Advanced Metal-Free Synthesis of Trifluoromethylated Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of Trifluoromethylated Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds that serve as the backbone of modern therapeutics. A pivotal advancement in this domain is detailed in patent CN113105402B, which discloses a highly efficient preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These nitrogen-containing five-membered heterocycles are not merely academic curiosities; they are critical structural motifs found in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, where the introduction of a trifluoromethyl group significantly enhances metabolic stability, lipophilicity, and bioavailability. The innovation presented in this patent addresses the longstanding challenges associated with synthesizing these specific trifluoromethylated and acyl-substituted triazoles, offering a pathway that bypasses the limitations of traditional transition-metal catalysis.
For R&D directors and process chemists, the significance of this technology lies in its operational simplicity and its ability to deliver high-purity intermediates without the burden of removing toxic metal residues. The method leverages a tandem sequence involving iodine-promoted oxidation and cyclization, utilizing dimethyl sulfoxide (DMSO) as both solvent and oxidant mediator. This approach represents a paradigm shift from conventional routes that often demand stringent anhydrous conditions, expensive ligands, or hazardous reagents. By enabling the direct transformation of readily available aryl ethyl ketones and trifluoroethylimide hydrazides into valuable triazole cores, this patent provides a strategic advantage for companies aiming to streamline their supply chains for active pharmaceutical ingredients (APIs) and advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, particularly those bearing both trifluoromethyl and acyl functionalities, has been fraught with synthetic inefficiencies. Traditional methodologies frequently rely on the use of heavy metal catalysts, such as copper or palladium complexes, which introduce significant downstream processing burdens. The removal of trace metal residues to meet the stringent regulatory standards required for pharmaceutical applications often necessitates additional purification steps, such as scavenging treatments or repeated recrystallizations, which inevitably erode overall yield and increase production costs. Furthermore, many existing protocols require strictly anhydrous and oxygen-free environments, demanding specialized equipment like gloveboxes or Schlenk lines, which complicates scale-up and increases capital expenditure for manufacturing facilities.
Another critical drawback of prior art methods is the limited substrate scope and poor functional group tolerance. Harsh reaction conditions often lead to the decomposition of sensitive functional groups or the formation of complex impurity profiles that are difficult to separate. The reliance on exotic or unstable starting materials further exacerbates supply chain vulnerabilities, making it difficult for procurement teams to secure consistent quality raw materials at competitive prices. Consequently, the industry has faced a bottleneck in accessing diverse libraries of trifluoromethylated triazoles for drug discovery programs, slowing down the development of next-generation therapeutics that rely on these privileged scaffolds for their pharmacological activity.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the methodology outlined in patent CN113105402B introduces a streamlined, metal-free strategy that utilizes elemental iodine as a benign and inexpensive promoter. This novel approach capitalizes on the dual role of iodine and DMSO to facilitate a Kornblum-type oxidation of aryl ethyl ketones, generating reactive aryl diketone intermediates in situ. These intermediates then undergo a seamless condensation and cyclization with trifluoroethylimide hydrazides to forge the target 1,2,4-triazole ring system. The elegance of this one-pot transformation lies in its ability to merge multiple bond-forming events into a single operational sequence, drastically reducing solvent consumption and waste generation while maximizing atom economy.
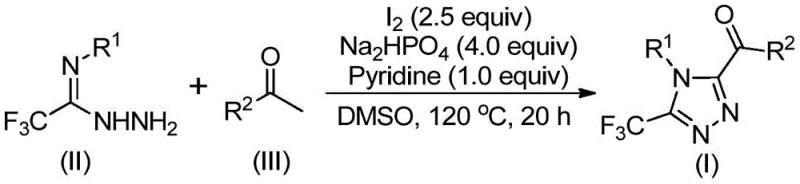
The practical implications of this new route are profound for industrial application. The reaction proceeds efficiently at moderate temperatures ranging from 90°C to 130°C and does not require inert atmosphere protection, allowing it to be performed in standard glass-lined reactors commonly found in multipurpose chemical plants. The use of cheap and commercially available starting materials, such as substituted acetophenones and simple hydrazides, ensures a stable and cost-effective supply chain. Moreover, the method exhibits remarkable versatility, accommodating a wide array of substituents on both the N1 and C5 positions, including electron-donating and electron-withdrawing groups, as well as heteroaryl moieties. This broad scope empowers medicinal chemists to rapidly explore structure-activity relationships (SAR) without being constrained by synthetic feasibility.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
To fully appreciate the robustness of this synthesis, one must delve into the mechanistic intricacies that drive the transformation. The reaction initiates with the iodination of the aryl ethyl ketone, activated by the polar aprotic solvent DMSO. This step effectively converts the methyl ketone into an alpha-iodo ketone or directly to an alpha-dicarbonyl species via a Kornblum oxidation mechanism. The presence of iodine acts as a Lewis acid and an oxidant, facilitating the activation of the C-H bond adjacent to the carbonyl group. This activation is crucial as it renders the substrate susceptible to nucleophilic attack by the hydrazide nitrogen. The subsequent dehydration condensation between the generated dicarbonyl species and the trifluoroethylimide hydrazide yields a hydrazone intermediate, which serves as the precursor for the final ring closure.
The final cyclization step is promoted by the synergistic action of the base (sodium dihydrogen phosphate/pyridine system) and the remaining iodine species. This environment facilitates the intramolecular nucleophilic attack of the terminal nitrogen onto the adjacent carbonyl carbon, followed by oxidative aromatization to establish the stable 1,2,4-triazole aromatic system. The inclusion of the trifluoromethyl group at the 3-position is preserved throughout this sequence due to the stability of the C-CF3 bond under these specific oxidative conditions. From an impurity control perspective, the mild nature of the iodine/DMSO system minimizes side reactions such as over-oxidation or polymerization, which are common pitfalls in harsher oxidative cyclizations. This results in a cleaner crude reaction profile, simplifying the downstream purification process and ensuring that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry.
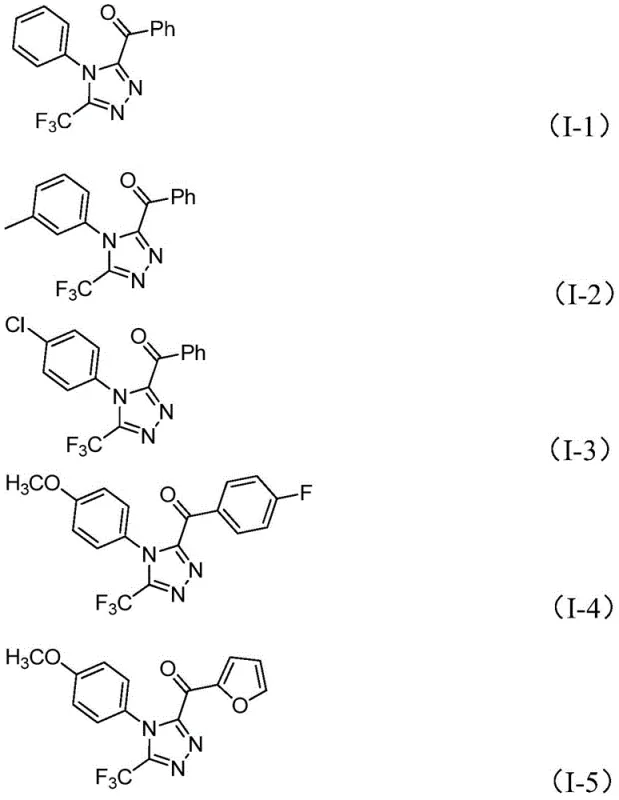
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to ensure optimal yield and reproducibility. The process is designed to be user-friendly, avoiding the need for specialized handling of air-sensitive reagents. The key to success lies in the precise control of reaction temperatures and the stoichiometric balance of the iodine promoter and base additives. By following the optimized protocol derived from the patent data, manufacturers can achieve consistent results across different batches, facilitating a smooth transition from gram-scale optimization to kilogram-scale production. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Oxidation Phase: React aryl ethyl ketone with elemental iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the mixture, then heat to 110-130°C for 12-20 hours.
- Purification: Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 3,4,5-trisubstituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage is the drastic simplification of the raw material portfolio. By relying on commodity chemicals like acetophenones and elemental iodine, companies can mitigate the risks associated with sourcing exotic or single-source catalysts. This diversification of supply sources enhances supply chain resilience, ensuring continuity of operations even during market fluctuations. Furthermore, the elimination of heavy metal catalysts removes the necessity for expensive metal scavengers and the associated waste disposal costs, leading to significant operational expenditure (OpEx) reductions. The simplified workup procedure, which involves basic filtration and standard chromatography, reduces the turnaround time for batch production, thereby improving overall asset utilization.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is substantial. By removing the dependency on precious metal catalysts such as palladium or rhodium, the direct material costs are significantly lowered. Additionally, the avoidance of strict anhydrous conditions means that solvents do not require energy-intensive drying processes, further reducing utility costs. The high atom economy of the tandem reaction minimizes waste generation, aligning with green chemistry principles and reducing the financial burden of environmental compliance and waste treatment. These cumulative factors contribute to a more lean and cost-competitive manufacturing process for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply chain reliability. Since the reaction tolerates a wide range of functional groups and does not require ultra-pure reagents, the risk of batch failure due to minor variations in raw material quality is minimized. The scalability of the process, demonstrated from gram to multi-gram levels in the patent data, suggests that it can be readily adapted for ton-scale production without encountering the heat transfer or mixing issues often associated with exothermic metal-catalyzed reactions. This scalability ensures that suppliers can meet sudden spikes in demand for key intermediates without compromising on delivery timelines or product quality.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a superior profile compared to traditional alternatives. The use of DMSO, a high-boiling polar solvent, allows for safe operation at elevated temperatures without the risk of high-pressure buildup associated with volatile organic solvents. The absence of toxic heavy metals simplifies the regulatory filing process for new drug applications, as residual metal limits are easier to meet. Moreover, the reduced number of unit operations and the potential for solvent recycling contribute to a lower carbon footprint for the manufacturing process, supporting corporate sustainability goals and enhancing the company's reputation as a responsible chemical manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their production pipelines. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely eliminating the need for costly and toxic heavy metal catalysts often found in traditional triazole synthesis.
Q: What are the key advantages regarding reaction conditions and safety?
A: The process operates under ambient atmospheric conditions without the need for strict anhydrous or oxygen-free environments, significantly simplifying operational requirements and enhancing safety profiles for large-scale manufacturing.
Q: Is this method suitable for producing diverse triazole derivatives?
A: Yes, the protocol demonstrates excellent functional group tolerance, allowing for the synthesis of various derivatives with different substituents on both the N1 and C5 positions, making it highly versatile for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the potential of the iodine-promoted synthesis described in patent CN113105402B and is prepared to leverage this technology to support your drug development programs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable partner committed to innovation and excellence. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your project timelines while reducing overall costs.
