Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in modern medicinal chemistry. These heterocyclic scaffolds are pivotal in the development of next-generation therapeutics, exhibiting potent anticancer, antifungal, and anti-inflammatory activities, as seen in various kinase inhibitors and antiviral agents. The strategic incorporation of the trifluoromethyl group significantly modulates the physicochemical properties of the parent molecule, improving lipophilicity and membrane permeability without compromising structural integrity. This technological advancement represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, base-promoted cycloaddition strategy.
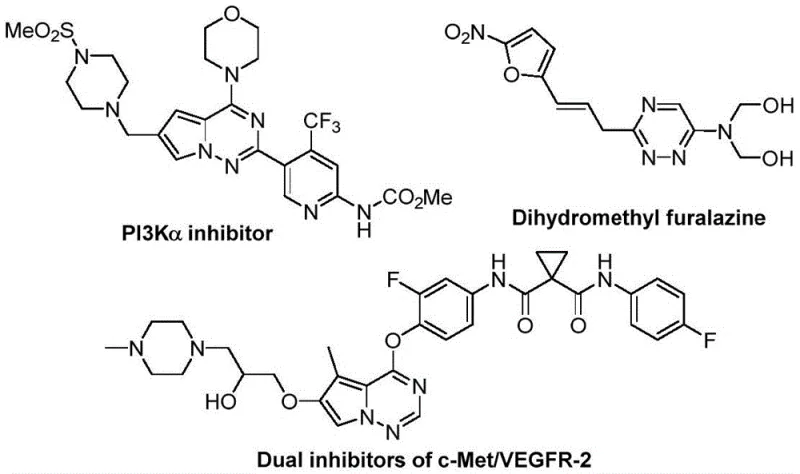
For R&D directors and process chemists, the ability to access diverse trifluoromethyl-triazine libraries rapidly is essential for lead optimization campaigns. The disclosed methodology leverages readily available starting materials—chlorohydrazones and trifluoroacetyl sulfur ylides—to construct the six-membered nitrogenous ring with high atom economy. Unlike legacy methods that often suffer from harsh reaction conditions or limited substrate tolerance, this novel approach operates efficiently under ambient conditions. The versatility of the reaction allows for the introduction of various substituents at the R1, R2, and R3 positions, enabling the rapid generation of structure-activity relationship (SAR) data. This flexibility is crucial for pharmaceutical companies aiming to diversify their pipeline with novel chemical entities that possess superior pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazine derivatives has relied heavily on the condensation of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical pathways have served the community for decades, they are increasingly viewed as suboptimal for modern high-throughput synthesis. Conventional methods frequently necessitate the pre-synthesis of complex reaction substrates, which adds multiple steps to the overall process and reduces the overall yield. Furthermore, many traditional cyclization protocols require elevated temperatures, strong acids, or stoichiometric amounts of oxidizing agents, leading to significant safety hazards and waste generation. The structural diversity achievable through these older routes is often restricted, limiting the chemical space that medicinal chemists can explore. Additionally, the purification of products from these reactions can be arduous due to the formation of polymeric byproducts or difficult-to-remove inorganic salts.
The Novel Approach
In stark contrast, the method described in patent CN116253692A utilizes a synergistic [3+3] cycloaddition between a nitrile imine intermediate and a sulfur ylide, promoted simply by potassium carbonate. This innovative strategy bypasses the need for transition metal catalysts such as palladium or copper, which are not only expensive but also pose significant challenges regarding residual metal limits in final drug substances. The reaction proceeds smoothly at room temperature (20-40°C) in common organic solvents like tetrahydrofuran (THF), eliminating the energy costs associated with heating or cooling. The use of air as the reaction atmosphere further simplifies the operational setup, removing the need for rigorous inert gas protection systems. This streamlined protocol not only accelerates the synthesis timeline but also drastically simplifies the downstream processing, as the byproducts are generally benign and easily separable.
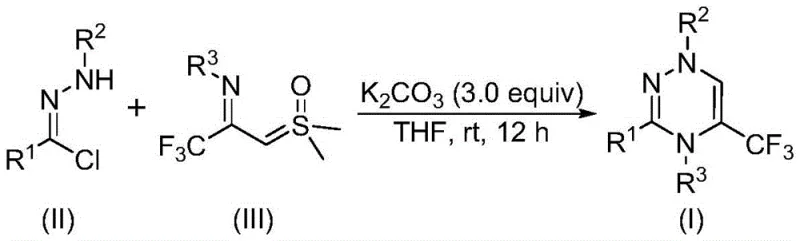
Mechanistic Insights into Base-Promoted [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the in situ generation of highly reactive intermediates under mild basic conditions. Upon addition of potassium carbonate, the chlorohydrazone precursor undergoes dehydrohalogenation to release hydrogen chloride, generating a transient nitrile imine species. This 1,3-dipole is perfectly poised to engage in a concerted cycloaddition with the electron-deficient trifluoroacetyl sulfur ylide. The sulfur ylide acts as a three-atom component, contributing the carbon-sulfur-carbon framework that eventually forms part of the triazine ring after the elimination of dimethyl sulfoxide (DMSO). This [3+3] annulation is highly regioselective, ensuring that the trifluoromethyl group is installed at the desired position on the heterocyclic core. The absence of radical intermediates or high-energy transition states contributes to the clean reaction profile observed in the experimental data, minimizing the formation of regioisomers or oligomeric impurities.
From an impurity control perspective, the choice of potassium carbonate as the promoter is critical. As a mild inorganic base, it effectively drives the elimination of HCl without promoting unwanted side reactions such as hydrolysis of the sensitive imine bonds or decomposition of the sulfur ylide. The reaction's tolerance to air and moisture suggests that the key intermediates are sufficiently stable under ambient conditions, or that the rate of the desired cycloaddition outcompetes potential degradation pathways. This robustness is a significant advantage for scale-up, as it reduces the sensitivity of the process to minor fluctuations in reactor conditions. Furthermore, the structural diversity demonstrated in the patent examples, ranging from simple phenyl groups to bulky naphthyl and heteroaryl substituents, confirms that the steric and electronic properties of the substrates do not significantly hinder the cyclization efficiency.
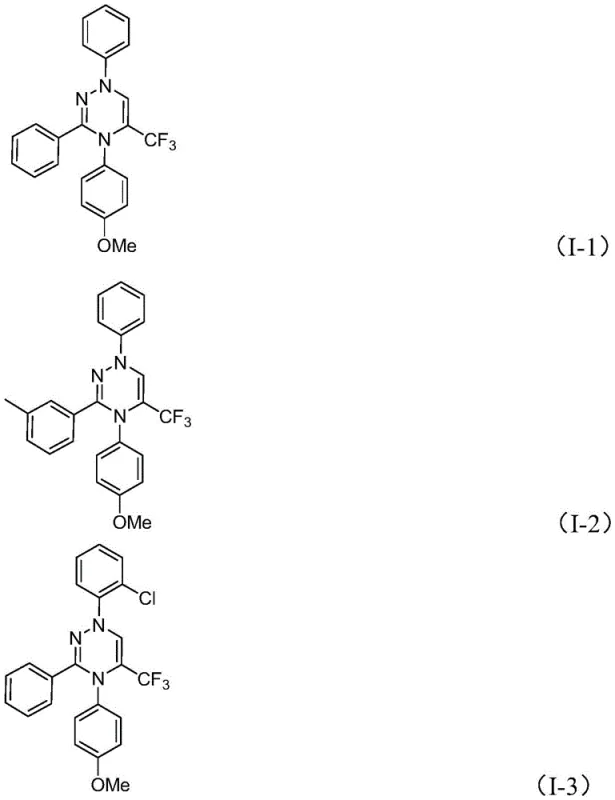
How to Synthesize Trifluoromethyl 1,2,4-Triazines Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow a straightforward protocol that emphasizes precise stoichiometry and adequate mixing. The process begins with the suspension of potassium carbonate in the chosen organic solvent, typically THF, followed by the sequential addition of the chlorohydrazone and the trifluoroacetyl sulfur ylide. Maintaining the reaction temperature between 20°C and 40°C is sufficient to drive the conversion to completion within 10 to 14 hours. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal quenching point, although the patent indicates that extended reaction times do not significantly degrade the product. Upon completion, the solid inorganic salts are removed by filtration, and the filtrate is concentrated. The crude product is then purified using standard silica gel column chromatography to afford the high-purity triazine derivative suitable for biological testing.
- Mix chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate (3.0 equiv) in an organic solvent like THF under air atmosphere.
- Stir the reaction mixture at room temperature (20-40°C) for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits. The elimination of precious metal catalysts removes a major cost driver from the bill of materials, as palladium and rhodium complexes are subject to volatile market pricing and supply constraints. Moreover, the removal of heavy metals from the process flow negates the need for expensive scavenging resins or complex extraction protocols designed to meet strict regulatory limits for residual metals in APIs. This simplification directly translates to reduced manufacturing costs and shorter cycle times. The use of commodity chemicals like potassium carbonate and common solvents like THF ensures a stable and reliable supply chain, mitigating the risk of production delays caused by the shortage of specialized reagents. The operational simplicity of running reactions under air atmosphere further reduces infrastructure costs, as existing reactors can be utilized without modification for inert gas handling.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with inexpensive inorganic bases like potassium carbonate results in substantial raw material savings. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, leading to a lower overall cost of goods sold (COGS). The high atom economy of the [3+3] cycloaddition minimizes waste generation, aligning with green chemistry principles and reducing environmental compliance burdens.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and sulfur ylides, are derived from widely available bulk chemicals such as acyl chlorides, hydrazines, and sulfoxides. This reliance on commodity feedstocks ensures long-term supply security and protects against the volatility associated with specialized fine chemical intermediates. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands without compromising product quality.
- Scalability and Environmental Compliance: The absence of pyrophoric reagents or high-pressure requirements makes this process inherently safer and easier to scale from gram to ton quantities. The mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint. Furthermore, the avoidance of toxic heavy metals simplifies wastewater treatment and solid waste management, facilitating easier regulatory approval and reducing the environmental impact of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazine synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this process for potential adoption. Understanding the nuances of the reaction mechanism and operational parameters is essential for successful technology transfer and scale-up.
Q: What are the primary advantages of this new triazine synthesis method?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild conditions (room temperature, air atmosphere), and uses inexpensive inorganic bases like potassium carbonate, significantly simplifying purification and reducing environmental impact.
Q: What is the reaction mechanism for forming the 1,2,4-triazine ring?
A: The reaction proceeds via a synergistic [3+3] cycloaddition. Potassium carbonate promotes the elimination of HCl from the chlorohydrazone to form a nitrile imine intermediate, which then reacts with the trifluoroacetyl sulfur ylide, followed by the elimination of dimethyl sulfoxide to yield the final heterocycle.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its operational simplicity. It does not require inert gas protection or specialized high-pressure equipment, and the starting materials are commercially available, making it ideal for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in accelerating drug discovery and development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering high-purity trifluoromethyl substituted 1,2,4-triazine compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources of complex building blocks.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall development costs. Let us help you bring your innovative therapies to market faster and more economically.
