclosed in patent CN114920707B introduces a groundbreaking approach that utilizes the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source. This innovation represents a paradigm shift in how complex nitrogen-containing heterocycles are constructed, moving away from multi-step sequences involving hazardous reagents toward a streamlined, one-pot tandem cyclization. For R&D directors and process chemists, this method offers a compelling solution to the challenges of scalability and cost-efficiency in API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing 3-trifluoromethyl-1,2,4-triazoles often suffer from significant operational complexities that hinder large-scale commercial adoption. Conventional routes typically rely on the condensation of hydrazides with specific C1 donors such as formamidine acetates or orthoesters, which frequently necessitate harsh reaction conditions, including high temperatures and strong acidic or basic environments. Furthermore, many existing protocols demand strictly anhydrous and anaerobic conditions to prevent side reactions or decomposition of sensitive intermediates, thereby requiring specialized equipment like gloveboxes or Schlenk lines that increase capital expenditure. The reliance on stoichiometric amounts of expensive coupling reagents or transition metal catalysts also introduces substantial cost burdens and complicates downstream purification due to heavy metal residue concerns. These factors collectively contribute to longer lead times and higher production costs, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel iodine-promoted cyclization described in the patent leverages the inherent reactivity of DMF to drive the formation of the triazole ring with remarkable efficiency. By employing molecular iodine as a mild promoter, the reaction activates the methyl or formyl group of DMF, allowing it to function as a versatile C1 building block that integrates directly into the heterocyclic framework. 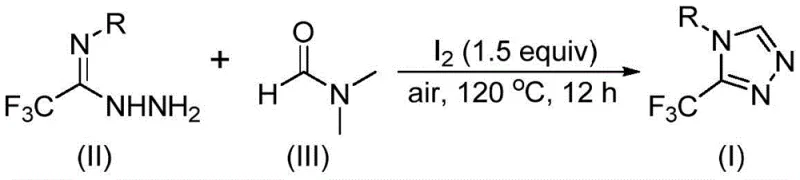 . This approach eliminates the need for external carbon sources, effectively merging the solvent and reactant roles into a single component. The process operates under ambient air pressure and does not require moisture exclusion, drastically simplifying the reactor setup and reducing energy consumption associated with drying solvents. This robustness makes the technology particularly attractive for contract manufacturing organizations seeking to optimize cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of product quality and safety.
. This approach eliminates the need for external carbon sources, effectively merging the solvent and reactant roles into a single component. The process operates under ambient air pressure and does not require moisture exclusion, drastically simplifying the reactor setup and reducing energy consumption associated with drying solvents. This robustness makes the technology particularly attractive for contract manufacturing organizations seeking to optimize cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of product quality and safety.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated example of oxidative cyclization driven by halogen activation. The reaction initiates with the interaction between molecular iodine and DMF, which generates an activated electrophilic species capable of engaging with the trifluoroethyliminohydrazide substrate. Depending on whether the formyl or the N-methyl group of DMF acts as the carbon donor, the mechanism diverges into two plausible pathways, both converging on the desired triazole product. In the formyl pathway, a condensation event forms a hydrazone intermediate, which subsequently undergoes intramolecular cyclization accompanied by the elimination of dimethylamine. Alternatively, when the N-methyl group participates, iodine facilitates the formation of an amine salt followed by nucleophilic addition and elimination of N-methylformamide to generate an azadiene intermediate. This intermediate then undergoes a final intramolecular nucleophilic attack and oxidative aromatization to yield the stable 3-trifluoromethyl-1,2,4-triazole core.  . Understanding these mechanistic nuances is vital for process optimization, as it highlights the critical role of iodine loading and temperature control in driving the equilibrium toward the aromatized product while minimizing the formation of polymeric byproducts.
. Understanding these mechanistic nuances is vital for process optimization, as it highlights the critical role of iodine loading and temperature control in driving the equilibrium toward the aromatized product while minimizing the formation of polymeric byproducts.
From an impurity control perspective, the simplicity of the reagent system inherently limits the complexity of the impurity profile. Since DMF is used in large excess as the solvent, the primary side reactions involve over-iodination or incomplete cyclization, both of which are readily managed through controlled reaction times and standard workup procedures. The absence of transition metals means that the final product is free from difficult-to-remove metal contaminants, a crucial advantage for meeting stringent regulatory specifications for API intermediates. The broad tolerance for functional groups on the aromatic ring, including electron-withdrawing halogens and electron-donating alkyl groups, further underscores the versatility of this catalytic system. This mechanistic robustness ensures that the process can be adapted to synthesize a wide array of analogues required for structure-activity relationship (SAR) studies without necessitating a complete re-optimization of reaction parameters for each new substrate.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis strategy in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the trifluoroethyliminohydrazide precursor and molecular iodine, followed by the addition of DMF which serves as both the reaction medium and the carbon source. The mixture is then heated to a temperature range of 110-130°C and stirred under an open air atmosphere for a duration of 10 to 15 hours. This straightforward procedure minimizes the need for complex inert gas manifolds or specialized drying trains, making it accessible for facilities with standard glassware or stainless steel reactors. The post-reaction workup is equally uncomplicated, typically involving filtration to remove inorganic salts followed by silica gel chromatography to isolate the target compound in high purity.
- Combine molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide substrate in a reaction vessel containing DMF as the solvent.
- Heat the reaction mixture to a temperature range of 110-130°C under an air atmosphere and maintain stirring for 10 to 15 hours to facilitate the tandem cyclization.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the pure 3-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMF-based synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary driver for cost optimization lies in the utilization of DMF, a commodity chemical available in bulk quantities at a fraction of the cost of specialized C1 reagents or organometallic catalysts. By eliminating the need for expensive transition metals such as palladium or copper, the process removes the costly and time-consuming step of heavy metal scavenging, which is often a regulatory requirement for pharmaceutical ingredients. This reduction in processing steps translates directly into lower manufacturing overheads and a reduced environmental footprint, aligning with green chemistry principles that are increasingly mandated by global regulatory bodies.
- Cost Reduction in Manufacturing: The economic impact of replacing proprietary reagents with commodity solvents is profound. Since DMF acts as both solvent and reactant, the material cost per kilogram of product is significantly lowered compared to traditional methods that require stoichiometric amounts of expensive formylating agents. Furthermore, the use of molecular iodine, which is relatively inexpensive and easily recoverable, avoids the financial volatility associated with precious metal catalysts. The simplified workup procedure reduces labor hours and consumable usage, such as filtration media and scavenger resins, contributing to substantial overall cost savings in the production budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available starting materials. Trifluoroethyliminohydrazides can be synthesized from readily accessible aromatic amines and trifluoroacetic acid derivatives, ensuring a stable upstream supply. The robustness of the reaction conditions, which tolerate air and moisture, reduces the risk of batch failures due to environmental fluctuations or operator error. This reliability allows for more accurate production scheduling and shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demands and urgent customer orders without compromising on quality assurance protocols.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of hazardous gases or pyrophoric reagents. The reaction can be safely conducted in standard stirred-tank reactors without the need for high-pressure autoclaves, lowering the barrier for technology transfer from R&D to commercial production. Additionally, the waste stream is simplified, consisting primarily of organic solvents and iodine salts that can be treated or recycled using established industrial protocols. This ease of waste management supports compliance with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and heavy metal discharge, positioning the manufacturer as a responsible and sustainable partner in the global value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of integrating this method into their existing process workflows for the production of complex heterocyclic scaffolds.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive formylating or methylating agents and significantly simplifying the reaction setup.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, reducing equipment costs.
Q: What types of substituents are tolerated on the aromatic ring of the substrate?
A: The method demonstrates broad substrate scope, successfully accommodating various functional groups including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the iodine-promoted DMF cyclization technology allows us to offer competitive pricing and reliable supply continuity for this valuable pharmaceutical intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data for our existing inventory and to receive detailed route feasibility assessments for your custom synthesis projects, ensuring that your supply chain is optimized for both performance and profitability.
