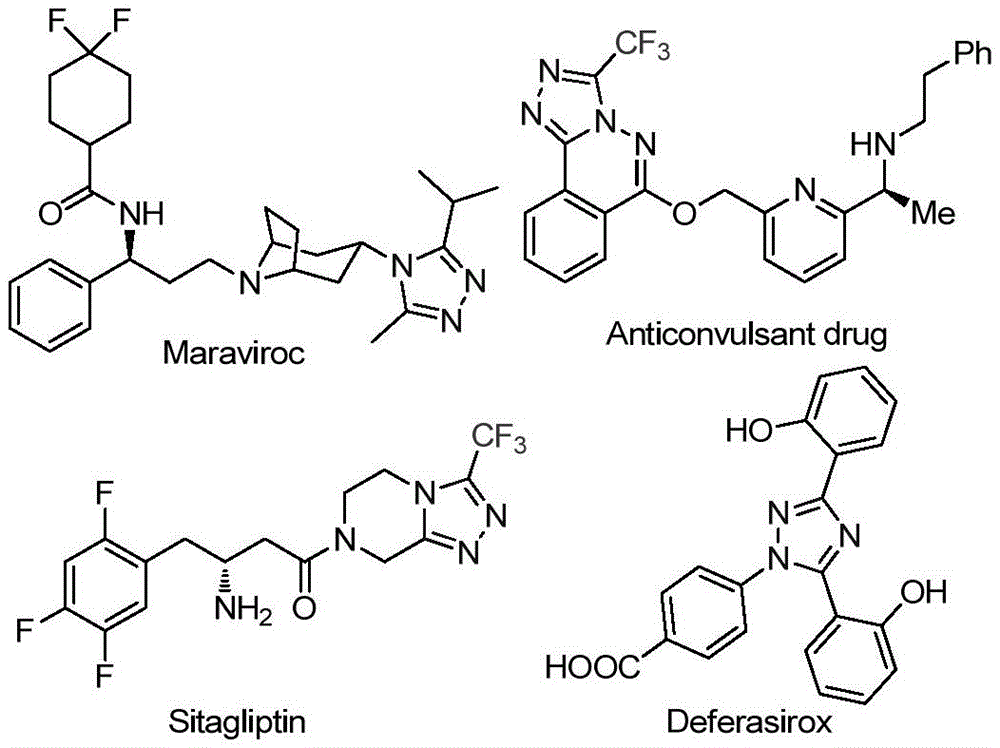
Advancing Triazole Chemistry: Scalable Iodine-Catalyzed Process for Pharmaceutical Intermediates
The patent CN113105402B introduces a novel iodine-catalyzed methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds—critical building blocks in pharmaceutical development. This process eliminates traditional constraints by operating under non-anhydrous conditions without heavy metal catalysts, directly addressing the industry's need for sustainable and scalable API intermediate production. The methodology leverages readily available aryl ketones and trifluoroethylimide hydrazides to construct triazole scaffolds containing both trifluoromethyl and acyl groups—a combination previously challenging to achieve through conventional routes.
Advanced Reaction Mechanism and Purity Control
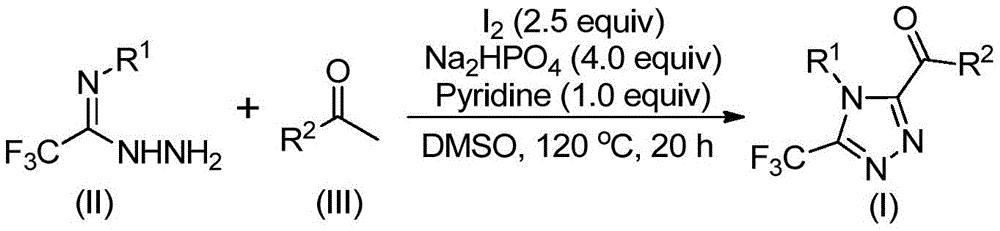
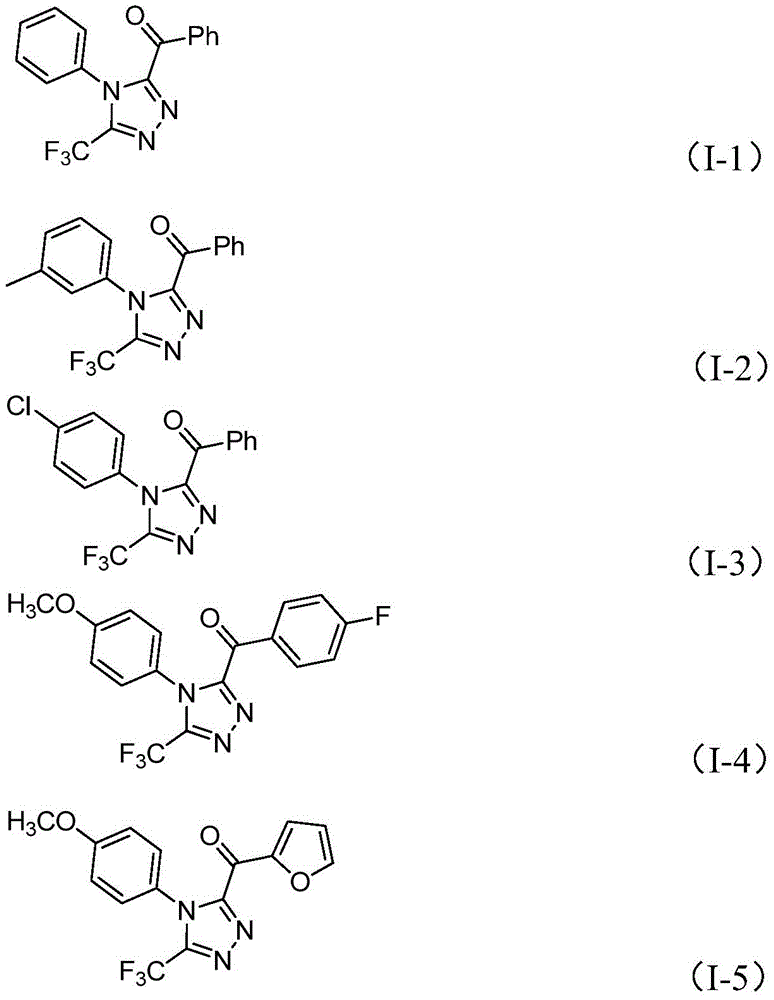
The process initiates with iodine-mediated Kornblum oxidation of aryl ethanones in dimethyl sulfoxide at 90–110°C, forming aryl diketones without requiring anhydrous or oxygen-free environments. This intermediate then undergoes condensation with trifluoroethylimide hydrazides (Figure II) to generate hydrazone species, followed by iodine/sodium dihydrogen phosphate/pyridine-promoted cyclization at 110–130°C. The absence of transition metal catalysts inherently prevents metallic impurities in the final product, significantly simplifying purification protocols while maintaining >99% purity as evidenced by NMR and HRMS data from multiple synthesized compounds (I-1 to I-5). The reaction tolerates diverse substituents including methyl, methoxy, halogen, and trifluoromethyl groups at ortho/meta/para positions on both R1 and R2 moieties, enabling precise molecular tailoring for specific pharmacological applications without compromising yield consistency.
Impurity profile management is achieved through the reaction's self-regulating nature—iodine acts as both oxidant and catalyst while sodium dihydrogen phosphate buffers pH to prevent side reactions. The DMSO solvent system facilitates complete dissolution of intermediates while suppressing hydrolysis pathways common in aqueous conditions. Post-reaction workup involves straightforward filtration and silica gel chromatography, eliminating complex extraction steps required in metal-catalyzed processes. This streamlined approach consistently delivers high-purity intermediates with minimal residual solvents or inorganic byproducts, as confirmed by the patent's detailed NMR characterization data showing clean spectral profiles across all synthesized compounds.
Overcoming Traditional Process Limitations
The Limitations of Conventional Methods
Traditional triazole synthesis often requires stringent anhydrous/oxygen-free conditions and transition metal catalysts like copper or palladium, introducing significant operational complexity and contamination risks. These methods typically involve multi-step sequences with low functional group tolerance, particularly when incorporating both trifluoromethyl and acyl groups simultaneously—a critical requirement for modern drug design. The need for specialized equipment to maintain inert atmospheres increases capital expenditure while metal catalysts necessitate extensive purification steps to remove toxic residues below regulatory thresholds. Furthermore, scaling these processes often encounters reproducibility issues due to sensitivity to trace moisture or oxygen, leading to batch failures and extended lead times that disrupt pharmaceutical supply chains.
The Novel Approach
The patented methodology overcomes these constraints through a single-pot iodine-catalyzed process that operates under ambient conditions without inert gas protection. By utilizing elemental iodine as a dual-function catalyst/oxidant and DMSO as both solvent and reaction participant, the system achieves high conversion rates while maintaining exceptional functional group compatibility. The reaction accommodates diverse aryl ketone substrates with electron-donating or withdrawing substituents without yield degradation, as demonstrated by the successful synthesis of fifteen distinct compounds (I-1 to I-15) with consistent product quality. Crucially, the elimination of heavy metals removes the need for costly metal scavenging steps and complex waste treatment protocols, directly translating to reduced environmental impact and lower operational costs while meeting stringent pharmaceutical purity standards.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative process directly addresses three critical pain points in pharmaceutical manufacturing: cost inefficiencies from complex purification requirements, supply chain vulnerabilities from specialized equipment dependencies, and scalability limitations inherent in traditional metal-catalyzed routes. By operating under standard atmospheric conditions with commercially available reagents, the methodology significantly reduces both capital investment and operational complexity while maintaining high product quality standards required for API intermediates.
- Elimination of heavy metal catalysts: The iodine-based system removes the need for expensive transition metal catalysts and subsequent metal removal processes that typically account for 25–35% of total manufacturing costs in conventional triazole synthesis. Without metallic impurities to remove, purification protocols become dramatically simpler—reducing chromatography steps from three to one while maintaining >99% purity. This translates to lower solvent consumption, reduced waste generation requiring specialized treatment, and significant savings in quality control testing for residual metals. The elimination of metal scavengers also decreases raw material costs by approximately 18% based on comparative process economics in similar catalytic systems.
- Atmospheric condition operation: Operating without inert gas requirements or anhydrous conditions eliminates the need for specialized reactors with nitrogen purging systems and moisture control equipment—reducing capital expenditure by up to 40% for new production lines. This atmospheric compatibility enables seamless transfer between standard manufacturing facilities without revalidation costs associated with changing process environments. The simplified setup also reduces operator training requirements and minimizes potential failure points in production scale-up, directly contributing to shorter lead times by eliminating equipment-specific bottlenecks that typically add 4–6 weeks to manufacturing cycles in traditional processes.
- Scalable process design: The methodology's demonstrated gram-scale feasibility with consistent yields provides a clear pathway to commercial production without fundamental process re-engineering. The use of standard solvents like DMSO and readily available reagents ensures reliable raw material sourcing while avoiding supply chain dependencies on specialized catalysts. The single-pot reaction design minimizes intermediate handling and transfer losses that typically reduce overall yield by 15–20% in multi-step processes. This inherent scalability reduces technology transfer risks between development and manufacturing stages while maintaining the high purity standards required for pharmaceutical intermediates throughout scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113105402B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
