Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in modern medicinal chemistry. These heterocyclic scaffolds are pivotal in the development of anticancer, antifungal, and anti-inflammatory agents, as evidenced by their presence in complex drug candidates targeting kinases and other biological pathways. The integration of the trifluoromethyl group is strategically vital, as it modulates electronegativity and lipophilicity, often turning a lead compound into a viable drug candidate. This new methodology represents a significant leap forward, offering a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates.
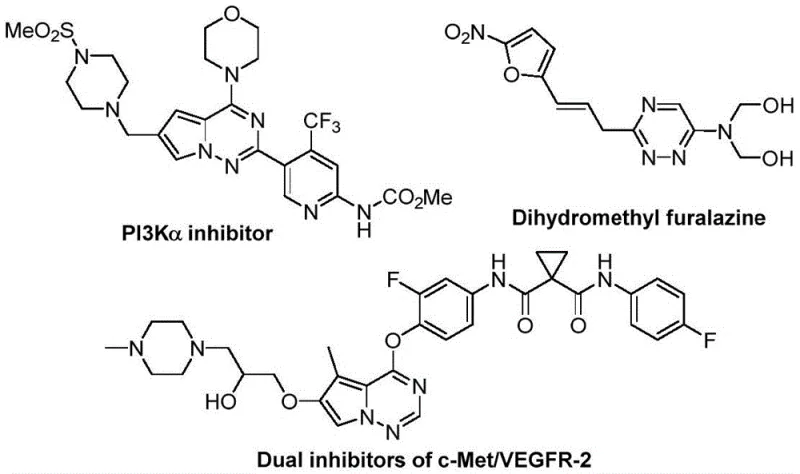
The limitations of conventional methods for synthesizing 1,2,4-triazine compounds have long hindered rapid drug discovery and process optimization. Traditional approaches typically rely on the condensation of amidrazones with 1,2-diketones or alkynes, or multicomponent reactions involving hydrazides and dicarbonyl compounds. These legacy processes frequently suffer from harsh reaction conditions, requiring high temperatures or strong acids that limit functional group tolerance. Furthermore, the structural diversity achievable through these older routes is often poor, restricting the chemical space available to medicinal chemists. In contrast, the novel approach detailed in the patent utilizes a synergistic [3+3] cycloaddition strategy that operates under remarkably mild conditions. By employing chlorohydrazones and trifluoroacetyl sulfur ylides as key building blocks, this method bypasses the need for pre-functionalized substrates that are difficult to synthesize.
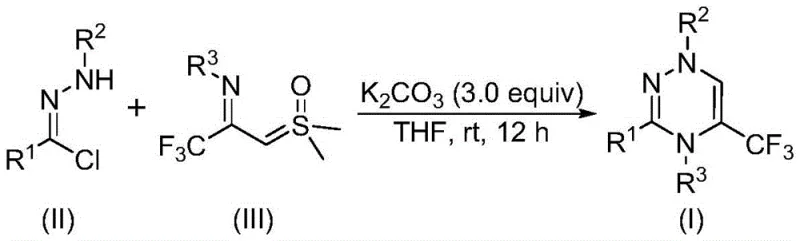
The mechanistic insights into this potassium carbonate-promoted cyclization reveal a sophisticated yet operationally simple transformation. The reaction initiates with the base-mediated dehydrohalogenation of the chlorohydrazone, generating a reactive nitrile imine intermediate in situ. This dipolar species then engages in a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, a unique reagent that serves as both a carbon source and a leaving group provider. The subsequent elimination of dimethyl sulfoxide drives the aromatization of the ring, yielding the stable 1,2,4-triazine core. This mechanism is particularly advantageous for impurity control; because the reaction proceeds at room temperature without transition metals, there is minimal risk of metal-catalyzed side reactions or decomposition of sensitive functional groups. The absence of heavy metals also simplifies the downstream purification process, ensuring that the final API intermediates meet stringent regulatory standards for residual metals.
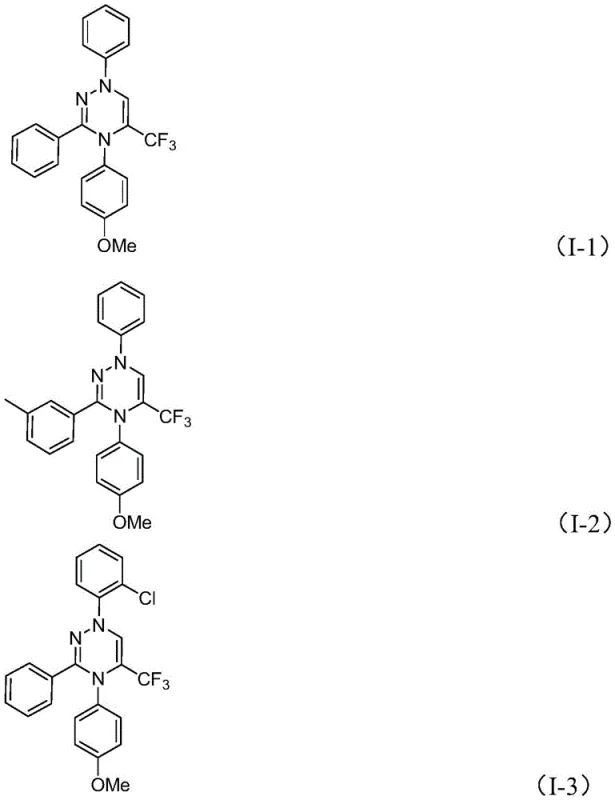
For research and development teams looking to implement this technology, the protocol is designed for ease of execution and reproducibility. The synthesis involves mixing the chlorohydrazone and trifluoroacetyl sulfur ylide in a polar aprotic solvent such as tetrahydrofuran, with potassium carbonate acting as the promoter. The reaction is allowed to stir at ambient temperature for a period of 10 to 14 hours, after which standard workup procedures involving filtration and silica gel chromatography afford the pure product. This straightforward workflow eliminates the need for specialized equipment like gloveboxes or high-pressure reactors, making it accessible for laboratories of all sizes. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below, providing a clear roadmap for replicating these high-yielding transformations in your own facility.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive transition metal catalysts, such as palladium or copper, which are common in traditional cross-coupling or cyclization reactions, results in a drastic reduction in raw material costs. Moreover, the reliance on potassium carbonate, an inexpensive and non-toxic inorganic salt, further lowers the cost of goods sold (COGS). The simplicity of the reaction conditions—operating effectively at room temperature and under an air atmosphere—means that energy consumption is minimized, and the process does not require costly inert gas infrastructure. These factors combine to create a highly cost-effective manufacturing process that enhances the overall economic viability of producing complex heterocyclic intermediates.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts from the synthetic route eliminates the need for expensive scavenging resins and complex purification steps required to meet strict ppm limits for metal residues in pharmaceuticals. This simplification of the downstream processing significantly reduces solvent usage and waste disposal costs, leading to substantial overall savings in the manufacturing budget. Additionally, the high atom economy of the cycloaddition reaction ensures that raw materials are efficiently converted into the desired product, minimizing waste generation.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including various acyl chlorides, hydrazines, and aromatic amines, are commodity chemicals that are readily available from multiple global suppliers. This abundance ensures a stable supply chain, reducing the risk of production delays caused by raw material shortages. The robustness of the reaction, which tolerates a wide range of functional groups and does not require stringent anhydrous or anaerobic conditions, further guarantees consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable from gram to multi-kilogram levels without loss of efficiency, making it suitable for commercial-scale production. The use of benign reagents and the avoidance of toxic heavy metals align perfectly with green chemistry principles and increasingly strict environmental regulations. This compliance reduces the regulatory burden on manufacturers and facilitates faster approval times for new drug applications, while the mild reaction conditions ensure safe operation in large-scale reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific project needs. The answers provided reflect the proven capabilities of the method as described in the intellectual property documentation, ensuring accuracy and reliability for decision-makers.
Q: What are the primary advantages of this new triazine synthesis method?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild conditions (room temperature, air atmosphere), and uses inexpensive inorganic bases like potassium carbonate, significantly simplifying purification and reducing environmental impact.
Q: What is the reaction mechanism for forming the 1,2,4-triazine ring?
A: The reaction proceeds via a synergistic [3+3] cycloaddition. Potassium carbonate promotes the elimination of HCl from the chlorohydrazone to form a nitrile imine intermediate, which then reacts with the trifluoroacetyl sulfur ylide, followed by the elimination of dimethyl sulfoxide to yield the final heterocycle.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its operational simplicity. It does not require inert gas protection or specialized high-pressure equipment, and the starting materials are commercially available, making it ideal for kilogram-to-ton scale manufacturing.
- Mix chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate (3.0 equiv) in an organic solvent like THF under air atmosphere.
- Stir the reaction mixture at room temperature (20-40°C) for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl triazine compound.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in accelerating drug discovery pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of clinical trials or preparing for market launch. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of trifluoromethyl-substituted 1,2,4-triazine we deliver meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for multinational pharmaceutical companies seeking dependable sources of complex building blocks.
We invite you to contact our technical procurement team to discuss how this innovative metal-free synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient route. We are ready to provide specific COA data and route feasibility assessments tailored to your unique molecular targets, helping you optimize your development timeline and reduce overall project costs.
