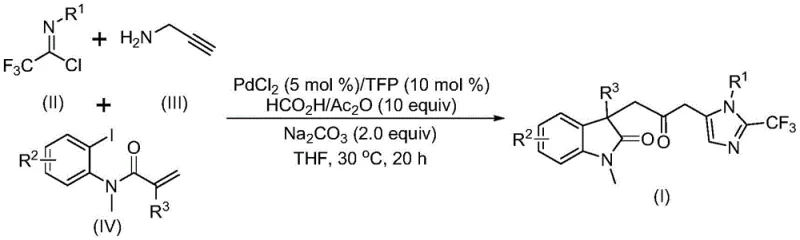
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates
Introduction to Patent CN115353511A
The pharmaceutical industry continuously seeks efficient routes to complex heterocyclic scaffolds that serve as core structures for bioactive molecules. Patent CN115353511A discloses a groundbreaking multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. These biheterocyclic systems are prevalent in natural products and drug candidates due to their broad-spectrum biological activities. The disclosed technology represents a significant leap forward in organic synthesis by replacing hazardous traditional carbonylation reagents with a safe, in-situ carbon monoxide generation system. This innovation not only streamlines the synthetic workflow but also enhances the safety profile of manufacturing processes, addressing critical concerns for both research laboratories and industrial production facilities seeking reliable pharmaceutical intermediate supplier partnerships.
At the heart of this invention lies a transition metal palladium-catalyzed cascade reaction that efficiently constructs multiple chemical bonds in a single operation. The method utilizes cheap and readily available starting materials, including trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, to assemble the complex architecture. By operating at mild temperatures around 30°C and avoiding the need for external toxic carbon monoxide gas cylinders, this process offers a distinct advantage over prior art methods that often require harsh conditions or specialized high-pressure equipment. The ability to introduce diverse substituents through rational substrate design further underscores the versatility of this approach, enabling the rapid generation of diversified libraries for drug discovery programs while maintaining high reaction efficiency and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic compounds has been fraught with significant synthetic challenges that hinder efficient production. Traditional strategies often rely on the direct coupling of two pre-formed heterocyclic substrates, which can suffer from low atom economy and poor regioselectivity. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles frequently require stoichiometric oxidants and harsh reaction conditions that are incompatible with sensitive functional groups. Perhaps the most prohibitive limitation in existing literature is the reliance on transition metal-catalyzed carbonylation using exogenous carbon monoxide gas. Handling toxic CO gas necessitates specialized high-pressure reactors and rigorous safety protocols, drastically increasing capital expenditure and operational complexity. Furthermore, these conventional routes often struggle with substrate compatibility, limiting the structural diversity accessible to medicinal chemists exploring structure-activity relationships.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent employs a sophisticated palladium-catalyzed multicomponent reaction that elegantly bypasses these obstacles. By utilizing a formic acid and acetic anhydride mixture as a safe, liquid carbon monoxide surrogate, the process eliminates the need for dangerous gas handling infrastructure. This one-pot transformation seamlessly integrates the formation of the indolinone core via an intramolecular Heck-type cyclization with the subsequent installation of the carbonyl bridge and the imidazole ring. The reaction proceeds under remarkably mild conditions, typically at 30°C in tetrahydrofuran (THF), ensuring high functional group tolerance. This methodology allows for the simultaneous construction of multiple bonds, significantly reducing step count and waste generation. The result is a robust, scalable protocol that transforms simple, commercially available building blocks into high-value biheterocyclic intermediates with exceptional efficiency.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the precision of modern organometallic catalysis. The cycle likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by a crucial intramolecular Heck reaction, where the palladium center inserts into the alkene moiety to generate a divalent alkyl-palladium intermediate, effectively closing the five-membered indolinone ring. Subsequently, the carbon monoxide released from the formic acid/acetic anhydride system coordinates to the palladium center, undergoing migratory insertion to form a reactive acyl-palladium species. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted intermolecular process to form a trifluoroacetamidine intermediate, which subsequently isomerizes. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that releases the final carbonyl-bridged biheterocyclic product and regenerates the active catalyst.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in maintaining product integrity. Higher temperatures often promote side reactions such as polymerization of the acrylamide or decomposition of the sensitive imidoyl chloride. The use of sodium carbonate as a base ensures neutralization of acidic byproducts without inducing unwanted hydrolysis of the trifluoromethyl group or the amide linkage. Furthermore, the choice of tetrahydrofuran as the solvent optimizes the solubility of all three distinct components, preventing phase separation issues that could lead to incomplete conversion. This precise control over reaction parameters ensures a clean impurity profile, which is critical for downstream purification and meeting the stringent quality standards required for high-purity pharmaceutical intermediates intended for clinical applications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The synthesis of these complex scaffolds is streamlined through a standardized protocol that balances reagent stoichiometry with operational simplicity. The process begins by charging a reaction vessel with the palladium catalyst system, specifically palladium chloride and tris(2-furyl)phosphine (TFP) ligand, along with sodium carbonate and the CO-generating mixture. To this heterogeneous mixture, the three key organic components—trifluoroethylimidoyl chloride, propargylamine, and the functionalized acrylamide—are added in a specific molar ratio, typically favoring a slight excess of the amine and acrylamide to drive the equilibrium forward. The reaction is conducted in anhydrous THF to prevent moisture-induced degradation of the acid chloride. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield across different batch sizes.
- Combine palladium chloride, TFP ligand, sodium carbonate, and the CO source system (formic acid/acetic anhydride) in THF solvent.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12-20 hours, then filter and purify via column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical yield. The primary economic driver is the elimination of high-pressure carbon monoxide infrastructure, which represents a significant capital expenditure and ongoing maintenance cost in traditional carbonylation facilities. By switching to a liquid CO source, manufacturers can utilize standard glass-lined or stainless steel reactors, drastically simplifying the engineering requirements and reducing the barrier to entry for production. Additionally, the starting materials, such as propargylamine and acryloyl chloride derivatives, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and ensuring supply continuity even during market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several efficiency factors. The use of inexpensive palladium chloride instead of more exotic catalysts, combined with the avoidance of specialized high-pressure equipment, leads to substantial cost savings in both CAPEX and OPEX. Furthermore, the one-pot nature of the reaction eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption, labor hours, and waste disposal costs associated with multi-step sequences. The high atom economy of the multicomponent approach ensures that a greater proportion of raw material mass is incorporated into the final product, minimizing waste generation and aligning with green chemistry principles that are increasingly mandated by regulatory bodies.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this synthetic route. Because the reaction tolerates a wide array of functional groups—including halogens, nitro groups, and trifluoromethyl moieties—manufacturers can source diverse substituted precursors without needing to re-optimize the core process for each variant. This flexibility allows for rapid adaptation to changing customer specifications or raw material availability. Moreover, the mild reaction conditions reduce the risk of thermal runaways or equipment failure, ensuring consistent batch-to-batch quality and reliable delivery schedules. The ability to scale this process from gram-level discovery to multi-kilogram production without fundamental changes to the chemistry provides a seamless path from R&D to commercial supply.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental and safety challenges, but this methodology is inherently designed for scalability. The absence of toxic gas feeds simplifies the permitting process for new production lines and reduces the environmental footprint of the facility. Waste streams are primarily composed of organic solvents and inorganic salts, which are easier to treat and dispose of compared to heavy metal-contaminated wastes from other catalytic systems. The high selectivity of the reaction minimizes the formation of difficult-to-remove byproducts, simplifying the purification workflow and reducing the volume of silica gel or other chromatographic media required. This alignment with environmental, social, and governance (ESG) goals makes the process attractive for companies aiming to reduce their overall environmental impact while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the safety advantages of this carbonylation method compared to traditional routes?
A: Unlike conventional carbonylation which requires toxic high-pressure carbon monoxide gas, this patent utilizes a formic acid/acetic anhydride system to generate CO in situ, significantly enhancing operational safety and reducing equipment costs.
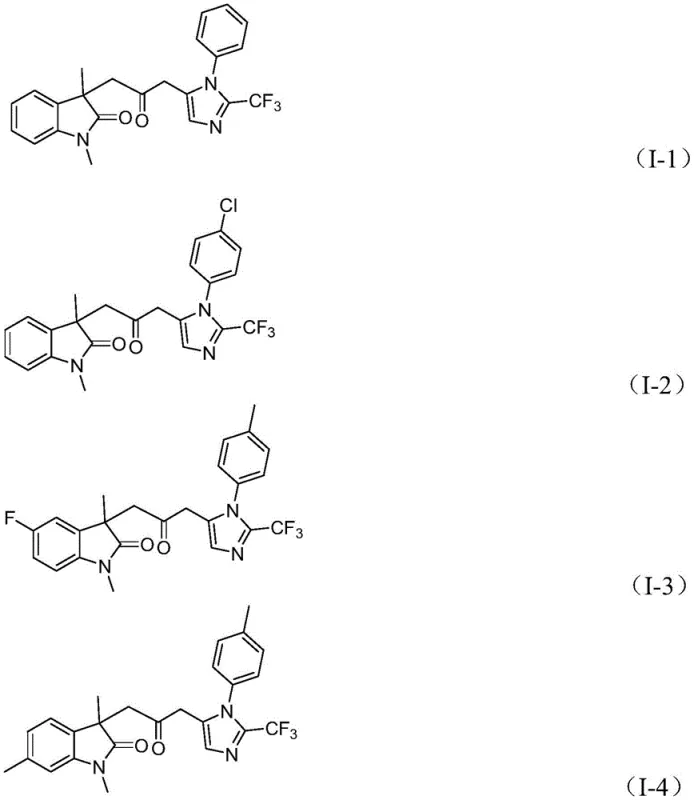
Q: What is the substrate scope for the R1 and R3 positions in this synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating alkyl, substituted aryl (including halogens, nitro, trifluoromethyl, and alkoxy groups), and benzyl substituents, allowing for diverse library generation.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the protocol operates at mild temperatures (30°C) with commercially available catalysts and avoids hazardous gases, making it highly amenable to scale-up from gram-level optimization to industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the pharmaceutical sector, partnering with an experienced CDMO is essential for successful project execution. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to industrial plant is seamless and efficient. Our team of expert chemists is well-versed in optimizing palladium-catalyzed reactions to meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. We understand that time-to-market is critical, and our established supply chains for key reagents allow us to initiate production rapidly upon project approval.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development program. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can accelerate the delivery of life-saving medicines to patients worldwide through innovative chemistry and reliable manufacturing excellence.
