Scalable Metal-Free Synthesis of Trifluoromethyl-Triazoles for Advanced Pharmaceutical Intermediate Manufacturing
Scalable Metal-Free Synthesis of Trifluoromethyl-Triazoles for Advanced Pharmaceutical Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Sitagliptin, Maraviroc, and Deferasirox. This innovation addresses the longstanding challenge of introducing both trifluoromethyl and acyl groups onto the triazole ring simultaneously, a transformation that historically required complex multi-step sequences or harsh conditions. By leveraging a non-metallic iodine promotion system in dimethyl sulfoxide (DMSO), this technology offers a streamlined pathway that aligns perfectly with modern green chemistry principles and cost-effective manufacturing strategies.
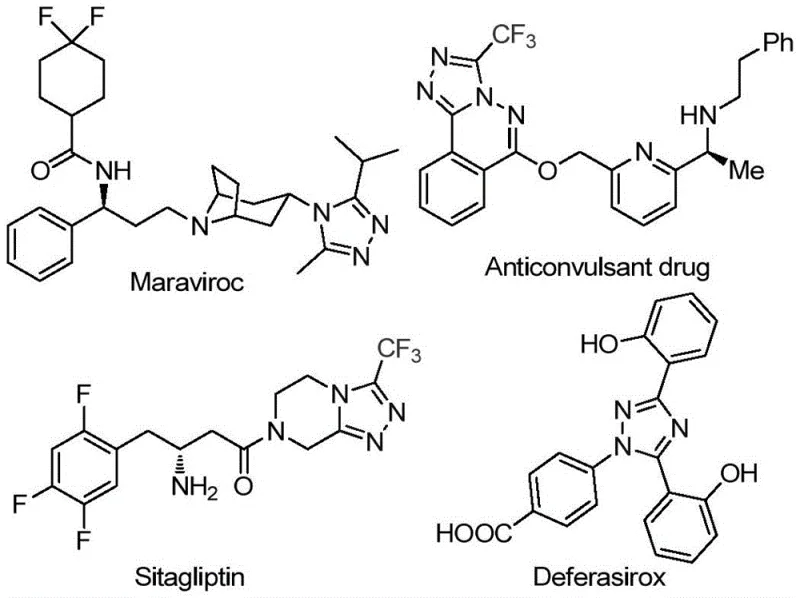
The strategic importance of this scaffold cannot be overstated, as the incorporation of trifluoromethyl groups significantly enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule. For R&D directors and process chemists, the ability to access these diversified cores efficiently translates directly into accelerated lead optimization cycles. Furthermore, the method's reliance on commodity chemicals rather than specialized organometallic catalysts positions it as a highly attractive option for procurement teams focused on supply chain resilience. As we delve deeper into the technical specifics, it becomes evident that this patent represents a significant leap forward in the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of polysubstituted 1,2,4-triazoles has been fraught with significant operational and economic hurdles that impede efficient commercial scale-up. Conventional methodologies often rely heavily on transition metal catalysts, such as copper or palladium complexes, which introduce substantial cost burdens due to the high price of the metals and the requisite ligands. Beyond the direct material costs, the presence of residual heavy metals in the final product necessitates rigorous and expensive purification steps to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). Additionally, many existing protocols demand strictly anhydrous and oxygen-free environments, requiring specialized reactor setups and inert gas handling that increase capital expenditure and operational complexity. These factors collectively contribute to extended lead times and reduced overall process efficiency, creating bottlenecks in the manufacturing of complex heterocyclic intermediates.
The Novel Approach
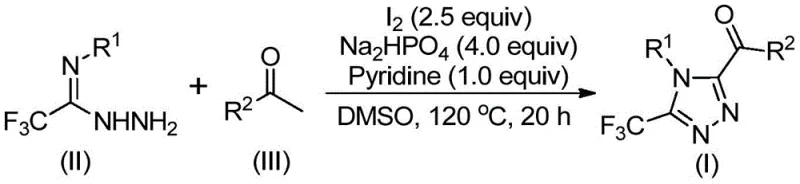
In stark contrast, the methodology outlined in patent CN113105402B introduces a paradigm shift by utilizing elemental iodine as a promoter in conjunction with DMSO, effectively bypassing the need for transition metals entirely. This novel approach capitalizes on the dual functionality of the reaction system, where DMSO acts not merely as a solvent but also participates in the oxidative transformation of aryl ethyl ketones via a Kornblum-type oxidation mechanism. The process operates under relatively mild thermal conditions, typically ranging from 90°C to 130°C, and critically, does not require exclusion of air or moisture. This tolerance to ambient conditions dramatically simplifies the operational workflow, allowing for the use of standard glassware or reactor vessels without the need for Schlenk lines or gloveboxes. The result is a robust, user-friendly protocol that maintains high substrate tolerance while delivering the desired 3,4,5-trisubstituted products with excellent efficiency.
Mechanistic Insights into Iodine-Promoted Cyclization and Oxidation
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions driven by the unique properties of the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes an iodination followed by a Kornblum oxidation mediated by DMSO to generate an aryl diketone intermediate in situ. This reactive species then engages in a dehydration condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine serves to buffer the reaction medium and facilitate the subsequent intramolecular cyclization. Under the continued influence of iodine and heat, the hydrazone undergoes oxidative cyclization to close the triazole ring, ultimately yielding the target 3,4,5-trisubstituted 1,2,4-triazole compound. This cascade sequence elegantly constructs the heterocyclic core while installing the crucial trifluoromethyl and acyl substituents in a single pot.
From an impurity control perspective, this mechanism offers distinct advantages regarding the purity profile of the final product. The absence of transition metals eliminates the risk of metal-induced side reactions or difficult-to-remove metal-ligand complexes that often plague conventional syntheses. Furthermore, the use of stoichiometric iodine and inorganic bases like sodium dihydrogen phosphate generates byproducts that are generally water-soluble or easily separable during the workup phase. The reaction demonstrates broad functional group tolerance, accommodating various substituents on the aryl rings such as methyl, methoxy, chloro, and trifluoromethyl groups without significant degradation or side-product formation. This high level of chemoselectivity ensures that the crude reaction mixture is cleaner, thereby reducing the burden on downstream purification processes like column chromatography and enhancing the overall yield of high-purity material suitable for pharmaceutical applications.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for maximum operational simplicity, making it accessible for both laboratory-scale discovery and pilot-plant production. The protocol involves a sequential addition strategy where the aryl ethyl ketone and iodine are first heated in DMSO to initiate the oxidation phase, followed by the introduction of the hydrazide and base components to drive the cyclization. This two-stage temperature profile, starting at 90-110°C and ramping to 110-130°C, optimizes the kinetics of each step to ensure complete conversion. Detailed standardized operating procedures regarding specific molar ratios, solvent volumes, and precise temperature ramps are critical for reproducibility and are essential for any technical team looking to implement this route.
- Mix aryl ethyl ketone and elemental iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route presents a compelling value proposition centered on cost optimization and supply security. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like palladium can fluctuate wildly based on geopolitical factors. Moreover, the reliance on commodity feedstocks such as acetophenones and elemental iodine ensures a stable and diverse supplier base, mitigating the risk of single-source dependency that often plagues specialized reagent supply chains. The simplified workup procedure, which avoids complex metal scavenging steps, further contributes to significant cost reduction in API manufacturing by shortening cycle times and reducing solvent consumption.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of expensive catalytic systems with inexpensive stoichiometric reagents. By removing the requirement for transition metals, manufacturers avoid the substantial costs associated with metal recovery, waste disposal, and analytical testing for residual metals. The use of DMSO, a low-cost polar aprotic solvent, further drives down raw material expenses compared to specialized organic solvents. Additionally, the high atom economy of the tandem reaction minimizes waste generation, leading to lower environmental compliance costs and more efficient use of raw materials throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials. Aryl ethyl ketones and iodine are produced on a massive industrial scale for various applications, ensuring consistent availability and competitive pricing. This contrasts sharply with custom-synthesized ligands or sensitive organometallic catalysts that may have long lead times and limited production capacity. The robustness of the reaction conditions, which do not require inert atmospheres, also reduces the logistical burden on facility operations, allowing for more flexible scheduling and faster turnaround times from order to delivery for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration at the gram level with straightforward potential for kilogram and tonne-scale expansion. The absence of toxic heavy metals simplifies the environmental, health, and safety (EHS) profile of the process, facilitating easier permitting and waste management. Waste streams are primarily composed of organic residues and inorganic salts, which are more manageable than hazardous metal-contaminated waste. This alignment with green chemistry principles not only supports corporate sustainability goals but also future-proofs the manufacturing process against increasingly stringent global environmental regulations regarding heavy metal discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and strategic advantages of the method. Understanding these details is crucial for stakeholders evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the key advantages of this iodine-promoted method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, significantly simplifying downstream purification and reducing environmental compliance costs while maintaining high yields.
Q: Can this synthesis protocol be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is easily expandable to gram levels and uses cheap, commercially available raw materials like aryl ethyl ketones, making it highly suitable for commercial scale-up.
Q: Does the reaction require strict anhydrous or oxygen-free conditions?
A: No, one of the primary operational benefits is that the reaction does not require anhydrous or oxygen-free conditions, which drastically reduces equipment complexity and operational overhead.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this iodine-promoted process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering consistent quality and reliability for complex heterocyclic building blocks.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data for our triazole intermediates and to discuss detailed route feasibility assessments that can optimize your supply chain and reduce your overall manufacturing costs.
