Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
Introduction to Next-Generation Imidazole Synthesis
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and safer synthetic routes to bioactive scaffolds. Patent CN111423381B introduces a groundbreaking methodology for the preparation of 2-trifluoromethyl substituted imidazole compounds, a class of molecules renowned for their significant impact on drug pharmacokinetics. The introduction of a trifluoromethyl group into a heterocyclic core often results in markedly improved electronegativity, metabolic stability, and lipophilicity, making these structures highly desirable in modern medicinal chemistry programs. As illustrated in the structural diversity of bioactive imidazoles shown below, these motifs are foundational to numerous therapeutic agents.
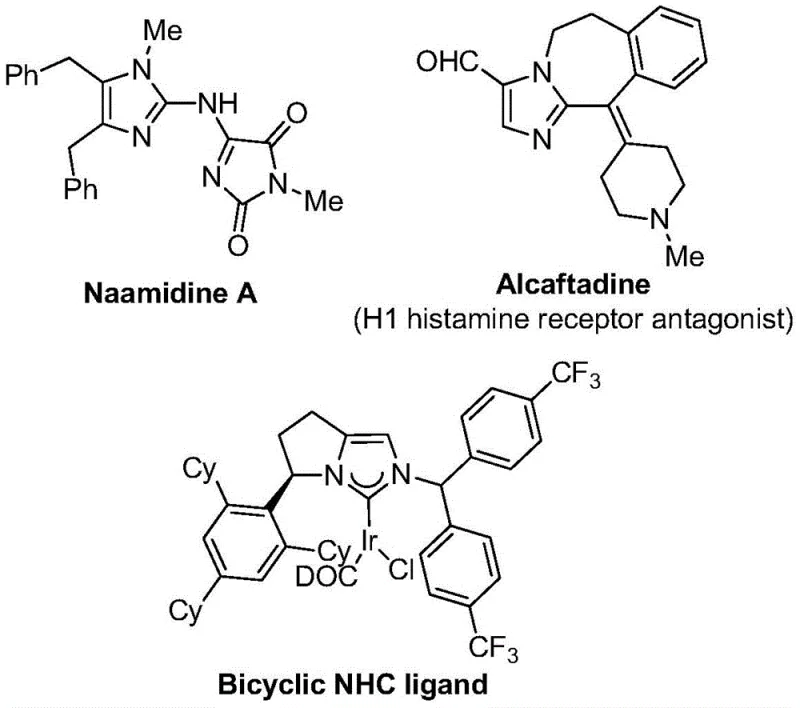
This novel approach distinguishes itself by leveraging a transition metal palladium-catalyzed carbonylation series reaction. Unlike traditional methods that may rely on hazardous reagents or extreme conditions, this invention utilizes cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The process operates under remarkably mild thermal conditions, typically around 30 °C, which not only enhances safety but also broadens the functional group tolerance, allowing for the synthesis of diversified derivatives essential for structure-activity relationship (SAR) studies in drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has presented significant challenges to process chemists. Conventional literature often describes methods involving the direct reaction of synthons with trifluoromethyl groups, such as trifluorodiazoethane, which poses severe safety risks due to its explosive nature and toxicity. Furthermore, many existing protocols require harsh reaction conditions, including high temperatures and strong bases, which can lead to the decomposition of sensitive substrates and the formation of complex impurity profiles. These limitations often result in low overall yields and necessitate cumbersome purification steps, thereby increasing the cost of goods and extending the timeline for developing reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in CN111423381B offers a transformative solution by employing a palladium-catalyzed cascade reaction that integrates carbonylation and cyclization in a single pot. As depicted in the general reaction scheme below, the process巧妙地 combines trifluoroethylimidoyl chloride, propargylamine, and a diaryl iodonium salt in the presence of a palladium catalyst and a carbon monoxide surrogate generated in situ from formic acid and acetic anhydride. This strategy eliminates the need for external CO gas cylinders, significantly improving operational safety. The reaction proceeds efficiently at 30 °C over 16 to 24 hours, demonstrating high atom economy and excellent compatibility with various substituents, thus providing a robust platform for the commercial scale-up of complex pharmaceutical intermediates.
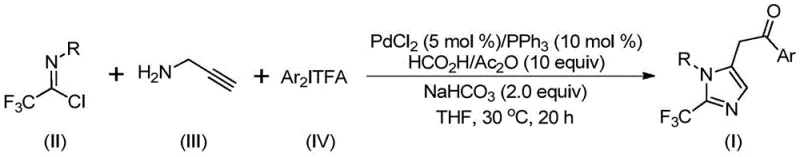
Mechanistic Insights into Palladium-Catalyzed Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific API candidates. The reaction initiates with the formation of a trifluoroacetamidine intermediate through an intermolecular carbon-nitrogen bond promotion facilitated by the base. Subsequently, isomerization occurs, followed by the palladation of the alkyne moiety of the propargylamine derivative to generate a key alkenyl palladium intermediate. This species undergoes further isomerization to form an alkyl palladium intermediate, setting the stage for the critical carbonylation step. The carbon monoxide, released slowly and controllably from the formic acid/acetic anhydride mixture, inserts into the palladium-carbon bond to yield an acyl palladium intermediate.
The catalytic cycle culminates with the oxidative addition of the diaryl iodonium salt to the acyl palladium species, forming a high-valent tetravalent palladium intermediate. Finally, reductive elimination releases the desired 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst. This intricate mechanism ensures high regioselectivity and minimizes side reactions. The versatility of this system is evidenced by the successful synthesis of various derivatives, including those with electron-rich and electron-deficient aryl groups, as shown in the specific examples below. Such broad substrate scope confirms the method's utility in generating diverse libraries for high-throughput screening.
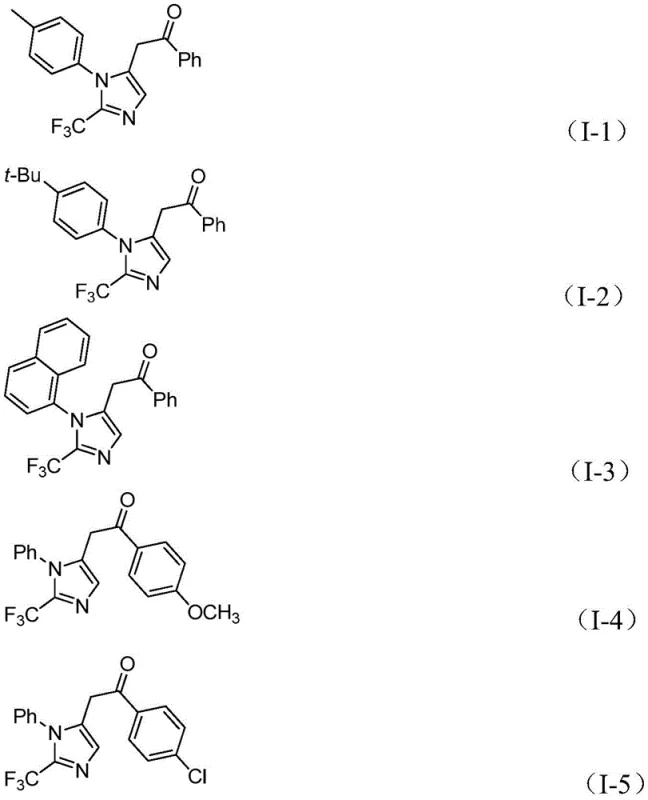
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and high purity. The procedure involves mixing the palladium catalyst, ligand, base, and reactants in an aprotic solvent like tetrahydrofuran (THF), which has been identified as the optimal medium for solubility and reaction efficiency. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the guide below to assist technical teams in replicating these results.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30 °C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted imidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. By shifting away from hazardous reagents like trifluorodiazoethane and high-pressure gas systems, companies can significantly reduce the capital expenditure associated with specialized safety infrastructure and regulatory compliance. The use of commercially available and inexpensive starting materials, such as aromatic amines and propargylamine, ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that often plague the production of specialty chemicals.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents, combined with the use of a relatively low loading of palladium chloride (5 mol%), leads to substantial cost savings in raw material procurement. Furthermore, the mild reaction temperature of 30 °C drastically reduces energy consumption compared to traditional high-temperature reflux processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: Since the key building blocks like trifluoroethylimidoyl chloride and diaryl iodonium salts can be readily synthesized from widely available precursors, the dependency on niche suppliers is minimized. This decentralization of the supply base enhances continuity of supply, ensuring that production schedules for critical API intermediates are maintained without interruption, even during global market fluctuations.
- Scalability and Environmental Compliance: The simplicity of the post-treatment process, which involves standard filtration and column chromatography, facilitates easy scale-up from gram to kilogram and eventually to ton-scale production. Additionally, the avoidance of toxic gases and the use of benign solvents align with increasingly stringent environmental regulations, reducing the burden of waste treatment and disposal while promoting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this technology, we have compiled a set of questions based on the specific experimental data and beneficial effects described in the patent documentation. These answers provide clarity on reaction conditions, catalyst performance, and substrate limitations, helping stakeholders make informed decisions about adopting this methodology for their specific projects.
Q: What is the primary advantage of using formic acid and acetic anhydride in this synthesis?
A: This combination serves as a safe and effective carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas while facilitating the carbonylation step efficiently.
Q: Can this method tolerate diverse functional groups on the aryl rings?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups such as nitro, chloro, and trifluoromethyl.
Q: Is the palladium catalyst loading economically viable for large-scale production?
A: The process utilizes a relatively low catalyst loading (5 mol% PdCl2), and since the reaction proceeds under mild conditions with high yields, it offers a cost-effective pathway suitable for scaling up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 2-trifluoromethyl imidazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive value and efficiency in your supply chain.
