Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
The efficient construction of fluorinated heterocycles remains a critical challenge in modern medicinal chemistry, particularly for the development of robust pharmaceutical intermediates. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing long-standing issues regarding safety and cost in heterocyclic synthesis. This technology leverages a base-promoted cyclization strategy that eliminates the need for transition metal catalysts and hazardous azide reagents, offering a streamlined pathway for producing high-purity triazole derivatives. For R&D directors and procurement specialists, this innovation represents a significant shift towards safer, more sustainable manufacturing protocols that do not compromise on yield or structural diversity. The ability to access these privileged scaffolds without complex purification steps for heavy metal removal is a distinct advantage in the competitive landscape of API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary methodologies, both of which present substantial operational hazards and economic inefficiencies. The first common approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation; this route necessitates the handling of toxic and potentially explosive azide compounds, posing severe safety risks in large-scale operations. Furthermore, the use of copper catalysts introduces the burden of rigorous downstream processing to remove residual heavy metals to meet stringent pharmaceutical purity standards. The second traditional method utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly suffers from the inherent instability and danger associated with azide reagents. These legacy processes often result in higher production costs due to specialized safety infrastructure requirements and complex waste treatment protocols for metal-containing effluents.
The Novel Approach
In stark contrast to these hazardous legacy methods, the technology disclosed in CN113121462B utilizes a metal-free, base-promoted reaction between readily available diazo compounds and trifluoroethylimidoyl chlorides. As illustrated in the reaction overview below, this novel pathway completely bypasses the use of organic azides and transition metal catalysts, fundamentally altering the safety profile of the synthesis. 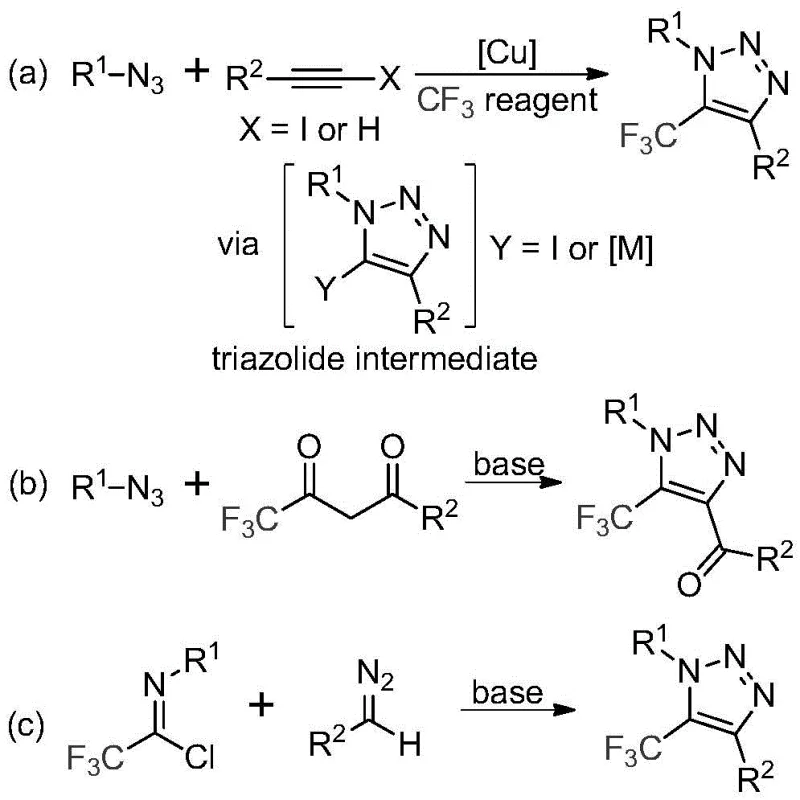
This approach not only mitigates the risk of explosion but also simplifies the supply chain by relying on stable, commercially accessible starting materials. The elimination of copper catalysts means that the costly and time-consuming steps associated with metal scavenging and validation are rendered unnecessary, directly translating to reduced manufacturing lead times. By shifting the mechanistic paradigm from metal-catalyzed cycloaddition to base-mediated cyclization, this method offers a cleaner, more atom-economical route that is highly attractive for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Base-Promoted Cyclization
The core of this innovative synthesis lies in a sophisticated base-promoted mechanism that facilitates the formation of the triazole ring through a sequence of nucleophilic additions and intramolecular cyclizations. The reaction initiates with the deprotonation of the diazo compound by cesium carbonate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular nucleophilic addition-elimination process promotes the critical carbon-carbon bond formation required for the scaffold assembly. Following this initial coupling, the intermediate undergoes a spontaneous intramolecular 5-endo-dig cyclization, driven by the electronic properties of the trifluoromethyl group and the stability of the resulting aromatic triazole system. This mechanistic pathway is highly efficient, avoiding the high-energy barriers often associated with metal-catalyzed cycles and ensuring high conversion rates under mild thermal conditions.
From an impurity control perspective, this mechanism offers superior predictability and cleanliness compared to radical-based or metal-mediated processes. The absence of transition metals eliminates the formation of metal-complexed side products, while the use of mild inorganic bases like cesium carbonate minimizes the risk of over-reaction or decomposition of sensitive functional groups. The specific reaction scheme below highlights the precise stoichiometry and conditions required to drive this transformation effectively. 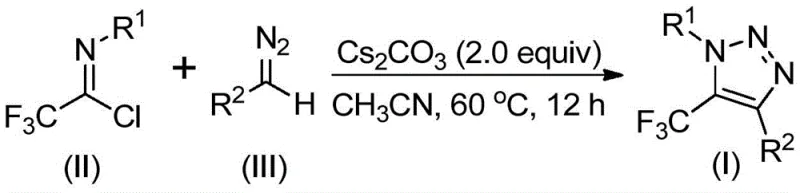
Furthermore, the tolerance for diverse substituents on both the imidoyl chloride and the diazo components allows for the fine-tuning of electronic and steric properties without disrupting the cyclization efficiency. This robustness ensures that the impurity profile remains manageable even when synthesizing structurally complex analogs, providing R&D teams with the confidence to explore broader chemical space for drug discovery programs.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for rapid adoption in process chemistry laboratories. The protocol requires standard laboratory equipment and does not demand inert atmosphere techniques beyond typical moisture sensitivity precautions, making it accessible for most manufacturing facilities. The reaction proceeds smoothly in common aprotic solvents such as acetonitrile, which are easy to recover and recycle, further enhancing the environmental profile of the process. Detailed standardized synthetic steps for implementing this methodology are provided in the guide below.
- Combine cesium carbonate, molecular sieves, trifluoroethylimidoyl chloride, and diazo compounds in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Perform post-treatment by filtration and silica gel mixing, followed by column chromatography purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing expensive and regulated azides and copper catalysts with stable diazo compounds and imidoyl chlorides, companies can secure a more resilient supply chain less susceptible to regulatory bottlenecks. The use of cesium carbonate as a promoter is particularly advantageous, as it is a commodity chemical with stable pricing and global availability, unlike specialized ligands or precious metal catalysts that are subject to volatile market fluctuations. This shift in reagent strategy directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering the bill of materials and reducing the complexity of inventory management.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavengers and the associated analytical validation steps required to prove low residual metal levels in the final API. This streamlining of the downstream processing workflow significantly reduces operational expenditures and shortens the overall production cycle time. Additionally, the high reaction efficiency and yield reported in the patent data suggest that raw material utilization is optimized, minimizing waste generation and maximizing the output per batch. The avoidance of hazardous azides also lowers insurance and safety compliance costs, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Relying on commercially available and stable starting materials such as trifluoroethylimidoyl chloride and diazo compounds ensures a consistent supply flow that is not disrupted by the strict transportation regulations governing explosive precursors. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that the process is less sensitive to minor variations in raw material quality, thereby reducing the rate of batch failures. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients who require just-in-time inventory solutions.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be easily extended to the gram level and beyond, indicating strong potential for multi-kilogram and ton-scale production without significant re-engineering. The use of acetonitrile as a preferred solvent aligns with industry preferences for solvents that are easier to handle and dispose of compared to chlorinated alternatives often used in older methodologies. Furthermore, the metal-free nature of the reaction results in aqueous waste streams that are simpler to treat, facilitating compliance with increasingly stringent environmental regulations regarding heavy metal discharge and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What are the safety advantages of this new triazole synthesis method compared to traditional routes?
A: Unlike conventional methods that rely on toxic and explosive organic azides or copper catalysts, this patent describes a metal-free process using stable diazo compounds and imidoyl chlorides, significantly reducing explosion risks and heavy metal contamination.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states that the method can be easily extended to the gram level and beyond, utilizing cheap and readily available starting materials like cesium carbonate, which supports cost-effective commercial scale-up.
Q: What is the substrate scope for the R1 and R2 groups in this triazole formation?
A: The method demonstrates broad functional group tolerance, accommodating alkyl, substituted or unsubstituted aryl groups for R1, and aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups for R2, allowing for diverse molecular design.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorinated chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates. We are committed to delivering high-purity triazole compounds that empower your drug development programs with speed and reliability.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market through superior chemical innovation.
