Advancing Pharmaceutical Intermediate Manufacturing with Safe Palladium-Catalyzed Carbonylation Technology
Introduction to Advanced Multicomponent Synthesis Strategies
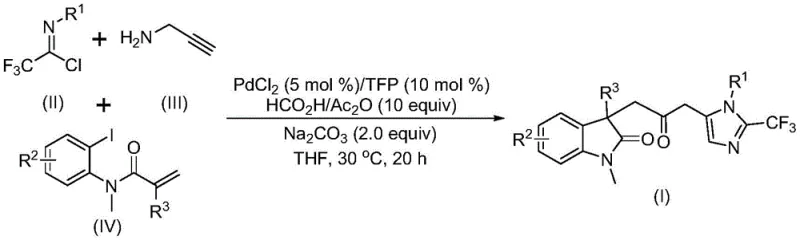
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, addressing critical challenges in modern organic synthesis. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. The significance of this invention lies not only in its chemical elegance but also in its operational safety and scalability. By replacing hazardous gaseous carbon monoxide with a liquid surrogate system, the process mitigates significant safety risks associated with traditional carbonylation reactions. Furthermore, the method operates under remarkably mild conditions, typically at 30°C, which preserves sensitive functional groups and reduces energy consumption. For R&D directors and process chemists, this represents a viable pathway to access diverse libraries of bioactive molecules containing indolinone and imidazole motifs, which are prevalent in numerous drug candidates and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocyclic systems has been fraught with significant technical and safety hurdles. Traditional approaches often rely on the direct coupling of two heterocyclic substrates or oxidative cyclization reactions that require harsh conditions and expensive oxidants. More critically, classical carbonylation strategies necessitate the use of carbon monoxide gas, which is highly toxic and requires specialized high-pressure equipment and rigorous safety protocols to handle. These requirements create substantial barriers to entry for many manufacturing facilities, increasing both capital expenditure and operational complexity. Additionally, conventional methods frequently suffer from poor substrate compatibility, limiting the diversity of substituents that can be introduced onto the final molecular scaffold. The need for multiple synthetic steps to install the carbonyl bridge further exacerbates cost and waste issues, making these routes less attractive for large-scale commercial production where efficiency and atom economy are paramount concerns for supply chain optimization.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by employing a one-pot, multicomponent reaction strategy that elegantly bypasses the limitations of prior art. Instead of using toxic CO gas, the process utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in situ, ensuring a steady and safe supply of the carbonyl source directly within the reaction medium. This innovation eliminates the need for high-pressure reactors, allowing the reaction to proceed at atmospheric pressure and a mild temperature of 30°C. The use of a palladium catalyst system, specifically palladium chloride paired with trifurylphosphine ligands, facilitates a cascade sequence involving Heck reaction, carbonylation, and cyclization in a single operational step. This telescoped approach not only streamlines the synthetic route but also enhances overall yield and purity by minimizing intermediate isolation steps. The broad substrate scope allows for the incorporation of various functional groups, including halogens, alkyls, and trifluoromethyl groups, providing medicinal chemists with unparalleled flexibility in designing novel analogues for structure-activity relationship studies.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring reproducibility at scale. The catalytic cycle likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction, which constructs the initial cyclic framework and forms a divalent alkyl-palladium species. Subsequently, the in situ generated carbon monoxide inserts into the palladium-carbon bond, yielding an acyl-palladium intermediate. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted intermolecular process to form a trifluoroacetamidine species, which may undergo isomerization. The final stage involves the activation of this amidine compound by the acyl-palladium intermediate, triggering an intramolecular cyclization that forge the second heterocyclic ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active palladium catalyst. This intricate dance of bond formations highlights the sophistication of the catalytic system and underscores the importance of precise stoichiometry and ligand selection.
Impurity control is another critical aspect governed by the reaction mechanism. The mild reaction temperature of 30°C plays a pivotal role in suppressing side reactions such as polymerization of the acrylamide or decomposition of the sensitive imidoyl chloride. The choice of tetrahydrofuran (THF) as the solvent is also strategic, as it effectively solubilizes all reactants while stabilizing the palladium intermediates. Furthermore, the use of sodium carbonate as a base ensures neutralization of acidic byproducts without promoting unwanted hydrolysis of the acid chloride or amide functionalities. Post-reaction processing involves simple filtration to remove inorganic salts and palladium residues, followed by silica gel column chromatography. This straightforward purification protocol is essential for achieving the high purity specifications required for pharmaceutical intermediates, ensuring that the final product meets stringent quality standards regarding residual metals and organic impurities, thereby facilitating downstream processing in API manufacturing.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to specific procedural guidelines regarding reagent addition and reaction monitoring. The process begins with the preparation of the catalytic system in an inert atmosphere to prevent oxidation of the palladium species. Precise weighing of the substrates according to the molar ratios specified in the patent is essential to maximize conversion and minimize waste. The reaction mixture should be stirred vigorously to ensure homogeneous mass transfer, particularly given the multiphase nature of some reagent interactions. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint, typically falling within the 12 to 20-hour window. Detailed standardized synthesis steps follow below to guide laboratory and pilot plant execution.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours to allow for the carbonylation cascade and cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The elimination of toxic carbon monoxide gas cylinders removes a major logistical and safety burden, significantly reducing the costs associated with hazardous material storage, transportation, and regulatory compliance. This shift to liquid reagents simplifies the procurement process, as formic acid and acetic anhydride are commodity chemicals available from multiple global suppliers, ensuring supply continuity even during market fluctuations. Moreover, the mild reaction conditions translate to substantial energy savings, as there is no need for heating to high temperatures or maintaining high-pressure environments. This reduction in energy intensity contributes to a lower carbon footprint, aligning with modern sustainability goals and potentially qualifying the manufacturing process for green chemistry incentives. The robustness of the reaction across a wide range of substrates means that a single production line can be utilized to manufacture a diverse portfolio of intermediates, maximizing asset utilization and flexibility.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this synthesis drastically reduces the number of unit operations required compared to stepwise conventional methods. By combining multiple bond-forming events into a single reactor charge, manufacturers save on solvent usage, labor hours, and equipment occupancy time. The avoidance of expensive high-pressure autoclaves further lowers capital expenditure requirements for new production lines. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproduct, directly lowering the cost of goods sold (COGS). The simplicity of the workup procedure, involving basic filtration and standard chromatography, minimizes the need for complex separation technologies, further driving down operational expenses.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as trifluoroethylimidoyl chloride and propargylamine ensures a reliable supply chain. Unlike specialized reagents that may have long lead times or single-source dependencies, the key inputs for this process are produced by numerous chemical manufacturers globally. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the scalability of the process from gram-scale to potential tonnage production, as evidenced by the patent data, provides confidence that supply can be ramped up quickly to meet market demand without requiring extensive process re-engineering. This agility is crucial for responding to the dynamic needs of the pharmaceutical market.
- Scalability and Environmental Compliance: The environmental profile of this process is superior to traditional carbonylation methods. By generating carbon monoxide in situ from liquid precursors, the risk of accidental gas leaks is virtually eliminated, enhancing workplace safety and reducing insurance premiums. The use of THF as a solvent, while requiring recovery systems, is well-established in the industry with mature recycling protocols. The reduction in waste generation due to higher selectivity and fewer purification steps aligns with strict environmental regulations regarding waste disposal. This compliance ease facilitates faster regulatory approvals for new manufacturing sites. The ability to operate at ambient pressure also simplifies the engineering controls required for scale-up, making the transition from pilot plant to commercial production smoother and less capital-intensive.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and descriptions provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing workflows.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly reducing workplace hazards and regulatory compliance burdens.
Q: What represents the key structural motif formed in this reaction?
A: The reaction constructs a complex carbonyl-bridged biheterocyclic scaffold, specifically linking an indolinone core with a trifluoromethyl-substituted imidazole ring, which is highly valued in medicinal chemistry for its biological activity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to gram-level synthesis with high yields and simple post-treatment procedures, indicating strong potential for commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN115353511A for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from laboratory discovery to market supply. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, including advanced solvent recovery and metal scavenging systems to meet stringent purity specifications. Our rigorous QC labs employ cutting-edge analytical techniques to verify the identity and purity of every batch, guaranteeing that the carbonyl-bridged biheterocyclic compounds we deliver meet the exacting standards required for drug development and manufacturing.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data for similar structures and comprehensive route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes safety, efficiency, and reliability, empowering you to bring life-saving medicines to market faster and more cost-effectively.
