Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are critical motifs in modern drug design due to their enhanced metabolic stability and lipophilicity. Patent CN111423381B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing significant bottlenecks in current synthetic strategies. This innovation leverages a transition metal palladium-catalyzed carbonylation series reaction that operates under remarkably mild conditions, specifically at 30°C, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The technical breakthrough lies not only in the high reaction efficiency and excellent substrate compatibility but also in the strategic use of formic acid and acetic anhydride as a safe carbon monoxide surrogate, eliminating the need for hazardous high-pressure CO gas cylinders. For R&D directors and process chemists, this represents a paradigm shift towards safer, more sustainable, and cost-effective manufacturing of high-value nitrogen-containing five-membered heterocycles that serve as pivotal scaffolds in bioactive molecules and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant operational challenges and safety hazards that impede large-scale commercial adoption. Conventional literature methods often rely on direct reactions between synthons containing trifluoromethyl groups and suitable substrates, frequently employing reagents like trifluorodiazoethane or trifluoroethylimidoyl halides under rigorous conditions. These legacy processes typically suffer from poor atom economy, require specialized high-pressure equipment to handle toxic carbon monoxide gas, and often exhibit limited functional group tolerance, leading to complex purification workflows and inconsistent yields. Furthermore, the use of unstable diazo compounds poses severe explosion risks in industrial settings, necessitating expensive safety infrastructure and strict regulatory compliance measures that drastically inflate the cost of goods sold. The inability to easily diversify the substitution patterns at the 1 and 5 positions of the imidazole ring further restricts the utility of these conventional routes for generating diverse libraries of drug candidates.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a sophisticated multicomponent coupling strategy that elegantly circumvents the drawbacks of prior art by integrating a palladium-catalyzed cascade reaction sequence. By employing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the core building blocks, this method achieves the construction of the imidazole core with simultaneous installation of the trifluoromethyl group in a single pot. The reaction proceeds efficiently at a mild temperature of 30°C over a period of 16 to 24 hours, utilizing a readily available catalyst system of palladium chloride and triphenylphosphine. Crucially, the generation of the necessary carbonyl moiety is achieved in situ through the decomposition of formic acid and acetic anhydride, providing a safe and controllable source of carbon monoxide without the logistical nightmares associated with gas handling. This methodology not only simplifies the operational procedure but also expands the practical utility of the synthesis by allowing for the easy modulation of substituents on the aryl rings, thereby facilitating the rapid generation of diverse molecular architectures.
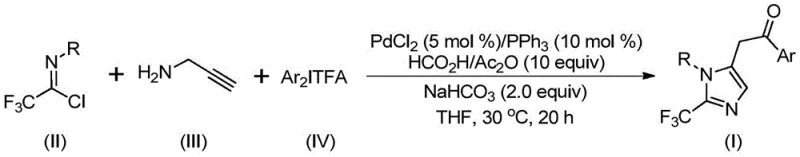
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of bond-forming events orchestrated by the palladium center. The process initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the alkaline additive, likely sodium bicarbonate, which generates a trifluoroacetamidine intermediate from the reaction of the imidoyl chloride and propargylamine. Following an initial isomerization, the palladium catalyst engages with the alkyne functionality of the amine to form a vinyl-palladium intermediate, which subsequently isomerizes into a more stable alkyl-palladium species. The pivotal step involves the carbonylation reaction, where the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond to generate an acyl-palladium intermediate. This reactive species then undergoes oxidative addition with the diaryl iodonium salt to form a high-valent tetravalent palladium complex, which finally collapses via reductive elimination to release the desired 2-trifluoromethyl-substituted imidazole product and regenerate the active catalytic species. Understanding this cycle is crucial for process optimization, as it highlights the delicate balance required between the various reagents to maintain catalytic turnover and prevent catalyst deactivation.
From an impurity control perspective, the specificity of this catalytic cycle offers distinct advantages in managing the杂质 profile of the final API intermediate. The use of diaryl iodonium salts as electrophiles is particularly beneficial because they are highly reactive yet selective, minimizing the formation of homocoupling byproducts that are common in other cross-coupling reactions. The mild reaction temperature of 30°C further suppresses thermal degradation pathways and side reactions that often plague high-temperature syntheses, resulting in a cleaner crude reaction mixture. Additionally, the choice of tetrahydrofuran (THF) as the preferred organic solvent ensures sufficient solubility of all reactants while effectively promoting the reaction kinetics, which contributes to high conversion rates and reduces the burden on downstream purification. The post-treatment procedure, involving simple filtration followed by silica gel mixing and column chromatography, is designed to efficiently remove palladium residues and inorganic salts, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, one must adhere to the precise stoichiometric ratios and reaction conditions outlined in the patent to maximize yield and reproducibility. The standard protocol involves charging a reaction vessel with palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2.0 equivalents), and a mixture of formic acid and acetic anhydride (10 equivalents) in THF. To this mixture, the trifluoroethylimidoyl chloride, propargylamine, and the specific diaryl iodonium salt are added in molar ratios that favor complete conversion, typically maintaining a slight excess of the imidoyl chloride and iodonium salt relative to the amine. The reaction is then stirred at 30°C for approximately 20 hours, after which the progress is monitored until completion. Detailed standardized synthesis steps for specific derivatives are provided in the guide below, ensuring that technical teams can replicate the high yields reported in the experimental data, which range from 65% to 97% depending on the electronic nature of the substituents.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture.
- Stir the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative potential for reducing the total cost of ownership and mitigating supply risks associated with complex heterocyclic intermediates. The primary economic driver is the utilization of commodity chemicals as starting materials; trifluoroethylimidoyl chloride can be rapidly synthesized from inexpensive aromatic amines, while propargylamine and diaryl iodonium salts are widely available from global chemical suppliers, ensuring a robust and competitive supply base. By eliminating the need for specialized high-pressure reactors and hazardous gas handling infrastructure, the capital expenditure (CAPEX) required for scaling this process is significantly reduced, allowing for faster deployment of manufacturing capacity. Furthermore, the operational simplicity of the workup procedure, which avoids complex extraction or distillation steps in favor of straightforward filtration and chromatography, translates directly into lower operational expenditures (OPEX) through reduced labor hours and solvent consumption. This streamlined process flow enhances the overall agility of the supply chain, enabling quicker response times to market demands for new drug candidates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the replacement of expensive and dangerous reagents with cost-effective alternatives, such as using formic acid and acetic anhydride instead of pressurized carbon monoxide cylinders. This substitution not only lowers the direct material costs but also eliminates the substantial overheads associated with safety compliance, gas storage, and specialized maintenance required for high-pressure systems. The high reaction efficiency and excellent yields observed across a broad range of substrates mean that less raw material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the use of a relatively inexpensive palladium catalyst system, which can potentially be recovered and recycled in optimized flows, further contributes to substantial cost savings in large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on commercially available and stable starting materials that are not subject to the same geopolitical or logistical constraints as exotic fluorinating agents. The ability to source trifluoroethylimidoyl chloride precursors from multiple vendors ensures continuity of supply even in the face of market fluctuations or disruptions. Moreover, the mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, leading to more predictable production schedules and reliable delivery timelines for downstream customers. This reliability is critical for pharmaceutical companies that require consistent quality and quantity of intermediates to maintain their own clinical trial and commercial manufacturing schedules without interruption.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles by minimizing waste generation and avoiding the use of toxic gases. The reaction can be easily scaled from gram-level laboratory experiments to multi-kilogram pilot plant operations without significant re-engineering, facilitating a smooth technology transfer from R&D to commercial manufacturing. The simplified post-treatment process reduces the volume of organic waste solvents and aqueous effluents, lowering the environmental footprint and disposal costs associated with production. This compliance with stringent environmental regulations not only avoids potential fines but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and investors in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this technology into existing manufacturing portfolios and for optimizing the supply chain strategy for fluorinated imidazole derivatives.
Q: What are the key advantages of this Pd-catalyzed method over traditional trifluoromethylation?
A: This method avoids hazardous trifluorodiazoethane and high-pressure CO gas, utilizing mild conditions (30°C) and inexpensive formic acid/acetic anhydride as a CO surrogate, significantly enhancing operational safety and cost-efficiency.
Q: What is the substrate compatibility for the aryl groups in this synthesis?
A: The process demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl substituents on both the imidoyl chloride and iodonium salt components.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the protocol uses commercially available starting materials and simple post-treatment procedures like filtration and column chromatography, making it highly scalable from gram-level laboratory synthesis to multi-kilogram commercial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development, and we are uniquely positioned to leverage this patented technology for your projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-trifluoromethyl imidazole intermediate meets the highest quality standards required by global regulatory agencies. Our commitment to excellence extends beyond mere manufacturing; we act as a strategic partner, offering deep technical insights and process optimization services to enhance the overall value of your supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits and efficiency gains achievable through this method. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics rather than theoretical projections. Let us collaborate to bring your next generation of fluorinated therapeutics to market faster and more cost-effectively.
