Advanced Industrial Synthesis of Agomelatine Intermediates via Green Chemistry and One-Pot Technology
Advanced Industrial Synthesis of Agomelatine Intermediates via Green Chemistry and One-Pot Technology
The pharmaceutical landscape for antidepressant therapies has been significantly reshaped by the introduction of Agomelatine, a novel melatonin receptor agonist and serotonin antagonist marketed under trade names such as Valdoxan® and Thymanax®. As global demand for this active pharmaceutical ingredient (API) continues to rise, the imperative for robust, scalable, and environmentally sustainable synthetic routes has never been more critical for supply chain stakeholders. Patent CN101759591A presents a groundbreaking methodology that addresses the longstanding inefficiencies of previous syntheses, offering a pathway that is not only chemically superior but also commercially viable for large-scale production. This technical insight report analyzes the proprietary innovations within this patent, focusing on the strategic replacement of toxic solvents, the implementation of a streamlined one-pot reduction-acetylation protocol, and the rigorous control of impurity profiles to ensure pharmaceutical-grade quality.
![Chemical structure of Agomelatine (N-[2-(7-methoxy-1-naphthyl)ethyl]acetamide)](/insights/img/agomelatine-intermediate-synthesis-pharma-supplier-20260305111437-02.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Agomelatine has been plagued by complex multi-step sequences that suffer from low overall yields and significant environmental liabilities. For instance, the method disclosed in European Patent EP 0447285 relies on an eight-step reaction sequence that results in an average yield of less than 30%, creating a bottleneck for cost-effective manufacturing. This legacy approach involves cumbersome aromatization and saponification steps to obtain the acid intermediate, followed by conversion to the amide and subsequent dehydration. A critical failure point in this conventional technology is the incomplete aromatization, which leads to persistent impurities that are difficult to remove without expensive purification techniques. Furthermore, other reported methods, such as those cited in Synthesis Communications (2001), utilize benzene as a solvent for the dehydration step. Benzene is a Class 1 carcinogen with severe toxicity, posing unacceptable risks to operator safety and creating massive regulatory burdens for waste disposal and environmental compliance in modern GMP facilities.
The Novel Approach
The methodology outlined in CN101759591A represents a paradigm shift towards green chemistry and process intensification. By utilizing 7-methoxy-1-tetralone as the starting material, the new route achieves the key naphthyl acetonitrile intermediate through a highly selective cyanation followed by DDQ-mediated aromatization. Crucially, the dehydration step is performed in cyclohexane rather than benzene. This solvent switch is not merely a safety improvement; it fundamentally alters the reaction kinetics to suppress the formation of undesirable side-chain alkene impurities, thereby enhancing the selectivity of the production. The culmination of this process is a sophisticated one-pot reduction and acetylation step. Instead of isolating the amine intermediate, the process directly converts the nitrile to the final acetamide in a single reactor using Raney-Nickel and acetic anhydride. This consolidation eliminates multiple unit operations, drastically reducing the physical footprint required for production and minimizing the potential for product loss during transfer and isolation.
Mechanistic Insights into DDQ-Mediated Aromatization and Impurity Control
At the heart of this improved synthesis lies a precise control over the aromatization and dehydration mechanisms, which are critical for defining the final purity profile of the API. The process begins with the generation of a cyanohydrin intermediate using n-Butyl Lithium at cryogenic temperatures (-78°C), ensuring high regioselectivity. Subsequent oxidation with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) in dichloromethane efficiently converts the tetrahydronaphthalene system into the dihydronaphthalene derivative. However, the true innovation is observed in the final dehydration step where the dihydro-intermediate is refluxed in cyclohexane with p-toluenesulfonic acid (TsOH). In traditional benzene-based systems, the reaction conditions often promote elimination reactions that generate styrene-like derivatives on the side chain. These alkene impurities are structurally similar to the target molecule and are notoriously difficult to separate via crystallization.
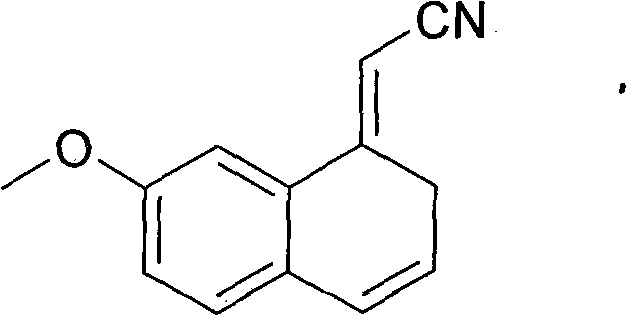
The use of cyclohexane as the solvent creates a specific solvation environment that favors the desired dehydration to the naphthyl acetonitrile while kinetically inhibiting the formation of the side-chain alkene impurity shown in the structural analysis above. By avoiding the generation of this specific contaminant at the source, the downstream purification burden is significantly lightened. This mechanistic advantage allows the final crude product to achieve high purity levels prior to even the final crystallization step. The subsequent one-pot hydrogenation utilizes Raney-Nickel under moderate hydrogen pressure (0.5-10 MPa) to reduce the nitrile group to the primary amine, which is immediately trapped by acetic anhydride present in the reaction mixture. This tandem reaction sequence prevents the accumulation of free amine, which can be prone to oxidation or dimerization, further safeguarding the chemical integrity of the final Agomelatine molecule.
How to Synthesize Agomelatine Efficiently
The transition from laboratory scale to commercial manufacturing requires a synthesis route that balances high yield with operational simplicity. The patented process described herein offers a clear roadmap for process engineers, moving away from the fragile multi-step sequences of the past toward a robust, convergent strategy. The key to success lies in the strict temperature control during the initial lithiation and the careful management of the one-pot hydrogenation parameters. By integrating the reduction and acetylation steps, manufacturers can achieve a significant reduction in cycle time and solvent usage. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for scaling this technology to multi-ton production capacities.
- Perform cyanation of 7-methoxy-1-tetralone using n-Butyl Lithium at -78°C to form the hydroxyl-acetonitrile intermediate.
- Execute aromatization using DDQ in dichloromethane, followed by dehydration in cyclohexane with tosic acid to prevent alkene impurities.
- Conduct a one-pot hydrogenation and acetylation using Raney-Ni and acetic anhydride, followed by crystallization with isopropyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis route described in CN101759591A translates directly into enhanced margin protection and supply security. The most immediate impact is seen in the drastic reduction of solvent consumption and waste generation. Traditional methods often require excessive volumes of solvents for extraction and purification due to poor selectivity. In contrast, this novel method utilizes a crystallization protocol with isopropyl ether that requires only 20 to 30 volumes of solvent to achieve high purity, compared to the 100 volumes typically needed in older processes. This reduction in solvent intensity directly correlates to lower raw material costs and reduced expenses associated with solvent recovery and hazardous waste disposal. Furthermore, the elimination of benzene removes a major regulatory liability, simplifying the permitting process for manufacturing sites and ensuring uninterrupted production schedules free from environmental compliance audits related to carcinogen handling.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the consolidation of reaction steps and the optimization of reagent stoichiometry. By executing the reduction and acetylation in a single vessel (one-pot), the need for intermediate isolation, drying, and re-dissolution is completely removed. This not only saves on labor and energy costs but also minimizes mechanical losses of material that occur during each transfer operation. Additionally, the use of Raney-Nickel, a relatively inexpensive heterogeneous catalyst, replaces more costly noble metal catalysts often found in fine chemical synthesis. The high selectivity of the cyclohexane-mediated dehydration ensures that the yield of the key nitrile intermediate remains high, preventing the compounding yield losses that typically erode profitability in long linear syntheses.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or hazardous reagents that have volatile market availability. This synthesis route relies on commodity chemicals such as cyclohexane, acetic anhydride, and standard inorganic bases, all of which are readily available in the global chemical market with stable pricing. The robustness of the process, characterized by its tolerance to standard industrial operating conditions (e.g., moderate hydrogen pressures and ambient temperature aromatization), means that it can be executed in a wide range of multipurpose chemical plants without requiring specialized exotic equipment. This flexibility allows for easier technology transfer between manufacturing sites, reducing the risk of supply disruption due to site-specific maintenance or capacity constraints.
- Scalability and Environmental Compliance: Scaling a chemical process often amplifies safety and environmental issues, but this methodology is inherently designed for green manufacturing. The avoidance of benzene and the reduction in solvent volumes significantly lower the facility's environmental footprint, aligning with increasingly stringent global ESG (Environmental, Social, and Governance) standards. The crystallization step, which uses isopropyl ether, is easily scalable and provides a high-purity solid product (HPLC > 99.5%) suitable for direct formulation or minimal further processing. This high quality at the intermediate stage reduces the burden on the final API manufacturing suite, allowing for faster batch turnover and higher overall plant throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Agomelatine synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, safety, and cost-efficiency. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for commercial production.
Q: How does the new synthesis route improve purity compared to EP 0447285?
A: The new route replaces the multi-step saponification and dehydration sequence with a direct DDQ aromatization and controlled dehydration in cyclohexane. This specifically avoids the formation of side-chain alkene impurities common in benzene-based solvents, achieving HPLC purity greater than 99.5% without extensive column chromatography.
Q: What are the safety advantages of using cyclohexane over benzene in this process?
A: Benzene is a known carcinogen with severe toxicity, posing significant health risks and regulatory hurdles in industrial settings. The patented method utilizes cyclohexane as the solvent for the dehydration step, which eliminates these toxicological hazards while simultaneously improving reaction selectivity and reducing environmental compliance costs.
Q: How does the one-pot reduction-acetylation step impact manufacturing costs?
A: By combining the reduction of the nitrile group and the subsequent acetylation into a single reactor vessel using Raney-Ni and acetic anhydride, the process eliminates intermediate isolation and purification steps. This consolidation drastically reduces solvent consumption, labor hours, and equipment turnaround time, leading to substantial operational expenditure savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agomelatine Supplier
The technical advancements detailed in patent CN101759591A underscore the complexity and sophistication required to produce high-quality antidepressant intermediates at a commercial scale. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Agomelatine intermediate meets the exacting standards required by global regulatory bodies. We understand that the transition from a patented laboratory method to a robust industrial process requires deep expertise in process engineering and hazard analysis, capabilities that are central to our CDMO service model.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By partnering with our technical procurement team, you can gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible economic benefits of our optimized synthesis. Let us collaborate to secure your supply chain with a reliable, cost-effective, and environmentally responsible source of high-purity Agomelatine intermediates.
