Advanced One-Pot Synthesis of Agomelatine: Enhancing Purity and Scalability for Global Pharmaceutical Markets
Advanced One-Pot Synthesis of Agomelatine: Enhancing Purity and Scalability for Global Pharmaceutical Markets
The pharmaceutical industry continuously seeks robust synthetic routes for high-value active pharmaceutical ingredients (APIs) that balance efficiency, safety, and cost-effectiveness. Patent CN102206170A introduces a transformative preparation method for Agomelatine, a potent melatonin receptor agonist and serotonin 2C receptor antagonist used in the treatment of major depressive disorder. This innovation addresses critical bottlenecks in existing manufacturing processes by employing a novel one-pot synthesis technique that utilizes (7-methoxy-1-naphthyl) acetonitrile as the starting material. By integrating the reduction and acetylation steps into a single operational sequence using Nickel Chloride Hexahydrate and a borohydride reducing agent, this method significantly streamlines production. The structural complexity of Agomelatine, as depicted below, demands precise control over reaction conditions to ensure high purity and minimize toxic impurities, a challenge this patent successfully overcomes through mild reaction parameters and simplified downstream processing.
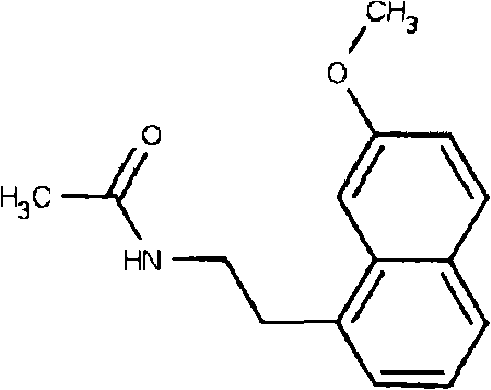
For R&D directors and process chemists, the implications of this technology are profound, offering a pathway to reliable pharmaceutical intermediate supplier status through superior process control. The shift from multi-step, harsh condition syntheses to a温和 (mild) one-pot protocol represents a significant leap forward in green chemistry principles applied to antidepressant manufacturing. This report analyzes the technical merits of this approach, evaluating its potential for commercial scale-up of complex pharmaceutical intermediates and its ability to deliver substantial cost savings without compromising on the stringent quality standards required for mental health therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Agomelatine has been plagued by inefficient synthetic routes that hinder cost reduction in API manufacturing. Early methodologies, such as those described in patent EP0447285, relied on 7-methoxy-1-tetralone as a starting material, necessitating a cumbersome eight-step sequence involving Reformatsky reactions, sulfur dehydrogenation, hydrolysis, chlorination, ammonification, dehydration, reduction, and finally acetylation. This excessive step count not only results in a dismal overall yield but also introduces numerous opportunities for impurity generation, complicating the purification landscape. Furthermore, alternative routes like those in patent CN 101643433A utilize Raney Nickel catalysts under high-temperature and high-pressure hydrogenation conditions. These harsh environments impose severe demands on reactor infrastructure, increase energy consumption, and present significant safety hazards due to the handling of high-pressure hydrogen gas and pyrophoric catalysts. Additionally, other methods involving chloroform as a solvent raise environmental and toxicity concerns, making them increasingly untenable in modern regulatory frameworks focused on sustainability and operator safety.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102206170A employs a highly efficient one-pot synthesis strategy that fundamentally reimagines the production workflow. By selecting (7-methoxy-1-naphthyl) acetonitrile as the direct precursor, the process bypasses the need for constructing the naphthalene ring system from scratch, immediately shortening the synthetic timeline. The core innovation lies in the simultaneous execution of reduction and acetylation within a single reaction vessel using methanol as a benign solvent medium. This approach eliminates the isolation of the intermediate amine, which is often unstable and prone to oxidation, thereby preserving material integrity. The use of Nickel Chloride Hexahydrate as a catalyst precursor, combined with sodium borohydride, allows the reaction to proceed at mild temperatures ranging from -10°C to 20°C under atmospheric pressure. This drastic reduction in reaction severity not only enhances operational safety but also lowers the barrier to entry for manufacturing facilities lacking specialized high-pressure equipment, effectively democratizing the production of this high-value antidepressant intermediate.
Mechanistic Insights into Nickel-Catalyzed Reductive Acetylation
The chemical elegance of this process is rooted in the in-situ generation of the active catalytic species and the seamless transition from nitrile reduction to amide formation. As illustrated in the reaction scheme below, the process begins with the dissolution of the nitrile starting material, Nickel Chloride Hexahydrate, and acetic anhydride in methanol. Upon the addition of the reducing agent, typically sodium borohydride, the nickel salt is reduced to an active nickel-boride species or a similar low-valent nickel complex capable of facilitating hydride transfer. This catalytic system selectively reduces the nitrile group (-CN) of (7-methoxy-1-naphthyl) acetonitrile to the corresponding primary amine (-CH2NH2) without affecting the methoxy substituent or the aromatic naphthalene system. The presence of acetic anhydride in the reaction mixture is critical; as soon as the amine intermediate is formed, it undergoes immediate nucleophilic attack on the acetic anhydride to form the stable acetamide bond. This tandem mechanism prevents the accumulation of free amine, which could otherwise lead to side reactions such as dimerization or over-reduction, ensuring a clean conversion to the target Agomelatine molecule.
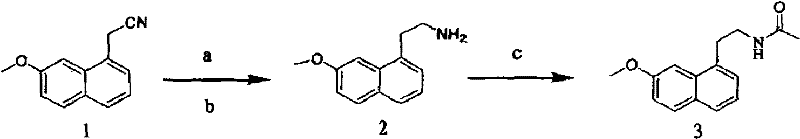
From an impurity control perspective, this mechanistic pathway offers distinct advantages for maintaining high-purity pharmaceutical intermediates. Traditional methods that isolate the amine intermediate often suffer from degradation products formed during storage or work-up. By trapping the amine in-situ as the acetamide, the process effectively locks the molecular structure into its final, stable form before any purification steps occur. The patent data highlights that optimizing the molar ratio of the reducing agent is crucial; specifically, a ratio of (7-methoxy-1-naphthyl) acetonitrile to sodium borohydride of 1:(3-5) ensures complete reduction while minimizing excess reagent waste. Furthermore, the choice of Nickel Chloride Hexahydrate over heterogeneous catalysts like Raney Nickel allows for a more homogeneous reaction environment, promoting better mass transfer and consistent reaction kinetics across large batches. This homogeneity is key to achieving the reported purity levels of 98.8% to 99.6%, as it minimizes the formation of localized hot-spots that often drive side reactions in heterogeneous catalysis.
How to Synthesize Agomelatine Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to maximize yield and safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to pilot plant operations with minimal re-optimization. The following guide summarizes the critical operational parameters derived from the patent embodiments, focusing on the preferred embodiment using sodium borohydride as the reducing agent. Operators must pay close attention to temperature control during the exothermic addition of the reducing agent to prevent thermal runaway, which could compromise product quality. Detailed standardized synthesis steps see the guide below.
- Dissolve (7-methoxy-1-naphthyl) acetonitrile, Nickel Chloride Hexahydrate, and Acetic Anhydride in methanol at room temperature, then cool the mixture to -15°C to -10°C.
- Add the reducing agent (preferably Sodium Borohydride) slowly while maintaining the temperature between -10°C and 20°C, stirring until the reduction is complete.
- Perform separation and purification via acid elution, ethyl acetate extraction, decolorization with activated carbon, and recrystallization using an ethyl acetate and isopropyl ether mixed solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates directly into tangible strategic benefits regarding cost structure and supply continuity. The shift away from high-pressure hydrogenation and multi-step sequences fundamentally alters the economic model of Agomelatine production. By eliminating the need for expensive high-pressure reactors and the rigorous safety protocols associated with hydrogen gas handling, capital expenditure (CAPEX) for manufacturing facilities is significantly reduced. Furthermore, the simplification of the process from potentially eight steps down to a concise one-pot operation drastically cuts down on labor hours, solvent consumption, and utility usage. This lean manufacturing approach aligns perfectly with the goals of cost reduction in API manufacturing, allowing suppliers to offer more competitive pricing while maintaining healthy margins. The use of readily available and inexpensive reagents like Nickel Chloride and Sodium Borohydride further insulates the supply chain from volatility in raw material markets, ensuring a stable and predictable cost base for long-term contracts.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of costly unit operations and hazardous reagents. Traditional routes relying on Raney Nickel require specialized filtration equipment and extensive safety measures due to the pyrophoric nature of the catalyst, adding hidden costs to the production bill. In contrast, the homogeneous nickel chloride system is easier to handle and quench. Additionally, the one-pot nature of the reaction removes the need for intermediate isolation, drying, and purification steps, which are typically the most resource-intensive parts of a synthetic sequence. This consolidation of steps leads to substantial cost savings by reducing solvent volumes, waste disposal fees, and energy consumption for heating and cooling cycles. The high yield of approximately 80.5% reported in the patent further amplifies these savings by maximizing the output per kilogram of starting material, effectively lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the new synthetic route. Complex multi-step syntheses are inherently fragile; a failure or delay in any single step can halt the entire production line, leading to missed delivery windows. By condensing the synthesis into a single pot, the number of potential failure points is minimized, resulting in a more reliable production schedule. Moreover, the reagents used—methanol, acetic anhydride, and sodium borohydride—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities, increasing the pool of qualified contract manufacturing organizations (CMOs) and reducing lead time for high-purity pharmaceutical intermediates. This flexibility is crucial for mitigating risks associated with geopolitical disruptions or regional supply constraints.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method is inherently designed for industrial viability. The absence of high-pressure hydrogen gas removes a major bottleneck for scale-up, as standard glass-lined or stainless steel reactors can be used without modification. From an environmental perspective, the replacement of chlorinated solvents like chloroform with methanol and ethyl acetate aligns with green chemistry initiatives and stricter environmental regulations. The reduction in waste generation, due to fewer purification steps and higher atom economy, simplifies wastewater treatment and lowers the environmental footprint of the manufacturing site. This compliance advantage is increasingly important for multinational corporations seeking sustainable partners, ensuring that the supply of Agomelatine remains uninterrupted by regulatory crackdowns on polluting processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Agomelatine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN102206170A, offering clarity on how this method compares to established industry standards. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary safety advantages of this Agomelatine synthesis method compared to traditional hydrogenation?
A: Unlike traditional methods utilizing Raney Nickel and high-pressure hydrogen gas which pose significant explosion risks and require specialized high-pressure equipment, this patented process operates at atmospheric pressure using Sodium Borohydride. This eliminates the need for high-temperature and high-pressure reactors, drastically reducing potential safety hazards and equipment costs.
Q: How does the one-pot strategy impact the purity and yield of the final Agomelatine product?
A: The one-pot synthesis strategy minimizes the number of isolation steps, thereby reducing product loss and the formation of impurities associated with multiple work-ups. Experimental data from the patent indicates that this method achieves an average yield of 80.5% with a content purity ranging from 98.8% to 99.6%, significantly outperforming older multi-step routes.
Q: Why is Nickel Chloride Hexahydrate preferred over Raney Nickel in this catalytic system?
A: Nickel Chloride Hexahydrate serves as a more cost-effective and easier-to-handle catalyst precursor compared to pyrophoric Raney Nickel. When combined with Sodium Borohydride in situ, it generates the active catalytic species under mild conditions, avoiding the severe reaction conditions and complex disposal issues associated with traditional heterogeneous hydrogenation catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agomelatine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the competitive landscape of antidepressant therapeutics. Our technical team has thoroughly analyzed the innovations presented in CN102206170A and possesses the expertise to implement this advanced one-pot synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to market availability is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this nickel-catalyzed process, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity Agomelatine intermediates that meet the exacting standards of global regulatory bodies, providing our partners with a secure and high-quality supply source.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their Agomelatine projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your manufacturing costs with our proven expertise in fine chemical synthesis and API intermediate production.
